20.109(S14):Begin Western protein analysis (Day2)
Introduction
Last time you got familiar with the two cell lines that we will be using during Module 2, and plated a known quantity of each cell type in preparation for protein analysis. Today you will lyse the cells, isolate the protein fraction, and separate the proteins on an acrylamide gel. The teaching faculty will probe the gels for Ku80 expression with a primary antibody, and a week from now you will complete the final steps of this assay and visualize your Western blot.
After completing the Day 1 exercise and hearing the Day 2 lecture, you should be well poised to understand that performing this Western blot serves a true research purpose. The Ku-80 deficient xrs6 cell line can revert to wild-type Ku80 expression under certain circumstances, and thus we want to validate that our current crop of xrs6 are not revertants. Recall that xrs6 have one XRCC5 allele that codes for truncated, non-functional Ku80, but also another, wild-type allele that is normally silenced by methylation. Should this allele become demethylated, the cells will no longer be repair deficient, and we’ll have some really boring flow cytometry results! Of course, even if the xrs6 strain couldn’t revert, we would want to sneak a Western into 20.109 for educational purposes. But isn’t it nice to know that you can formally motivate this experiment for your research article?
A great way to identify a specific protein from a complex mixture is to exploit antibodies – also called immunoglobulins. We will use antibodies to perform a Western blot in this module, and to perform an enzyme-linked immunosorbent assay (ELISA) during Module 3. We'll learn more about the structure of antibodies, and different contexts in which they are used, during Module 3. For now, we'll discuss where they come from.
Many species can be used to raise antibodies. Most commonly mice, rabbits, and goats are immunized, but other animals like sheep, chickens, rats and even humans can be used. The protein used to raise an antibody is called the antigen and the portion of the antigen that is recognized by an antibody is called the epitope. Each antibody can recognize only a small portion of its antigen, typically 5 to 6 amino acids. Some antibodies are monoclonal, or more appropriately “monospecific,” and recognize one epitope, while other antibodies, called polyclonal antibodies, are in fact antibody pools that recognize multiple epitopes.
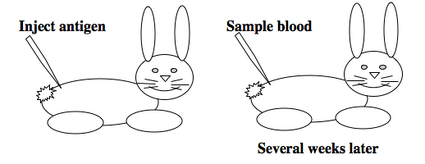
To raise polyclonal antibodies, the antigen of interest is first purified and then injected into an animal. To elicit and enhance the animal’s immunogenic response, the antigen is often injected multiple times over several weeks in the presence of an immune-boosting compound called adjuvant. After some time, usually 4 to 8 weeks, samples of the animal’s blood are collected and the cellular fraction is removed by centrifugation. What is left, called the serum, can then be tested in the lab for the presence of specific antibodies. Even the very best antisera have no more than 10% of their antibodies directed against a particular antigen. The quality of any antiserum is judged by its purity (that it has few other antibodies), its specificity (that it recognizes the antigen and not other spurious proteins) and its concentration (sometimes called its titer). Animals with strong responses to an antigen can be boosted with the antigen and then bled many times, so large volumes of antisera can be produced. However animals have limited life-spans and even the largest volumes of antiserum will eventually run out, requiring a new animal for immunization. The purity, specificity and titer of the new antiserum will likely differ from that of the first batch. High titer antisera against bacterial and viral proteins can be particularly precious since these antibodies are difficult to raise; most animals have seen these immunogens before and therefore don’t mount a major immune response when immunized. Antibodies against toxic proteins are also challenging to produce if they make the animals sick.
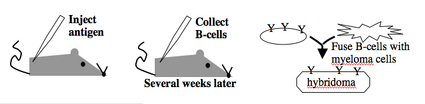
Monoclonal antibodies overcome many limitations of polyclonal pools in that they are specific to a particular epitope and can be produced in unlimited quantities. However, more time is required to establish these antibody-producing cells, called hybridomas, and it is a more expensive endeavor. In this process, normal antibody-producing B cells are fused with immortalized B cells derived from myelomas, and the two cell types are fused by chemical treatment with a limited efficiency. To select only heterogeneously fused cells, the cultures are maintained in medium in which myeloma cells alone cannot survive (often HAT medium). Normal B cells will naturally die out over time with no intervention, so ultimately only the fused cells, called hybridomas, remain. A fused cell with two nuclei can be resolved into a stable cell line after mitosis. Production of stable hybridomas is tedious and difficult but often worth the effort since monoclonal antibodies can recognize covalently-modified epitopes specifically. These are invaluable for experimentally distinguishing the phosphorylated or glycosylated forms of an antigen from the unmodified forms.
