User:Janaye M. Monica/Notebook/Biology 210 at AU
3/18 Zebra Fish Experiment
Purpose
The first purpose of this lab was to learn the stages of the embryonic development of zebra fish. The second purpose was to determine how retinoic acid might affect the embryonic development of zebra fish when it is added to the organism’s environment.
Materials and Methods
Collect two petri dishes.One of the petri dishes represents the control group and it will be filled with approximately 20 mLs of Deerpark water. The second petri dish will contain 20 mLs of water and the retinoic acid additive; this will represent the test variable. Make sure to label each petri dish accordingly. Once the appropriate solution is in each petri dish environments transplant 20 healthy and translucent embryos in both dishes. Use a dropper to transfer each embryo into their new environments. Observe your zebra fish and record their current developmental stages. Tape the petri dishes closed and place them in a safe environment. Determine your observational schedule. On day 4 and 5, remove 10 mLs of water and any dead embryos (they will appear discolored and fuzzy). Add approximately 25 mLs of fresh water. Observe the embryos through a compound microscope. On each observation day, count the number of dead eggs, the number of living embryos, the number of larvae. Also, note the embryos developmental stage and, if possible, their general movement, eye movement, possible heart rate, and development of swim bladder, mouth and fins. On day 7 of the experiment, remove 5mLs water and any dead embryos. Add 25mLs of fresh water once again. With a dropper, remove 3 embryos from both petri dishes and preserve them in a small tube of paraformaldehyde. At this stage of development, introduce food to the embryos and place two drops of paramecium into the petri dishes. This should be continued whenever the solution is changed. Between day 7 and 14, remove 5mls and add 10 mLs. Final observations should be made non day 14 of the experiment. Results
Day 1:
Number of living embryos control: 20
Number of living embryos experimental: 20
Number of dead embryos control: 0
Number of dead embryos experimental: 0
Day 3:
Developmental stage: 20-somites
Number of living embryos control: 20
Number of living embryos experimental: 20
Number of dead embryos control: 0
Number of dead embryos experimental: 0
Number of living or dead hatchlings control: 0
Number of living or dead hatchlings experimental: 0
Other observations were not possible at this point due to their early developmental stage and they were all still in their egg cage.
Day 5:
Developmental stage: 48 hours
Number of living hatchlings control: 14
Number of living hatchling experimental: 16
Number of dead embryos control: 6
Number of dead embryos experimental: 4
No noticeable differences between the hatchlings in the control and experiment. There was no heartbeat observed. The water and retinoic acid solution accidently spilled and was replaced by the instructor.
Day 7:
Developmental stage: Fully developed, past 48 hours
Number of living hatchlings control: 11
Number of living hatchling experimental: 14
Number of dead embryos control: 9
Number of dead embryos experimental: 6
Degree of body and tail pigmentation control: Gray/ black color
Degree of body pigmentation experimental: Yelow hue
Eye movement control and experimental: Eyes were developed but eye movement was not noticeable.
General Movement control: Slow
General Movement experimental: Shaking with quick movements, moved faster that the control group. Yoke sac size control and experimental: developed
Day 14:
Number of dead hatchlings control: 20
Number of dead hatchling experimental: 20
Conclusion
The hypothesis of this lab was that the introduction of retinoic acid into the environment of the zebra fish embryos, then the embryonic development of the zebra fish would be negatively affected. This hypothesis can’t be accepted due to the observations listed above. It appeared that the zebra fish inhabiting the retinoic acid solution moved faster than their counterparts. Additionally, there was no noticeable difference in the experimental embryos development except for their yellow discoloration. If the experiment was to be recreated, the petri dishes should be kept in a safer area to limit the amount of environmental disturbances. Also, the embryos should be observed more frequently.
JM
3/4 16S Sequence
Purpose
The purpose was to better identify the bacteria that inhabit each specific transect. Identification was achieved through the sequencing the 16S rRNA. The 16S rRNA is a highly conserved gene among all species of bacteria. Additionally the sequence is long, approximately 1500bp, thus providing valid measurements (Clarridge, 2004).
Methods
Select one nutrient agar plate and one tetracycline plate with the most complete bacteria characterization. Once the plates are chosen, transfer a single bacteria colony from each plate and place it in a sterile tube with 100 ul of water. From there, the bacteria’s DNA was isolated and then amplified through the PCR technique. To better identify the bacterial species, the 16S rRNA gene is amplified and casted onto the agarose gel. Specific sequences were provided to each transect group. The groups were then expected to input their nucleotide sequence into the online program, BLAST. BLAST identifies species based on the nucleotide sequence.
