User:Johnsy/Lipoprotein Modelling/Bile Acid Biosynthesis
Background
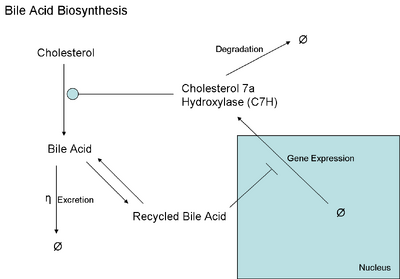
We first start off with a diagram of the model that we wish to use for the bile acid biosyntheis pathway.
In hepatocytes, cholesterol is converted to bile acids to be excreted into the intestine via the action of the enzyme cholesterol 7α hydroxylase (CYP7A1, C7H for short). The bile acid is excreted and stored in the gall bladder and is pooled until hormonal changes within the body signal to contract and excrete the bile acid into the gut during feeding. Bile acids are key to increasing the solubility of ingested lipids. Approximately 95% of the bile acids are returned to the circulation through the ileum (termed enterohepatic circulation). The returned bile acids then inhibit the gene expression of C7H through a complex unknown pathway, but modeled simply as a direct inhibitor as seen below. The returned bile acids are returned to the bile acid pool in the hepatocytes with a certain rate constant.
The Model
The equation governing the enzymatic degradation of cholesterol by the enzyme Cholesterol 7α Hydroxylase (C7H) is modeled using a simple Michaelis-Menten type equation as shown below. This equation will be combined with that from the de novo synthesis pathway to complete our hepatocyte model.
- [math]\displaystyle{ \frac{d[IC]}{dt} = - \frac{k_3[C7H][IC]}{k_{m2} + [IC]} }[/math]
The equation for the synthesis of bile acids follows from the degradation of cholesterol assuming that in this model cholesterol is only degraded to bile acid.
- [math]\displaystyle{ \frac{d[BA]}{dt} = \frac{k_3[C7H][IC]}{k_{m2} + [IC]} - d_3 \eta[BA] - r_1(1-\eta)[BA] + k_5[RBA] }[/math]
Individual terms are described below:
- [math]\displaystyle{ d_3 \eta[BA] }[/math] - This term describes the excretion of bile acids of which a fraction η is not returned to the liver (η is usually about 5% of the excreted bile acids).
- [math]\displaystyle{ r_1(1-\eta)[BA] }[/math] - This term describes the excretion of bile acids which are recycled to the blood stream and are converted to "Returned Bile Acids" (RBA). This was done for the convenience since the gene expression of C7H is dependent on the returned bile acid pool and not the bile acid pool already present in the hepatocyte.
- [math]\displaystyle{ k_5[RBA] }[/math] - This term describes the conversion of Returned Bile Acids to the Bile Acid pool.
The equation governing the production of the enzyme C7H is described below. This is a genetically controlled production and will be justified and described further below. The justification follows similar lines to the genetic justification for the production of HMGR in the de novo synthesis pathway.
- [math]\displaystyle{ \frac{d[C7H]}{dt} = \frac{k_4}{b_2 + [RBA]} - d_4[C7H] }[/math]
And finally, the equation relating to the returned bile acids is shown below.
- [math]\displaystyle{ \frac{d[RBA]}{dt} = r_1(1-\eta)[BA] - k_5[RBA] }[/math]
Parameters
- k3 - rate constant for the conversion of cholesterol to bile acids via C7H (equivalent to kcat value) - 3.59 min-1 (average value from P450 Type I and Type II) (Chiang) [1]
- km2 - Michaelis-Menten constant for C7H - 41.5 μM (average value from P450 Type I and Type II) (Chiang) [1]
- d3 - degradation (excretion) rate of bile acids which are not returned (rate at which ABCB11 transporter transfers bile acids from the hepatocyte into the bile) (see section below for calculation of this value)
- r1 - "degradation" rate of bile acids to recycled bile acids (assumed to be equal to d3 but in effect is attenuated by η)
- k5 - rate constant for the conversion of returned bile acid to bile acid (assume to be equal to r1)
- η - percentage of bile acids not recycled back to the liver - 5% (Redinger) [2]
- k4 - transcription rate (see section below for value)
- b2 - attenuation factor for the regulation of C7H by RBA (see section below for value)
- d4 - degradation rate of C7H (see section below for value)
Assumptions
Cholesterol Conversion to Bile Acid
In the conversion of cholesterol to bile acid, there are several enzymes involved, but the rate limiting step is the conversion of cholesterol to 7α-hydroxy cholesterol by the action of a P450 cytochrome reductase [3]. We assume that all the other steps in this conversion are fast and the rates are negligible compared to that of the rate limiting step. Hence, we can assume that the overall rate of production of bile acids from cholesterol is defined by the kinetics of this step.
