User:Peter Giang/Notebook/Biology 210 at AU
March 19, 2015 - Embryology & Zebrafish Development
Purpose
The purpose of this experiment is to learn the stages of embryonic development, compare embryonic development in different organisms, and develop an experiment to study how environmental conditions affect embryonic development. Many teratogens exist that affects developing embryos during this extremely sensitive time in their growth cycle. Today we will look specifically at the substrate fluoride and its role on embryogenesis. We hypothesize that embryos under the influences of fluoride will be greatly inhibited in their embryonic development and those unexposed to this mineral will be unaffected.
Materials and Methods
• Fluoride (10mg/ml)
• Deer Park Water
• 20 Healthy Tranlucent Zebrafish Embryos
• Incubator
• Pipette
• Two Covered Petro Dishes
• Microscope
• Paraformaldehyde
• Paramecium
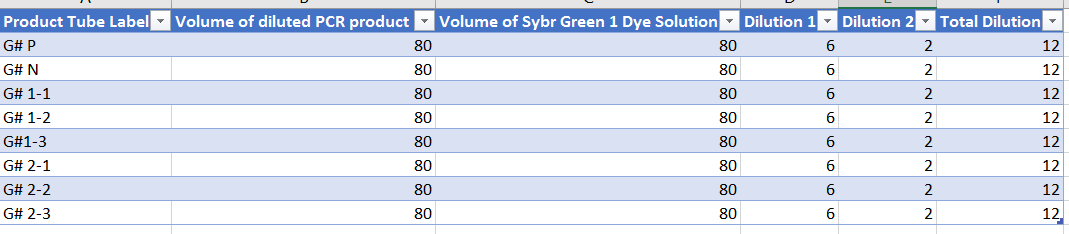
Zebrafish Larvae Observation Summary
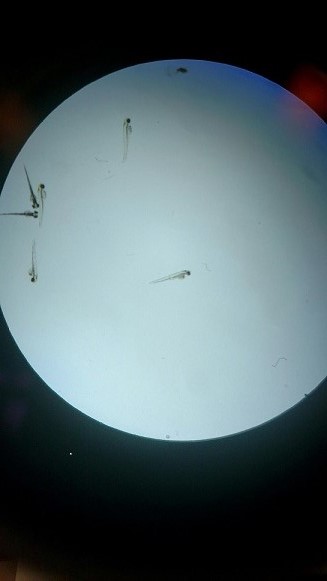
Day 1: 20 Healthy Translucent Zebrafish Embryoes [18-36 Hours Old]
We began this experiment with 20 healthy translucent zebrafish embryos (20 per petri dish) which are seen (as shown in above) to be small (~1mm in diameter), oblong and spherical in shape, translucent, nonmotile, and free from debris, hatchlings, or casings. We speculate that based on this observation these embryos are most likely in their high to oblong transition stage of embryonic development. Two experiment groups were staged: one group as a control where Deer Park exclusively water was given and another where our independent variable fluoride (10mg/ml concentration) was given.
