Biomod/2013/UT-Austin/Design
<html> <style type="text/css"> // </style>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin"><img src="http://openwetware.org/images/2/2f/G4262.png" width ="50%" height="50%"/>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin/Introduction"><img src="http://openwetware.org/images/0/0d/Intro.png" width="20%" alt="" /></a>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin/Design"><img src="http://openwetware.org/images/0/0a/Design.png" width="10%" alt="" /></a>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin/Methods"><img src="http://openwetware.org/images/0/07/MEthods.png" width="15%" alt="" /></a>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin/Results"><img src="http://openwetware.org/images/1/17/Results.png" width="12%" alt="" /></a>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin/Team Info"><img src="http://openwetware.org/images/3/32/Team.png" width="15%" alt="" /></a>
<a href="http://openwetware.org/wiki/Biomod/2013/UT-Austin/References"><img src="http://openwetware.org/images/0/0d/References.png" width="20%" alt="" /></a>
</html>
Design
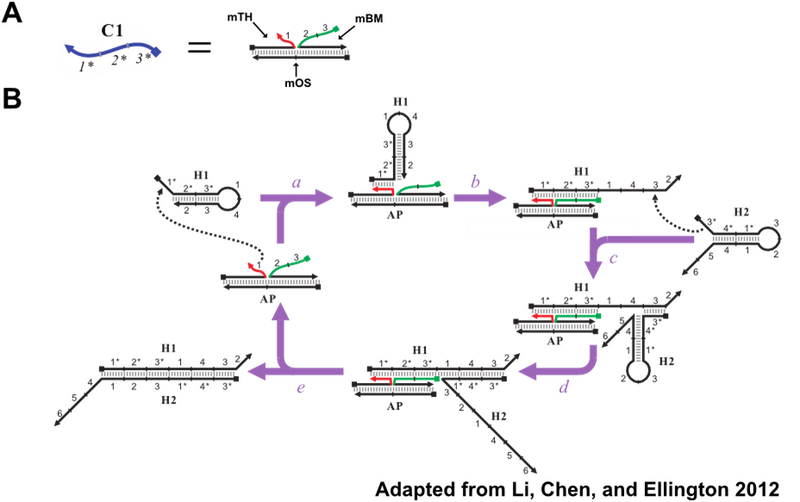
The CHA reaction described in the introduction allows for the detection of oligonucleotides. The oligo detected is C1, Figure 8A. C1 has 3 domains: the toehold binding domain (1*) and the domains that undergo branch migration in order to open H1. However, previous work has shown that these catalytic domains do not have to be found on a single oligo. These domains can be split between separate oligos and still trigger CHA when colocalized on a common template, Figure 8B. This modified variant of CHA will be referred to as split catalyst CHA (scCHA) in this work.
Further Modifications of the Catalyst
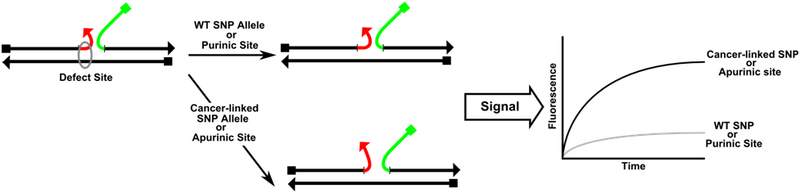
The scCHA catalyst can be further modified in order to detect minor structural defects in mOS. By including limited complementarity, 1-2 bp, between the 1 domain of mTH and mOS, the rate of catalysis can be greatly reduced, Figure 9. When the 1 domain is partially annealed to mOS it is hindered from annealing to the toehold on H1. However, if a defect is present in mOS in this region of complementarity, the 1 domain will be freed to function normally and the rate of catalysis is restored. For our purposes this defect can be an SNP allele or an AP site. mOS is the target sequence to be detected and characterized by our circuit.
MDM2 Circuit Design
In order to have a circuit that would specifically detect and characterize SNP309 of the MDM2 gene, new CHA circuit pieces were designed using CircDesigNA. Hairpins were designed to not interact with the MDM2 gene in the absence of the split catalyst pieces, Table 1. The split catalyst was designed as depicted in Figure 10. The MDM2 gene sequence determined a majority of the split catalyst sequences and the remaining sequences were designed by CircDesigNA.

| Oligo Name | Description | Sequence (5' to 3') |
|---|---|---|
| MDM2 | ||
| BioM_MDM2_Cat_1,2,3p1 | Continuous catalyst for testing circuit hairpins | CTTCAAACCCTAAATCGACACAAC |
| BioM_MDM2_Cat_1p1 | Split catalyst piece containing domain 1 | CACCTGCGATCATCCGGACCTCCCGCGCCGACACAAC |
| BioM_MDM2_Cat_2,3p1 | Split catalyst piece containing domains 2 and 3 | CTTCAAACCCTAAATCGCGGCCCCGCAGCCCCCGGCCCCCGTGAC |
| BioM_MDM2_H1p1 | Hairpin 1 for MDM2 detection and characterization | GTTGTGTCGATTTAGGGTTTGAAGCTCTCTCCCTTCAAACCCTAAATCCCTCCCTCCCTCCCTC |
| BioM_MDM2_H2p1 | Hairpin 2 for MDM2 detection and characterization | GTTTGAAGGGAGAGAGCTTCAAACCCTAAATCCTCTCTCC |
| BioM_MDM2_RepFp1 | Flurophore tagged reporter oligo for MDM2 detection and characterization | /56-FAM/GAGGGAGGGAGGGAGGGATTTAGG |
| BioM_MDM2_RepQp1 | Quencher tagged reporter oligo for MDM2 detection and characterization | CCTCCCTCCCTCCCTC/3IABkFQ/ |
| Apurinic Site | ||
| BioM_Cat_1,2,3p1 | Continuous catalyst for testing circuit hairpins | CTTTTCTGCATCTATCTCCTAACC |
| BioM_Cat_1p1 | Split catalyst piece containing domain 1 | GAACCCCCGGTTCCTCTCCTAACC |
| BioM_Cat_2,3p1 | Split catalyst piece containing domains 2 and 3 | CTTTTCTGCATCTATCGTACTGAGGGGGCGTA |
| BioM_H1p1 | Hairpin 1 for AP site detection and characterization | GGTTAGGAGATAGATGCAGAAAAGCAATTGTCCTTTTCTGCATCTATCGAAGTAAGGTAGTGTG |
| BioM_H2p1 | Hairpin 2 for AP site detection and characterization | CAGAAAAGGACAATTGCTTTTCTGCATCTATCCAATTGTC |
| BioM_RepFp1 | Flurophore tagged reporter oligo for AP site detection and characterization | /56-FAM/GTAGTGTGGAAGTAAGGATAGATG |
| BioM_RepQp1 | Quencher tagged reporter oligo for AP site detection and characterization | CTTACTTCCACACTAC/3IABkFQ/ |
Apurinic Circuit Design
The apurinic circuit was designed in the same fashion as the MDM2 circuit. The catalyst pieces were designed to assemble on the A4324 loop of the 28S rRNA, Figure 11. The remaining circuit pieces for the apurinic design can be found in Table 1.