Furthermore, we also assume that the concentration of C7H is not constant and will vary over time defined by the equation above governing production of C7H. Although the derivation of the Michaelis-Menten equation assumed that the concentration of enzyme was kept constant, we assume that the steady state value of the enzyme will be reached before any significant changes in the level of enzyme. This is supported by the fact that there is a delay of approximately 40 minutes between regulation of gene expression and the presence or reduction of protein in the cell.
We currently assume that the only degradation by cholesterol is through bile acids, but we will couple the cholesterol governing equation with the same from the de novo synthesis pathway and from the lipoprotein pathway. By separating each component, we can compartmentalize and have modularity of the model.
Unlike the conversion of HMG-CoA to cholesterol, we cannot assume that the enzyme will be rate limiting. Although studies that have probed the kinetic parameters of C7H have assumed cholesterol levels that are much higher than enzyme levels, in our model, there is a dynamical equilibrium set up between the amount of cholesterol in the cell and the amount converted to bile acids.
Enterohepatic Circulation
A complex pathway is required for the return of bile acids from the ileum to the liver through chylomicrons and portal vein circulation. Unfortunately, the exact mechanism for this return is unknown [2] and we just assume a linear relationship between the rate of return and the amount of bile acid present in the intercellular bile acid pool. Furthermore, we also assume a linear relationship in the rate of conversion from the recycled bile acid pool to the intercellular bile acid pool, since literature review has not given us any significant insight into this metabolic pathway in hepatocytes.
We have also assumed that only the returned bile acid pool regulates the gene expression of C7H. In fact, there is a complex negative feedback through multiple nuclear hormone receptors from the returned bile acid which regulates the production of transcription factors that regulate the transcription of the genes required for bile acid synthesis. [2] As usual for modeling, we are neglecting these complications and simplifying our model as much as possible yet attempting to maintain the key biochemical elements involved.
The bile acids themselves return to from the ileum through the hepatic portal vein through the action of ileal bile acid binding proteins which facilitate the movement of bile acids across the cell and into the portal vein. The bile acids are then taken up by a transporter on the surface of the hepatocyte and then secreted into the bile duct again for another round of recycling. [4]
For the conversion of returned bile acid to bile acid, we can assume that this happens fast and that the rate of influx will always equal to the rate of conversion of returned bile acids to the bile acid pool. This effectively maintains the rate of returned bile acids equal to zero to make the quasi-steady state approximation valid.
Quasi-steady state approximation
Similar to what was performed with the de novo synthesis of cholesterol, we can again take the quasi-steady state approximation for several of the equation above to reduce our dimensionality. First, since the recycled bile acids are being converted to bile acids, we can also assume that the concentration of recycled bile acids will be at steady state such that:
- [math]\displaystyle{ \frac{d[RBA]}{dt} = 0 }[/math]
Solving for the steady state concentration of returned bile acids, we obtain:
- [math]\displaystyle{ [RBA]^* = \frac{r_1}{k_5}(1-\eta)[BA] }[/math]
We then assume that the concentration of the enzyme will be at steady state such that:
- [math]\displaystyle{ \frac{d[C7H]}{dt} = 0 }[/math]
Solving for the steady state concentration of C7H, we obtain:
- [math]\displaystyle{ [C7H]^* = \frac{k_4}{d_4(b_2 + [RBA])} }[/math]
Substituting our expression for the steady state concentration of the recycled bile acids:
- [math]\displaystyle{ [C7H]^* = \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-\eta)[BA])} }[/math]
Because we have assumed that the concentration of recycled bile acids is constant, our equation for the rate of change of bile acids is also simplified.
- [math]\displaystyle{ \frac{d[BA]}{dt} = \frac{k_3[C7H][IC]}{k_{m2} + [IC]} - d_3 \eta[BA] }[/math]
And our final equations governing both cholesterol levels and bile acid levels are shown below.
- [math]\displaystyle{ \frac{d[BA]}{dt} = \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-\eta)[BA])} \frac{k_3[IC]}{k_{m2} + [IC]} - d_3 \eta[BA] }[/math]
- [math]\displaystyle{ \frac{d[IC]}{dt} = - \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-\eta)[BA])} \frac{k_3[IC]}{k_{m2} + [IC]} }[/math]
Justification of the Genetic Regulatory Term for the Production of C7H
We use the following network to justify the inverse regulatory component from the recycled bile acids inhibits the production of C7H:
Similar to what was done with the genetic regulation of the production of HMGR, we can come up with the following equations to describe the genetic system at work.
