User:Odom:Schmidt
|
Odom laboratory
|
 |
email: Dominic.Schmidt487 'at' gmail 'dot' com |
Qualifications and Personal History
Education
|
Research
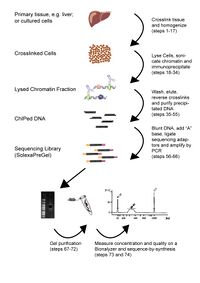
Research accomplishmentsCTCF-binding locations represent regulatory sequences that are highly constrained over the course of evolution. To gain insight into how these DNA elements are conserved and spread through the genome, we defined the full spectrum of CTCF-binding sites, including a 33/34-mer motif, and identified over five thousand highly conserved, robust, and tissue-independent CTCF-binding locations by comparing ChIP-seq data from six mammals. Our data indicate that activation of retroelements has produced species-specific expansions of CTCF binding in rodents, dogs, and opossum, which often functionally serve as chromatin and transcriptional insulators. We discovered fossilized repeat elements flanking deeply conserved CTCF-binding regions, indicating that similar retrotransposon expansions occurred hundreds of millions of years ago. Repeat-driven dispersal of CTCF binding is a fundamental, ancient, and still highly active mechanism of genome evolution in mammalian lineages.: 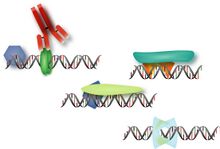 Transcription factors (TFs) direct gene expression by binding to DNA regulatory regions. To explore the evolution of gene regulation, we experimentally determined the genome-wide occupancy of two TFs, CEBPA and HNF4A, in livers of five vertebrates. Although each TF displays highly conserved DNA binding preferences, most binding is species-specific, and aligned binding events present in all five species are rare. Regions near genes with expression levels dependent on a TF are often bound by the TF in multiple species, yet show no enhanced DNA sequence constraint. Binding divergence between species can be largely explained by sequence changes to the bound motifs. Among the binding events lost in one lineage, only half are recovered by another binding event within 10 kilobases. Our results reveal large interspecies differences in transcriptional regulation and provide insight into their evolution: We recently showed that cohesin co-binds across the genome with transcription factors independently of CTCF, plays a functional role in estrogen-regulated transcription, and may help mediate tissue-specific transcriptional responses via long-range chromosomal interactions:
|
Publications
|
Miscellaneous
|