Day 4: Zebrafish Embryoes Hatch and Begin Motility
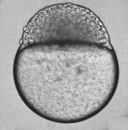
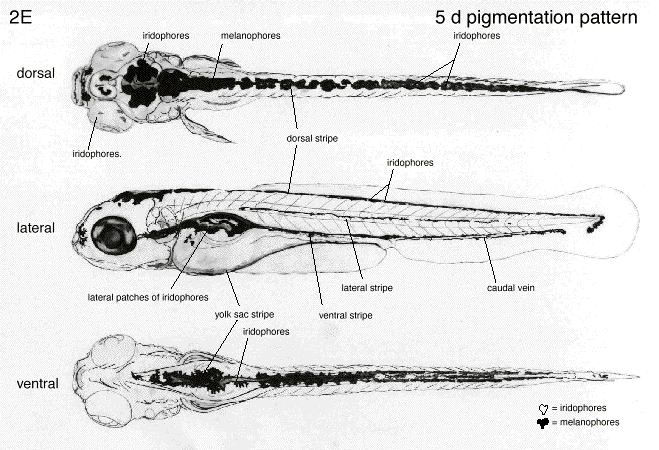
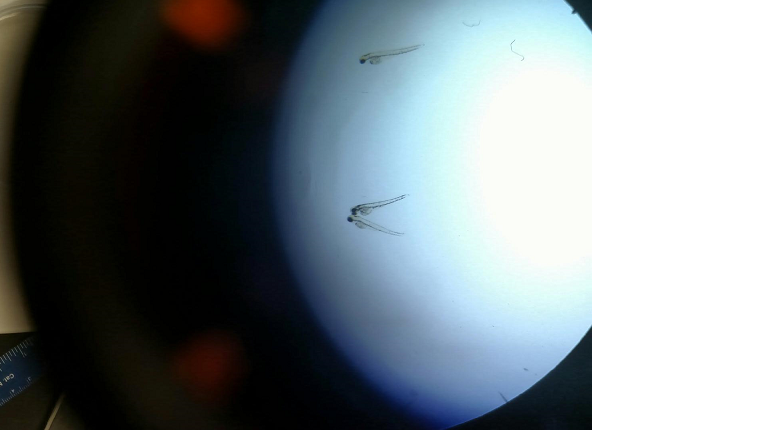
Our embryos that we started with have now begun to grow to their near adult size on day 4. This was evident not only by the large amount of empty hatchlings and casings that were collected, but also by the zebrafish's now new ability of motility as they were seen swimming aimlessly around our petri dish. These embryos are now stages to be in their 36-48 hour year old stage of embryonic development. As we examine closer under the 4x objective, we can see the clear formation of the body and delineating features of tail pigmentation (melanophores). Eyes and eye movement are also seen, along with other new developmental features including the eyes, gills, heart, pectoral fin, yolk sac, swim bladder, and the mouth. When accessing general movement, we used an external stimuli (i.e. suction bulb) to initiate the startle reflex which is highly intact.
Day 7: Zebrafish Embryoes: Few Left Viable
In the span of only 3 days, the healthy zebrafish in both our control and experiment group, are now seen to be mostly deceased with exception to one and two in our experimental and control groups respectively. Those few left intact have now come to there own in full length and growth (i.e. eyes, fin, mouth, yolk sac. etc), but external stimuli via the suction bulb has a less dramatic response by those still left viable. We did what we could to optimize survival by replenish water, removing debris and dead zebrafish carcass, along with providing a food sources of Paramecium in each dish.
Day 11: Final Day of Our Observation
Today in our final day of observation, we came to lab with a startling revelation: all of our zebrafish have now died.
Conclusion
In this experiment we sought to examine the effects of our test variable fluoride on the embryonic development of a zebrafish. We initially hypothesize that fluoride would have a dramatic impact affecting not only embryonic growth during its early stages, but continue to alter growth in this organism as it develops into adulthood. The findings drawn from the conclusion of this study disproved this initial hypothesis. We saw in fact the opposite to be true: more zebrafish were alive in the final days of this experiment under the exposure of fluoride that those without having had this exposure. Looking at studies done in the past under similar conditions, researchers found that there was a latent period of up to seven days post-fertilization or even possibly longer, where little to no morphological changes were seen. It would not be surprising then to find similar conclusions drawn then from our own experiment as we saw neither control nor experiment exhibiting any contrasting changes in morphology or behavior. What conclusions then can we draw from these findings? It is important to note that during each step in the experiment there was a great deal where human error may have inadvertently altered the integrity of this study, for instance, during one of the lab an accidental drop of both our dishes lead to both the contamination and death of our embryos about day three into our experiment. Other instances for error may have been the inaccurate measurement of fluoride concentration making the solution either too weak or too strong. Looking forward, we would welcome the opportunity to repeat this experiment but under different conditions. We would like to standardize our environment: pH, light, temperature, noise, both water and fluoride concentrations, food source, etc in order to optimize growth to greater than seven days in order see what long term effects fluoride may have on the developing embryo. For example we know that the mouth and inner oral structure of a zebrafish usually develops later in its evolving stage. Studying this single characteristic feature then would allow us to better understand both the short and long term effects to prolong exposure to this element.