- [math]\displaystyle{ \frac{d[mRNA]}{dt} = \frac{g_1g_m^n}{g_m^n + [IC]^n} - w_1[mRNA] }[/math]
- [math]\displaystyle{ \frac{d[C7H]}{dt} = g_2[mRNA] - w_2[C7H] }[/math]
The parameters of the this model are defined below with relevant values found in literature:
- g1 - transcription rate - 1 min-1 (Barrio)[5]
- g2 - translation rate - 1 min-1 (Barrio)[5]
- gm - DNA Dissociation Rate - 10-100 (no units) (Barrio)[5]
- n - Hill cooperativity - assumed to be 1 for this case
- w1 - mRNA degradation rate - 0.029 min-1 (Barrio)[5]
- w2 - C7H degradation rate (assumed to be an ordinary protein degradation rate) - 0.031 min-1 (Barrio)[5]
We again assume that the mRNA will be at steady state:
- [math]\displaystyle{ \frac{d[mRNA]}{dt} = 0 }[/math]
So that we can come up with the fixed point and substitute into the C7H equation:
- [math]\displaystyle{ \frac{d[C7H]}{dt} = \frac{g_1 \ g_2 \ g_m^n}{w_1(g_m^n + [IC]^n)} - w_2[C7H] }[/math]
Comparing this with the above equation for C7H, we can see some equivalents to the equation which will help us to determine the value of the parameters that we are using. For the equations above, we assume that the Hill cooperativity constant n = 1, as we assume that only one molecule of bile acid will be sufficient to repress transcription of the genes required to express C7H.
- [math]\displaystyle{ k_4 = \frac{g_1 \ g_2 \ g_m}{w_1} = \frac{1 \ min^{-1} \times 1 \ min^{-1} \times 55}{0.029 \ min^{-1}} = 1896 \ min^{-1} }[/math]
Also,
- [math]\displaystyle{ b_2 = g_m = 55 \mbox{ (no units)} }[/math]
and
- [math]\displaystyle{ d_4 = w_2 = 0.031 \ min^{-1} }[/math]
Bile Acid Binding Resin Effects on the Model
Bile Acid resins are another method by which physicians have helped to lower the plasma cholesterol levels. They are taken orally and work by binding the bile acids so that they are not taken up again in the ileum. This results in a lower amount of recycled bile acid which will in turn increase the production of C7H, and hence increase the conversion of cholesterol to bile acid, lowering the cholesterol levels. The effect on LDL levels is seen when considering the entire model when lipoprotein cholesterol contribution is taken into account.
Modeling this effect is relatively easy, and we assume an inverse proportion of resins to the amount that of bile acid that is not recycled. We take this into account by slightly modifying the model such that instead of η, we now use:
- [math]\displaystyle{ k_6 \eta [Resin] }[/math]
Here, k6 is the binding rate of the resin to the bile acids and the entire term will replace η in all of our equations as shown below.
- [math]\displaystyle{ \frac{d[BA]}{dt} = \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-k_6 \eta [Resin])[BA])} \frac{k_3[IC]}{k_{m2} + [IC]} - d_3 k_6 \eta [Resin][BA] }[/math]
- [math]\displaystyle{ \frac{d[IC]}{dt} = - \frac{k_4}{d_4(b_2 + \frac{r_1}{k_5}(1-k_6 \eta [Resin])[BA])} \frac{k_3[IC]}{k_{m2} + [IC]} }[/math]
Calculation of bile acid degradation (excretion) rate
The human bile acid salt pool circulates 6 to 10 times per day with a daily excretion of approximately 20-40 g. However, only 0.5 g of bile salts are lost through fecal excretion each day of an average individual.[6]
Assuming that an average individual is approximately 60 kg and this is approximately 60 L of water volume:
- [math]\displaystyle{ d_3 = \frac{0.5 \ g}{60 \ kg \times day} \times \frac{1 \ kg \ H_2O}{1 \ L \ H_2O} \times \frac{1 \ day}{1440 \ min} \times \frac{1 \ mol}{393 \ g} = 1.5 \times 10^{-8} \frac{M}{min} }[/math]
References
- Chiang JY, Miller WF, and Lin GM. Regulation of cholesterol 7 alpha-hydroxylase in the liver. Purification of cholesterol 7 alpha-hydroxylase and the immunochemical evidence for the induction of cholesterol 7 alpha-hydroxylase by cholestyramine and circadian rhythm. J Biol Chem. 1990 Mar 5;265(7):3889-97.
- Redinger RN. The coming of age of our understanding of the enterohepatic circulation of bile salts. Am J Surg. 2003 Feb;185(2):168-72. DOI:10.1016/s0002-9610(02)01212-6 |
- Russell DW and Setchell KD. Bile acid biosynthesis. Biochemistry. 1992 May 26;31(20):4737-49. DOI:10.1021/bi00135a001 |
- Russell DW. Nuclear orphan receptors control cholesterol catabolism. Cell. 1999 May 28;97(5):539-42. DOI:10.1016/s0092-8674(00)80763-1 |
- Barrio M, Burrage K, Leier A, and Tian T. Oscillatory regulation of Hes1: Discrete stochastic delay modelling and simulation. PLoS Comput Biol. 2006 Sep 8;2(9):e117. DOI:10.1371/journal.pcbi.0020117 |
- Trauner M and Boyer JL. Bile salt transporters: molecular characterization, function, and regulation. Physiol Rev. 2003 Apr;83(2):633-71. DOI:10.1152/physrev.00027.2002 |