User:Johnsy/Lipoprotein Modelling/Compartmental Modelling
Compartmental Modelling
We now wish to separate the model of the human body into three compartments to see the dynamics established within the body. These three compartments are the hepatocytes, the epithelial cells (and other cells in the body), and the blood plasma. Each have their own unique identities which require a slightly different model for each. Most importantly, the mode of cholesterol regulation is very different between hepatocytes and epithelial cells and each is able to control different aspects of cholesterol metabolism. What occurs within epithelial cells is crucial to the local control of cholesterol metabolism, while the functionality afforded in the hepatocytes enables a global control. The transmission mechanism between the two types of cells is, of course, the lipoproteins in the blood plamsa.
A Model of the Hepatocyte
Introduction
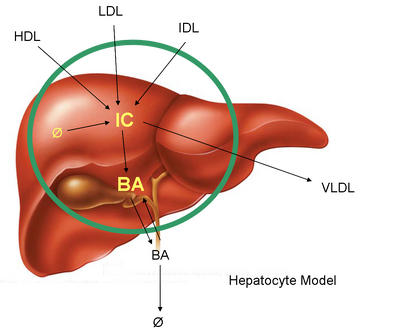
The hepatocyte is unique amongst all other cells in terms of cholesterol metabolism since it is the only cells in the body that can excrete cholesterol from the body via bile acids. Although other cells in the body are capable of performing the bile acid synthesis pathway, it is within the liver cell that it predominates through the enzyme cholesterol 7α hydroxylase.
Furthermore, hepatocytes are also the major suppliers of VLDL and HDL nascent particles. VLDL acts as a method for redistributing cholesterol around the body through its conversion to IDL and further to LDL while HDL is responsible for reverse cholesterol transport, or moving excess cholesterol from peripheral cells back to the liver. In our model, we neglect the cholesterol output from the hepatocyte as the nascent particles do not contain significant amounts of cholesterol. Instead, we focus on the uptake of cholesterol from HDL molecules from either an endocytotic pathway where the entire lipoprotein is ingested or via selective removal of choelsterol without uptake of the whole particle.[1]
The figure below shows the main players in the cholesterol flux through the hepatocyte.
For this compartment, we will have three main equations governing the cell: intracellular cholesterol, LDL receptors, and bile acid.
The Model
For Cholesterol
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[IC-H]}{dt} = & \frac{k_1}{d_1(b_1 + [IC-H])} \frac{k_2[HMG-CoA]}{k_{m1}+ [HMG-CoA] + \frac{k_{m1}}{k_i}[statin]} \\ & - \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-\eta)[BA])} \frac{k_3[IC-H]}{k_{m2} + [IC-H]} \\ & + (d_I \frac{[IDL-C]}{\chi_I} + d_L \frac{[LDL-C]}{\chi_L})\phi_{HLR} \\ & + d_5[LDL-C] \\ & + h[HDL-C] \\ & - u_v[IC-H] \\ \end{alignat} }[/math]
For Bile Acid
- [math]\displaystyle{ \frac{d[BA]}{dt} = \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-\eta)[BA])} \frac{k_3[IC-H]}{k_{m2} + [IC-H]} - d_3 \eta[BA] }[/math]
For LDL Receptors
- [math]\displaystyle{ \frac{d[\phi_{HLR}]}{dt} = -b(d_I \frac{[IDL-C]}{\chi_I} + d_L \frac{[LDL-C]}{\chi_L})\phi_{HLR} + c\frac{1-\phi_{HLR}}{b_3 + [IC-H]} }[/math]
Parameters
Lipoprotein Parameters