March 5, 2015 - Identifying Bacteria Through Constructing PCR for 16S Sequencing
Purpose
The purpose of this lab is to identify bacteria from our Hay infusion (obtained from our marsh transect) through establishing a PCR reaction, that is selectively amplifying the 16S rRNA gene (a well-studied primer in the scientific community) and then sequencing its DNA against known species of bacteria. Once we've identified the bacteria, we will be able to get a better understanding of some of its characteristic features including: growth patterns under antibiotic conditions, and cell morphology.
Materials and Methods
• Agar Petri Dish (4)
• Sterile Loop
• Sterile Tube with 100µL of Water
• Incubator
• Centrifuge
• 20µL of Primer/Water Mixture
• 5µL of Supernantant
• Agarose Gel
• Basic Local Alignment Search Tool
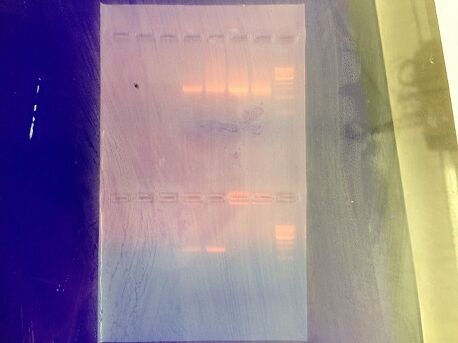
After setting up our PCR reaction and allowing one week for our product to form, we were able to successfully sequence our sample using Basic Local Alignment Search Tool (BLAST) whose results are supported by the National Institute of Health, through using the following sequence:
Step 1: Use https://clims3.genewiz.com/links.aspx?oId=4p6LZ4Isjwk=&ref=00 to retrieve sequencing reactions from the forward primer MB51.
Step 2: Download trace file for MB51 -For_16S by clicking on "View"
Step 3: Sequence File : MB51-For_16S.seq
Step 4: Navigate to BLAST at http://blast.ncbi.nlm.nih.gov/Blast.cgi and enter sequence reaction for forward primer MB51
Step 5: Top results from sequences producing significant alignments:
MB51-For_16S_C07.ab1 GNNNNNNNNNNNNNNNNNNNNNNNTGCAGCCGAGCGGTAGAGATTCTTCGGATCTTGAGAGCGGCGTACGGGTGCGGAAC ACGTGTGCAACCTGCCTTTATCAGGGGGATAGCCTTTCGAAAGGAAGATTAATACCCCATAATATATTGAATGGCATCAT TTAATATTGAAAACTCCGGTGGATAGAGATGGGCACGCGCAAGATTAGATAGTTGGTGAGGTAACGGCTCACCAAGTCTA CGATCTTTAGGGGGCCTGAGAGGGTGATCCCCCACACTGGTACTGAGACACGGACCAGACTCCTACGGGAGGCAGCAGTG AGGAATATTGGACAATGGGTGCGAGCCTGATCCAGCCATCCCGCGTGAAGGACGACGGCCCTATGGGTTGTAAACTTCTT TTGTATAGGGATAAACCTAGATACGTGTATCTAGCTGAAGGTACTATACGAATAAGCACCGGCTAACTCCGTGCCAGCAN CCGCGGTAATACGGAGGGTGCAAGCGTTATCCGGATTTATTGGGTTTAAAGGGTCCGTAGCGGATTTGTAAGTCAGTGNT GAAATCTCACAGCTTAACTGTGAAAACTGCCATTGATACTGCAAGTCTTGAGTGTTGTTGAATANCTGGAATAANTAGTG TANCGGTGAAATGCATANATTTTACTTANAACACCNNTGGCAAGGCNGGTTACTAANCACAACTGACGCTNATGGAN
Chryseobacterium vietnamense strain GIMN1.005 16S ribosomal RNA gene, partial sequence
Conclusion
Chryseobacterium vietnamense bacteria, designated GIMN1.005T, is a gram-reaction-negative bacterial strain, first discovered and isolated from a forest soil sample in Vietnam. This strain was first seen to have characteristics features including its distinct yellow-pigmentation, growth only under aerobic conditions, specifically between 5 °C and 37 °C, and cells that were non-motile, non-gliding and non-spore-forming. Considering it's first origins, it would not be uncommon then for this isolate to also be found from our own marsh (forest-like) transect having very similar types of environmental conditions. Others features such as motility may or may not give further support, simply because their were a myriad of both motile and non-motile cells seen from our transect.
