User:Pedrobeltrao/Notebook/Structural analysis of phosphorylation sites
IntroductionThe main objective of this study is to try to find systematic ways to study how phosphorylation might regulate proteins based on the analysis of their structures/domains. Only 15% of the known phosphorylation sites of S. cerevisiae map to known folded domains but we can try to find how these generally regulate domain activity, interfaces and how are they conserved in 3D space instead of sequence space.
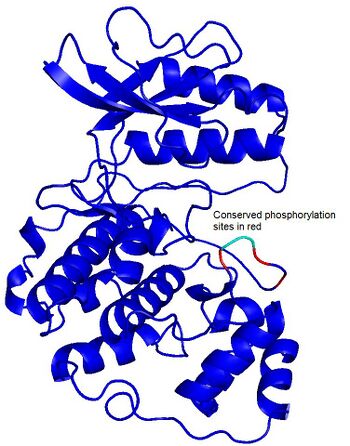 To the right we show an example of conserved phosphorylation sites in the MAPK Hog1 of S. cerevisiae. The conserved phosphorylation events in orthologous proteins of other species are shown in red. We think this sort of analysis might help to identify novel ways in which domains are regulated by phosphorylation and how this regulation changes during evolution Ongoing work
Ressources | |