- dI - binding rate of IDL to LDL receptors describing receptor mediated uptake - 1 h-1 (August) [2]
- dL - binding rate of LDL to LDL receptors describing receptor mediated uptake - 0.01 h-1 (August)[2]
- d5 - rate of non receptor mediated uptake of LDL - 0.0075 h-1 (Dietschy) [3]
- χI - proportional cholesterol contained in IDL - 0.35 (Adiels)
- χL - proportional cholesterol contained in LDL - 0.45 (Adiels)
- h - internalization rate of HDL
- uv - production rate of VLDL
- b - internalization rate of lipoproteins through LDL receptors
- c - receptor recycling rate - 0.05 g(lh)-1 (Goldstein)[4]
- b3 - attenuation factor for the regulation of LDL receptors by cholesterol
De Novo Synthesis Parameters
- k1 – transcription rate for HMGR gene - 1896 min-1 (see De Novo Modelling)
- b1 – attenuation factor for the regulation of HMGR by cholesterol - 55 (see De Novo Modelling)
- d1 – degradation rate of HMGR (see below for value) - 0.031 min-1 (see De Novo Modelling)
- k2 – rate constant for conversion of HMG-CoA to cholesterol mediated by HMGR (equivalent to kcat value) - 22.6 sec-1 = 1356 min-1 (Theivagt) [5]
- km1 – Michaelis-Menten constant for HMGR (using NADPH as substrate)- 20 μM (Theivagt) [5]
- ki – dissociation constant for the action of statins 0.2 - 3.6 nM (Louis-Flamberg) [6]
Bile Acid Synthesis Parameters
- k3 - rate constant for the conversion of cholesterol to bile acids via C7H (equivalent to kcat value) - 3.59 min-1 (average value from P450 Type I and Type II) (Chiang) [7]
- km2 - Michaelis-Menten constant for C7H - 41.5 μM (average value from P450 Type I and Type II) (Chiang) [7]
- d3 - degradation (excretion) rate of bile acids which are not returned (rate at which ABCB11 transporter transfers bile acids from the hepatocyte into the bile) - 1.8 x 10-8 Mmin-1 (see Bile Acid Modelling)
- r1 - "degradation" rate of bile acids to recycled bile acids (assumed to be equal to d3 but in effect is attenuated by η)
- k5 - rate constant for the conversion of returned bile acid to bile acid (assume to be equal to r1)
- η - percentage of bile acids not recycled back to the liver - 5% (Redinger) [8]
- k4 - transcription rate for C7H - 1896 min-1 (see Bile Acid Modelling)
- b2 - attenuation factor for the regulation of C7H by RBA - 55 (see Bile Acid Modelling)
- d4 - degradation rate of C7H - 0.031 min-1 (see Bile Acid Modelling)
A Model of Peripheral Cells
Introduction
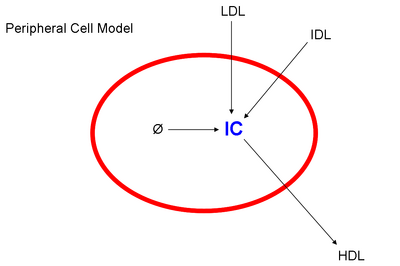
Peripheral cells are generally involved in autonomous cholesterol regulation and are usually only concerned about control within the cell itself. Both LDL and IDL particles are taken up by receptor mediated endocytosis and the cholesterol output is via HDL. De novo synthesis also occurs, but is usually a very small contributor to overall cholesterol concentrations since the cholesterol requirement is usually filled by the intake of lipoproteins. The cholesterol concentration is generally kept constant within the cell regardless of the concentration of lipoproteins in the blood plasma.
A general model of the peripheral cell is shown below.