February 24, 2015 - Learning the Wonderful World of Invertebrates
Purpose The purpose of this lab is to study the soil invertebrates from our transect in order to better understand their role and function and how they evolved from simple organ systems to those of higher complexity. We hypothesize that there will be many invertebrates sampled from our leaf litter due to the large and nutrient rich resources from our march transect.
Materials and Methods (Procedure I: Acoelomates, Pseudocoelomates, and Coelomates)
• Dissecting scope
• Wet stain mount
Materials and Methods (Procedure II: Arthropods)
• Various organisms from each of the major classes: ararchnida, diplopoda, chilopoda, insect, and crustacea
Materials and Methods (Procedure III: Analyzing the Invertebrates Collected by the Berlese Funnel)
• Berlese Funnel
• Dissecting Microscope
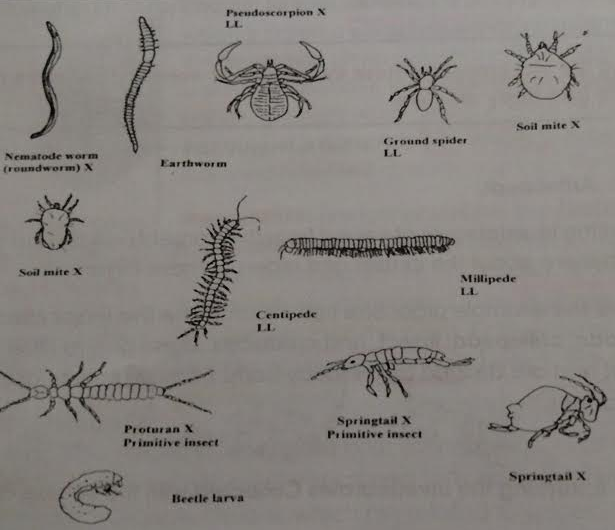
• Figure 3: Common Soil Invertebrates Diagram
• Petri dish
• 10-15mL of Water
• Probe
Data and Observation (Procedure I: Acoelomates, Pseudocoelomates, and Coelomates)
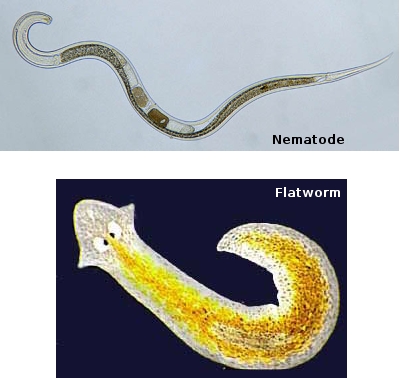
The slender, and semi-transparent body and shape of the Planaria allows this organism to seemingly glide smoothly and evenly over or between environmental surfaces little to no apparent effort powered by cilia. The nematodes is seen with a slightly thinner and greater transparent cross-section allowing it to move faster through water surfaces in a undulating fashion. The last organism in this category, the Annelida has greater percentage of body composition and unlike the both the Planaria and the nematodes, movement involves extending the body, anchoring it to a surface, and contracting body muscles in an almost wavelike contraction/relaxation motion.