For Western analysis, a high quality antibody can have a relatively low affinity for its target protein. This is because the target is localized and concentrated on a blot, allowing the antibody to bind using both antibody “arms” thereby strengthening the association. Even an antibody that is loosely bound to the blot under these circumstances may dissociate then re-associate quickly since the local concentration of the target protein is high. The lower limit for protein detection is approximately 1 ng/lane, a value that varies with the size of the protein to be detected and the Western blotting apparatus that is used. For most acrylamide gels, the protein capacity for each lane is usually 100 to 200 ug (that would be 20 ul of a 5-10 ug/ul protein preparation). Thus 1 ng represents a protein that is approximately 0.001-0.002% of the total cellular protein (1 ng out of 100,000-200,000 ng). Obviously proteins that make up a more significant fraction of the total protein population will be easier to detect.
Protocols
Part 1: Prepare cell lysates
- You will each have an ice bucket at your bench with the following pre-chilled items inside: two empty eppendorfs, RIPA buffer, protease inhibitors, and PBS. Begin by labeling the eppendorf tubes as K1 and xrs6 (plus your section).
- Pick up your cell dish from the incubator in TC, and place it at a 30-45 degree angle tilted downward in your bucket.
- Add 2.5 μL of protease inhibitors to your 250 μL of aliquotted RIPA buffer.
- Aspirate the media from each well and add about 2 mL (very approximate!) of ice-cold PBS per well by pouring.
- Obtain two pre-chilled scrapers from the fridge.
- Aspirate the ice-cold PBS and repeat the wash once more – make sure to remove ALL of the PBS after this wash.
- Add 100 μL of lysis buffer across the top of each well, allowing it to run down the well.
- Collect the cells to the bottom of the well by scraping each well with a fresh cell scraper.
- First rub back and forth with the scraper to coat cells with lysis buffer. Then go from top to bottom windshield wiper style to pool them down towards the bottom of the tilted dish.
- Add the contents of each well to its respective eppendorf tube.
- Incubate the eppendorf tubes on ice for 15 min.
- Meanwhile, take two fresh eppendorf tubes and begin to chill them for a later step.
- Spin the tubes at max speed in the cold room centrifuge for 15 min to pellet insoluble material. Bring your eppendorf tubes to the TA who will spin them for you.
- This step is typically referred to as "clearing" the lysate.
- Transfer the supernatant to the new set of eppendorf tubes – be careful not to disturb the pellet at the bottom! Keep these samples on ice when not directly in use.
Part 2: Measure protein content
You will now measure the total protein concentration in each cell lysate to determine the volume required to evaluate equal protein amounts by Western blot. We are using the Precision Red Advanced Protein Assay from Cytoskeleton.
- Right before it is your turn to use the spectrophotometer, add 10 μL of cell lysate to a plastic cuvette: prepare K1, xrs6, and a blank "lysate" using your leftover RIPA buffer.
- Be careful not to allow 10 μL of lysate to sit in the cuvettes for more than a couple minutes before starting the next step -- it will dry up!
- Bring the cuvettes to the spectrophotometer and add 990 μL of Precision Red reagent. Mix by pipetting up and down a couple times without introducing bubbles.
- After 1 minute, measure each sample at 600 nm, using the RIPA sample as a blank.
- Calculate the two stock protein concentrations using the following information
- 1 absorbance unit = 100 ug protein/mL reagent / cm
- the path length of the spec is precisely 1 cm
- don't forget to account for the dilution factor
- Next, calculate the volumes of lysate and water required to add 20 μg of total protein to the SDS-PAGE gel in a total volume of 20 μL, per each lysate.
- if your concentration is greater than 1μg/μL, use water to make up the remaining volume
- if your concentration is less than 1μg/μL for at least one sample, scale both samples down to a lower amount, such as 10 μg
- Do not throw away the remainder of your cell lysate! We will store these samples for you at -80 °C in case another Western blot needs to be completed.
Part 3: Separate proteins by SDS-PAGE
Two teams will share one gel.
- Pipet 15 μL of pre-stained Kaleidoscope ladder into a labeled tube. It will be used to track gel progress.
- Working inside the hood, add 4 μL of 6X Laemlli sample buffer to each of the two lysates (but not to the ladder). Briefly vortex each tube, then quick-spin the solution back down.
- Toss your β-mercaptoethanol-soaked tips into the empty pipette tip box inside the hood so they can air out for a while before being dumped in a burn box. Humans can sense this stinky compound at the order of one part per million!
- Put lid locks on the eppendorf tubes – including the ladder – and boil for 5 minutes in the water bath that is in the fume hood. Repeat the vortex and quick-spin step.
- Meanwhile, get trained in groups of 2-4 people about how to load a protein gel.
- When your samples are ready, you should load them according to the scheme below.
- It is okay to let your samples boil 2-3 extra minutes if there is no gel box free, but don't boil them more than 10 min total.