For this compartment, we have two main equations governing cholesterol metabolism within these cells: Intracellular cholesterol and LDL receptors
The Model
For Cholesterol
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[IC-P]}{dt} = & \frac{k_1}{d_1(b_1 + [IC-P])} \frac{k_2[HMG-CoA]}{k_{m1}+ [HMG-CoA] + \frac{k_{m1}}{k_i}[statin]} \\ & + (d_I \frac{[IDL-C]}{\chi_I} + d_L \frac{[LDL-C]}{\chi_L})\phi_{PLR} \\ & + d_5[LDL-C] \\ & - d_{HDL} \frac{[HDL-C]}{\chi_H}([IC-P]-p[HDL-C]) \\ \end{alignat} }[/math]
For LDL Receptors
- [math]\displaystyle{ \frac{d[\phi_{PLR}]}{dt} = -b(d_I \frac{[IDL-C]}{\chi_I} + d_L \frac{[LDL-C]}{\chi_L})\phi_{PLR} + c\frac{1-\phi_{PLR}}{b_3 + [IC-P]} }[/math]
The Parameters
Those not mentioned previously:
- dHDL - transfer rate of cholesterol from peripheral cells to HDL
- χH - amount of cholesterol contained within an average HDL particle
- p - attenuation coefficient for cholesterol transfer to HDL from peripheral cells
A Model of the Blood Plamsa
Introduction
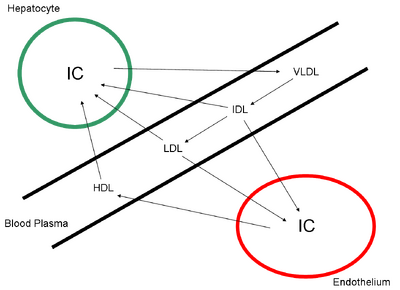
Probably the most well studied portion is the conversion of VLDL to IDL and the further conversion to LDL. These lipoproteins are present in the plamsa and serve to shuttle cholesterol from the liver to the extrahepatic cells as well as to take excess cholesterol from these extrahepatic cell back to the liver (reverse cholesterol transport). As mentioned previously, these lipoproteins are the mediators between our two compartments we are considering and is the the most clinically relevant aspect of the project. Clinicians refer to both LDL and HDL levels as a marker for cardiovascular disease and prescribe medications to correct these levels. Although it is probably the intracellular cholesterol levels that are actually responsible for the atherosclerosis phenomenon, the difficulty to assess intracellular cholesterol levels in patients has prevented it's use. Hence, clinicians still rely on statistics that have shown a correlation between lipoprotein levels and the prevalence of coronary heart disease (CHD).
The model that is used is shown below.
Here, we are concerned with all of the lipoproteins and the rate of change over time starting with VLDL, IDL, LDL, and HDL. The equations governing these are shown below.
The Model
For VLDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[VLDL-C]}{dt} = & u_v[IC-H] - k_v[VLDL-C] \\ & + k_{cetp}[HDL-C][VLDL-C]([HDL-C]-a_v[VLDL-C]) \end{alignat} }[/math]
For IDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[IDL-C]}{dt} = & k_v[VLDL-C] -k_i[IDL-C] \\ & + k_{cetp}[HDL-C][IDL-C]([HDL-C]-a_i[IDL-C]) \\ & - d_i[IDL-C]\phi_{HLR} - d_i[IDL-C]\phi_{PLR} \\ \end{alignat} }[/math]
For LDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[LDL-C]}{dt} = & k_i[IDL-C] - d_5[LDL-C] \\ & - d_l[LDL-C]\phi_{HLR} - d_l[LDL-C]\phi_{PLR} \\ & + k_{cetp}[HDL-C][LDL-C]([HDL-C]-a_l[LDL-C]) \\ \end{alignat} }[/math]
For HDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[HDL-C]}{dt} = & d_{HDL} \frac{[HDL-C]}{\chi_H}([IC-P]-p[HDL-C]) \\ & + k_{cetp}[HDL-C][VLDL-C]([HDL-C]-a_v[VLDL-C]) \\ & + k_{cetp}[HDL-C][IDL-C]([HDL-C]-a_i[IDL-C]) \\ & + k_{cetp}[HDL-C][LDL-C]([HDL-C]-a_l[LDL-C]) \\ & - h[HDL-C] \end{alignat} }[/math]
The Parameters
Those not shown previously:
- kv - conversion rate of VLDL to IDL - 0.3 h-1 (Packard)[9]
- ki - conversion rate of IDL to LDL- 0.05 h-1 (Packard)[9]
- kcept - transfer rate of Cholesterol Ester Transfer Protein (CETP)
- av - attenuation coefficient for cholesterol transfer from HDL to VLDL
- ai - attenuation coefficient for cholesterol transfer from HDL to IDL
- al - attenuation coefficient for cholesterol transfer from HDL to LDL
References
- Lewis GF and Rader DJ. New insights into the regulation of HDL metabolism and reverse cholesterol transport. Circ Res. 2005 Jun 24;96(12):1221-32. DOI:10.1161/01.RES.0000170946.56981.5c |
- August E, Parker KH, and Barahona M. A dynamical model of lipoprotein metabolism. Bull Math Biol. 2007 May;69(4):1233-54. DOI:10.1007/s11538-006-9132-6 |
- Dietschy JM, Turley SD, and Spady DK. Role of liver in the maintenance of cholesterol and low density lipoprotein homeostasis in different animal species, including humans. J Lipid Res. 1993 Oct;34(10):1637-59.