Results
Raw sequence for Sample A: NNNNNNNNNNNNGNNNTTANNNNTGCAGTCGNNCGANNGAGTAGCNCNNNNTNNCGGACGCTGACGAGTGGCGAACGGGT GAGTAATACTATCGGAACGTGCCCAGTCGTGGGGGATAACTACTCGAAAGAGTAGCTAATACCGCATACGATCTGAGGAT GAAAGCGGGGGACCTTCGGGCCTCGCGCGATTGGAGCGGCCGATGGCAGATTAGGTAGTTGGTGGGATAAAAGCTTACCA AGCCGACGATCTGTAGCTGGTCTGAGAGGACGACCAGCCACACTGGGACTGAGACACGGCCCAGACTCCTACGGGAGGCA GCAGTGGGGAATTTTGGACAATGGGCGAAAGCCTGATCCAGCAATGCCGCGTGCAGGATGAAGGCCTTCGGGTTGTAAAC TGCTTTTGTACGNAACGAAAAAGCTTCTCCTAATACNAGAGGCCCATGACGGTACCGTANNAATAAGCACCGGCTAACTA CGTGCCANCAGCCGCGGTANTACGNANGGTGCGAGCGTTNATCGCGANTTTACTGNGCGTAAAGCGTGCGCANGCNGNTA TGTNANGACANNATGTGAAATCCCCNNNNTCTNTCCTGNTNAACTGNCNTTTGTGNCTGCNNGGNTAAANTACGGNAGAG GNGTGNTGGAAT.
BLAST identified Sample A’s sequence as an “uncultured bacteria.”
Raw Sequence for Sample D: NNNNNNNNNNNNNNNNNNNNNNTGCAGCCGAGCGCTANGTTTCCTTCGNGGAGGACTGNAGANANNCGCACGGGTGCGGA ACATGTGTGCAACCTGCCTTTATCAGGGGGATAGCCTTTCGAAAGGAAGATTAATACCCCATAATATTTTGAGTGGCATC ACTTGAAATTGAAAACTCCGGTGGATAAAGATGGGCACGCGCAAGATTAGATAGTTGGTGAGGTAACGGCTCACCAAGTC TACGATCTTTAGGGGGCCTGAGAGGGTGATCCCCCACACTGGTACTGAGACACGGACCAGACTNNNNCGGGAGGCAGCNG TGAGGAATATTGGACGATGNGTGAGAGCCTGATCCAGCCATCCCGCGTGAAGGACGACGGCCCTATNGGTTGTAAACTTC TTTTGTATAGGGATAAACCTTTCCACGTGTGGGANGCTGAAGGTACTATACGAATAAGCACCGGCTAACTCCGTGCCAGC NGCCGCGGTAATACGGAGGGTGCAAGCGTTATCCGGATTTATTGGGTTTAAAGGGTCCGTAGGCGGATCTGTNAGTCAGT GGTGAAATCTCACAGCTTAACTGTGAAACTGCCATTGATACTGCNGGTCTTGAGTGNTGTTGAAGTAGCTGGAANAAGTN GTGTANCGGTGAAATGCATAGATNTNACNNACAACACCANTNGCGAAGGNNGGTTACTAANCAACAACTGANGCTNNNGG ACGAAGCGTGGGGAGCGAACAGGATTAGATACCCNGGTANTCCACGCNTNNNCGANGCNNNTCCGTTTTNGNTTTCNGAT CNNAGNCNAANCGAANGTGANCAGTAGCCACTGGGNGNNTNCGNCGNANTNNNNANNNTCNNNNNATTGACNGGGGNNNN CNNNNNNNGNNNNTTNTGTCNTTANTCNNNNANNNCGCNNGGNNCNNNNNNNCNNANNNGNNNNGNNAGNNNNNNNNNCN TNNNNGNNTTNNNNGNNNNNNNNNNNNNCANCNNNCCGNNNNGNNNANTANNNNNGNNNNNNCNNNNC.
BLAST identifies the bacteria as "Chryseobacterium sp. B."
Conclusion
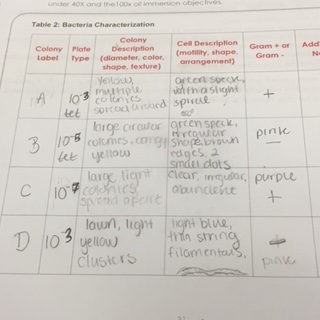
BLAST was unable to identify Sample A. However, Sample D was classified as Chryseobacterium. According to my lab group’s Bacteria Characterization Table, as seen in Lab 3, Sample D’s colony description was a light yellow lawn that grew in clusters. Also, it was categorized as gram negative. Chryseobacterium is a gram negative bacteria that exhibits a similar color and growing pattern as Sample D (Hugo, Segers, Host, Vancanneyt and Kersters, 2003). Due to these similarities, it is appropriate to assume that Sample D is Chryseobacterium.