Data and Observation (Procedure II: Arthropods)
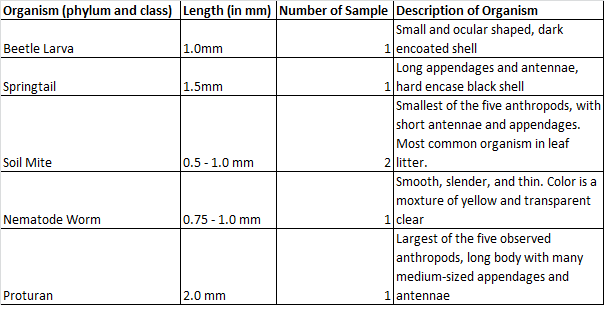
Figure 3: Common Invertebrates Soil Diagram
Organism #1 - Beetle Larva
Organism #2 - Springtail
Organism #3 - Soil Mite
Organism #4 - Nematode Worm
Organism #5 - Protura
Data and Observation (Procedure III: Analyzing the Invertebrates Collected with the Berlese Funnel)
Conclusion and Future Direction
The organisms obtained from our transect unfortunately did not support our initial hypothesis likely due to the extreme cold-weather conditions as many of the organisms inhabiting our transect may have died. It would be interesting to repeat the same experiment during a different time in the year (i.e. fall or summer months) to compare organisms that might be sampled or sample what organisms might be obtained from a different transect source (i.e. lake, pond, stream).
February 03, 2015 - Identifying Bacteria with DNA Sequences
Purpose The purpose of this lab is to better understand the different characteristics of bacteria by observing their morphology: growth and size, shape, motility, antibiotic-resistance, and lastly amplifying their growth (via PCR reactions) to better understand their DNA structure.
Although Archaea microorganisms can generally be found everywhere and are common in soil, I do not believe that this species would have grown in our agar plates because this organism has traditionally been very difficult to culture (Freeman, 2014).
As we observe our Hay Infusion Culture, it's evident that this medium has grown more murky in color, and much more odorous in smell. This makes sense because bacteria grows and reproduces very quickly and so over the span of a week, there was likely a significant growth of various organism contributing to both the smell and appearance in this culture.
There was a noticeably difference in bacterial growth of colonies seen between those that contained tetracycline and those that did not. The agar plates that did not contain tetracycline in the agar medium saw bacteria grew in large numbers to form lawns where hundreds to thousands of bacteria flourished. Agar plates that contained tetracycline in contrast, had significantly less total number of colony formation. The spectrum of activity of tetracyclines encompasses various protozoan parasites including: P. falciparum, Entamoeba histolytica, Giardia lamblia, Leishmania major, Trichomonas vaginalis, and Toxoplasma gondii (Chopra, I., & Roberts, M. (2001). Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiology and Molecular Biology Reviews, 65(2), 232–260. doi:10.1128/MMBR.65.2.232-260.2001)
Tetracyclines mechanism of action works by entering the bacterial cell wall in two ways: passive diffusion and an energy-dependent active transport system, mediated in a pH-dependent fashion. Once inside the cell, tetracyclines bind reversibly to the 30S ribosomal subunit at a position that blocks the binding of the aminoacyl-tRNA to the acceptor site on the mRNA-ribosome complex. Protein synthesis is ultimately inhibited, leading to a bacteriostatic effect (Chopra, I., & Roberts, M. (2001). Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiology and Molecular Biology Reviews, 65(2), 232–260. doi:10.1128/MMBR.65.2.232-260.2001)
Materials and Methods
Hay Infusion Culture, Wet Mount Slides, Agar Plates (4), Inoculating Loop, Microscope, Staining Tray, Permanent Marker, Crystal Violet Stain, Gram Iodine Mordant, Water Bottle, Kimwipe, Sterile Tube, Centrifuge, PCR primer, Pipette, 95% alcohol
Finally able to get access to OWW! This forum is new to me, but I think I will enjoy it. Gone are the days of handwritten lab manuals..
PG