- It is also okay to let them cool a couple of minutes.
- In your notebooks, document the starting and stopping time of electrophoresis, which will be initiated by the teaching faculty at 200 V and run for 30-40 minutes
| Lane | Sample | Volume to load |
|---|---|---|
| 1 | Group 1, K1 | 20 ul |
| 2 | Group 1, xrs6 | 20 μL |
| 3 | "Kaleidoscope" protein molecular weight standards | 10 μL |
| 4 through 6 | BLANK | N/A |
| 7 | "Kaleidoscope" protein molecular weight standards | 10 μL |
| 8 | Group 2, xrs6 | 20 μL |
| 9 | Group 2, K1 | 20 μL |
| 10 | BLANK | N/A |
Part 4: Transfer proteins to membrane
- Wearing gloves, disassemble the electrophoresis chamber.
- Blot the gel to nitrocellulose as follows:
- Place the gray side of the transfer cassette in a tupperware container which is half full of transfer buffer. The transfer cassette is color-coded so the gray side should end up facing the cathode (black electrode) and the clear side facing the anode (red).
- Place a pre-soaked ScotchBrite pad on the gray side of the cassette.
- Place 1 piece of filter paper on top of the ScotchBrite pad.
- Place your gel on top of the filter paper.
- Place a piece of nitrocellulose filter on top of the gel. The nitrocellulose filter is white and can be found between the blue protective paper sheets. Wear gloves when handling the nitrocellulose to avoid transferring proteins from your fingers to the filter.
- Gently but thoroughly press out any air bubbles caught between the gel and the nitrocellulose.
- Place another piece of filter paper on top of the nitrocellulose.
- Place a second pre-soaked ScotchBrite pad on top of the filter paper.
- Close the cassette, then push the clasp down and slide it along the top to hold it shut.
- Place the transfer cassette into the blotting tank so that the clear side faces the red pole and the gray side faces the black pole.
- Two blots can be run in each tank. When both are in place, insert the ice compartment into the tank. Fill the tank with buffer. Connect the power supply and transfer at 100 V for one hour.
- After an hour, turn off the current, disconnect the tank from the power supply, and remove the holders. Retrieve the nitrocellulose filter and confirm that the pre-stained markers have transferred from the gel to the blot. Cut the blot next to each ladder so that you end up with two blots, each only three or four lanes wide (not losing track of whose side is whose!), and then move each mini-blot to blocking buffer and store it in the refrigerator until next time.
The teaching staff will do step #4 for you if it runs past 5 pm.
For next time
- In preparation for next time, read the paper from Zhang lab about Ku80 in the context of lung cancer. You do NOT need to make a figure this time, but you DO need to review the guiding questions in the Day 3 protocol and be ready to participate in a discussion centered around them.
- During Module 1, we let you omit a methods section from your slide deck summary… but this blissful state of affairs cannot last forever! For next time, you will take a first stab at writing up the methods for your Module 2 research article. This early draft will include just the content/procedures from Days 1 and 2 of lab.
- Be sure to read the Methods section guidelines at this link before you begin; doing so may save you some effort.
- As you compose your methods for the first two days, do your best to think ahead about the scope of the experiment and how D1 and D2 fit into that overall context. For example, you probably want to establish your cell strains and general culture conditions just once.
- Recall that the Module 2 assignment will be done individually and with no formal revision, so it's even more important than before that you (a) complete each FNT and (b) put forth your best effort – in order to get meaningful feedback that you can use later on.
- Finally, recall that your primer design memo is due by 11 AM on the day of your next lab session (Tuesday or Wednesday depending on section).
Reagent list
Had been missing transfer buffer!
- Transfer Buffer
- 25 mM Tris
- 192 mM Glycine
- 20% v/v Methanol
From Boston Bioproducts unless otherwise noted:
- RIPA Lysis Buffer
- 50 mM Tris-HCL, pH 7.4
- 150 mM NaCL
- 1% NP-40
- 0.5% Sodium deoxycholate
- 0.1% SDS
- 100X Protease Inhibitor cocktail
- AEBSF
- Aprotinin
- E-64 Besstain Leupeptin
- EDTA
- Precision Red Advanced Protein Assay (Cytoskeleton, Inc.)
- 6x Reducing Laemlli Sample Buffer
- 375 mM Tris HCL, pH 6.8
- 9% SDS
- 50% Glycerol
- 9% Betamercaptoethanol
- 0.03% Bromophenol blue
From Bio-Rad
- 4-20% Mini-PROTEAN TGX gel
- TGS Buffer: 25 mM Tris, 192 mM glycine, 0.1% (w/v) SDS, pH 8.3
- Kaleidoscope marker sizes here
Navigation Links
Next Day: Choose system conditions and paper discussion Previous Day: Introduction to cell strains and plating