Work Cited
http://ijs.sgmjournals.org/content/53/3/771.full http://www.ncbi.nlm.nih.gov/pmc/articles/PMC523561/
JM
2/19 Invertebrate Lab
Purpose
The purpose of this lab was to identify various invertebrates that inhabit the leaves collected from my group’s transect. Invertebrates do not have internal skeletons and make up approximately 98% of the animal species in the world.
Materials and methods
Procedure I
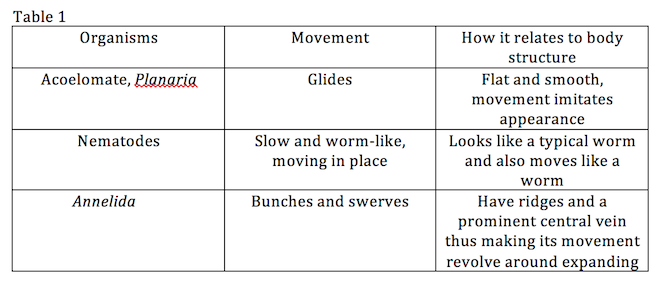
Observe Acoelomates, Pseudocoelomates and Coelomates with a dissecting microscope. Observe the movements of each worm and how this particular movement relates with their body structure.
Procedure II
Collect 500g of leaf litter from the transect and place it into a Ziploc bag.
Procedure III: Setting up the Berlese Funnel
1. Get a funnel, screen and conical tube.
2. Pour 25 mL of a 50:50 ethanol/water solution into the conical tube.
3. At the bottom of the funnel, tape a small piece of screening material into the bottom of the funnel; this is to ensure that the leaf litter does not fall into the conical tube.
4. Wrap the parafilm at the base of the funnel and the top of the conical tube to keep the ethanol from evaporating.
5. Set the funnel on a ring stand so that the Berlese funnel is upright.
6. Leave the funnel in the lab for one week
Procedure II: Analyzing the Invertebrates collected 1. After the week passes, take apart the Berlese Funnel and pour the top 10-15 mLs of liquid into a petri dish
2. Then pour the last amount into a second petri dish.
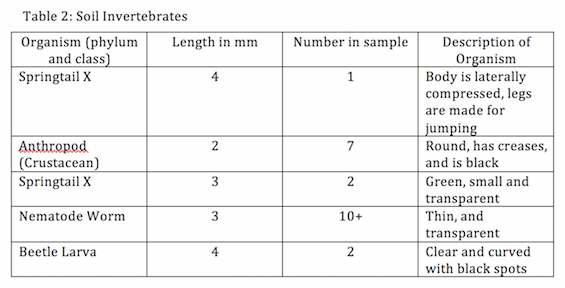
3. View the two petri dishes under a dissecting microscope and identify all the observed Athropods invertebrates.
Data
Procedure I
Procedure II and III
ConclusionAll of the five invertebrate samples that my group was able to identify were common soil invertebrates. The findings are due to the environment from which we collected the leaf samples.
Vertebrate Lab
Purpose
The purpose of this lab is to identify vertebrates in an attempt to better describe the transect environment.
Methods
We went to our transect during the late afternoon, around 4:30. The sun was low, it was extremely windy and cold.
Results
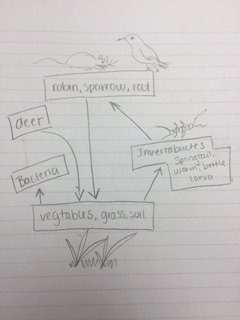
5 species inhabiting the transect: Rat, sparrow, American robin, pigeon and deer Vertebrate: Kingdom, Phylum, Class, Order, Family, Genus, Species Rat: Animalia, Chordata, Sarcopterygii, Rodentia, Muridae, Rattus Sparrow: Animalia, Chordata, Aves, Passeriformes, Emberizidae, Melospiza American Robin: Animalia, Chordata, Aves, Passeriformes, Turdidae, Turdus, T. migratorius Pigeon: Animalia, Chordata, Sarcoptergii, Aves, Columbiformes, Columbidae, Deer: Animalia, Chordata, Sarcopterygii, Mammalia, Artiodactyla, Cervidae
Food Web of the Community Garden Transect
Conclusions
A biotic feature that these organisms would benefit from within this transect would be the various vegetable patches. They provide food for the rat, sparrow, American robin, pigeon and deer. In fact, we found deer hoof prints in the vegetable patch. The rat, sparrow and American robin will also eat the invertebrates that live in the transect. In terms of abiotic features, rats and birds could utilize the random stick and twigs for shelter. The community of the transect is diverse and the organism interactions ultimately sustain the community. The two birds and the rat are at virtually the same trophic level since they all consume insects and vegetation. The carrying capacity refers to the maximum population of a certain species that can survive within a given environment. I assume that none of the species observe have reached their maximum carrying capacity.