- Goldstein JL and Brown MS. The low-density lipoprotein pathway and its relation to atherosclerosis. Annu Rev Biochem. 1977;46:897-930. DOI:10.1146/annurev.bi.46.070177.004341 |
- Theivagt AE, Amanti EN, Beresford NJ, Tabernero L, and Friesen JA. Characterization of an HMG-CoA reductase from Listeria monocytogenes that exhibits dual coenzyme specificity. Biochemistry. 2006 Dec 5;45(48):14397-406. DOI:10.1021/bi0614636 |
- Louis-Flamberg P, Peishoff CE, Bryan DL, Leber J, Elliott JD, Metcalf BW, and Mayer RJ. Slow binding inhibition of 3-hydroxy-3-methylglutaryl-coenzyme A reductase. Biochemistry. 1990 May 1;29(17):4115-20. DOI:10.1021/bi00469a014 |
- Chiang JY, Miller WF, and Lin GM. Regulation of cholesterol 7 alpha-hydroxylase in the liver. Purification of cholesterol 7 alpha-hydroxylase and the immunochemical evidence for the induction of cholesterol 7 alpha-hydroxylase by cholestyramine and circadian rhythm. J Biol Chem. 1990 Mar 5;265(7):3889-97.
- Redinger RN. The coming of age of our understanding of the enterohepatic circulation of bile salts. Am J Surg. 2003 Feb;185(2):168-72. DOI:10.1016/s0002-9610(02)01212-6 |
- Packard CJ, Demant T, Stewart JP, Bedford D, Caslake MJ, Schwertfeger G, Bedynek A, Shepherd J, and Seidel D. Apolipoprotein B metabolism and the distribution of VLDL and LDL subfractions. J Lipid Res. 2000 Feb;41(2):305-18.
Appendix
Kathryn's Original Model
For VLDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[VLDL-C]}{dt} = & \chi_u u_v - k_v[VLDL-C] \\ & + k_{cetp}[HDL-C][VLDL-C]([HDL-C]-a_v[VLDL-C]) \end{alignat} }[/math]
For IDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[IDL-C]}{dt} = & k_v[VLDL-C] -k_i[IDL-C] \\ & + k_{cetp}[HDL-C][IDL-C]([HDL-C]-a_i[IDL-C]) \\ & - d_i[IDL-C]\phi_{LR} \\ \end{alignat} }[/math]
For LDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[LDL-C]}{dt} = & k_i[IDL-C] - d_l[LDL-C]\phi_{LR} - d[LDL-C] \\ & + k_{cetp}[HDL-C][LDL-C]([HDL-C]-a_l[LDL-C]) \\ \end{alignat} }[/math]
For LDL Receptors:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[\phi_{LR}]}{dt} = -b(d_I \frac{[IDL-C]}{\chi_I} + d_L \frac{[LDL-C]}{\chi_L})\phi_{LR} + c\frac{1-\phi_{LR}}{[IC]} \end{alignat} }[/math]
For IC:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[IC]}{dt} = & (d_I [IDL-C] + d_L [LDL-C])\phi_{LR} \\ & + d[LDL-C] \\ & - d_{HDL} \frac{[HDL-C]}{\chi_H}([IC]-p[HDL-C]) \\ & - d_{IC}[IC] \end{alignat} }[/math]
For HDL:
- [math]\displaystyle{ \begin{alignat}{2} \frac{d[HDL-C]}{dt} = & d_{HDL} \frac{[HDL-C]}{\chi_H}([IC]-p[HDL-C]) \\ & + k_{cetp}[HDL-C][VLDL-C]([HDL-C]-a_v[VLDL-C]) \\ & + k_{cetp}[HDL-C][IDL-C]([HDL-C]-a_i[IDL-C]) \\ & + k_{cetp}[HDL-C][LDL-C]([HDL-C]-a_l[LDL-C]) \\ & - h[HDL-C] \end{alignat} }[/math]