JM
Sources:
http://animaldiversity.org/accounts/Cervidae/classification/ http://animaldiversity.org/accounts/Columbiformes/classification/ http://animaldiversity.org/accounts/Rattus_rattus/classification/ http://www.nhptv.org/natureworks/robin.htm http://www.sustainablescale.org/ConceptualFramework/UnderstandingScale/MeasuringScale/CarryingCapacity.aspx
2/11 Lab 4 Plantae and Fungi
Purpose of lab
The purpose of this lab is to understand the characteristics and diversity of the plants within the assigned transect. Land plants evolved from aquatic green algae, resembling the modern day phyla of Brophytes. Plants are can be categorized by the following three general features: presence of vascularization, presence of specialized structures and mechanisms of reproduction. In general, the overall purpose of this lab was to observe, identify and classify various plants located in the transect.
Procedures and materials
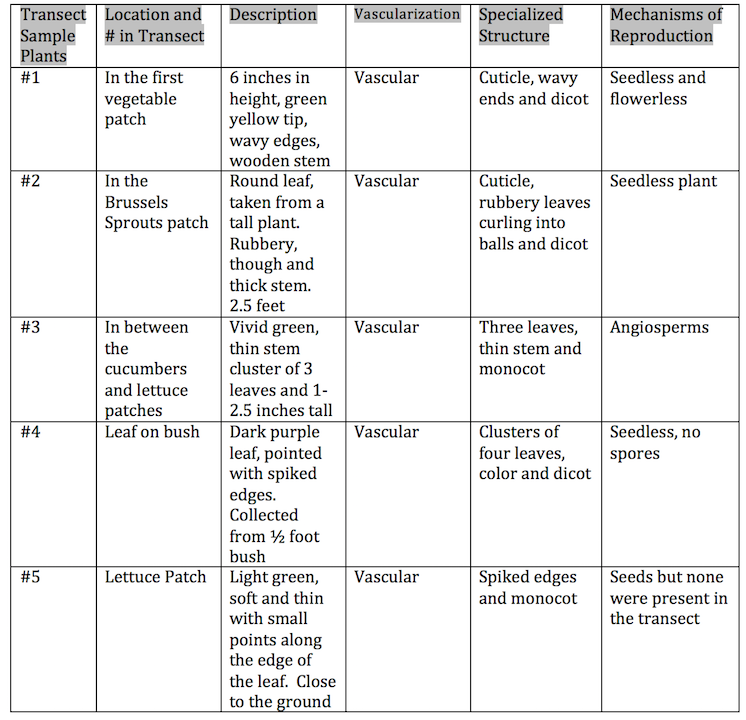
Procedure I Examine moss, Mnium, through a compound microscope and identify both the male and female gametophytes and sporophyte. Also observe the cross section slide of a lily stem and find xylem and phloem layers. Procedure II Obtain two Ziploc bags and gloves and walk to your designated transect. Collect a handful of dead leaves, approximately 500g, from the same area. Transfer the leaves, other plant matter and bits of soil into one of the bags. These leaves will be used in the Berlese Funnel to collect invertebrates for next week's lab. Then take five samples of diverse plants from various areas within the transect. If possible, gather the seeds of each sample. Make sure to record where you retrieve each sample. While collecting your samples, make sure not to disturb the surrounding environment. Once you have acquired your samples, bring them back to the lab. From there, examine the plants and record various characteristics, as displayed on Table 1. Record the height and describe the shape, size and cluster arrangement of the leaves.
Data and Observations
Table 1
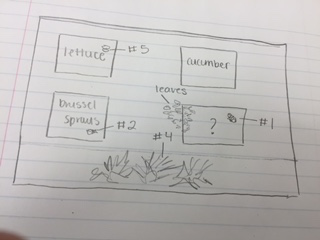
Image 1
The image above is a map depicting where within the transect the samples were taken.
Image 2
Image 2 displays each sample.
Conclusions
It was challenging to decipher if the plant’s observed were vascular and to determine their mechanism of reproduction by our small samples. I would not be surprised if some of our findings were not accurate. We also had trouble finding the seeds of our samples. If we were to repeat this lab, I believe we should collect our samples during a warmer season thus increasing the likelihood of finding seeds. All of the samples we found were vascular and represented a diverse array of plants.
JM
2/5 Lab 3 Microbiology and Identifying Bacteria with DNA Sequences
Purpose of lab
The objectives of this lab were to understand the characteristics of bacteria, to observe antibiotic resistance and comprehend how DNA sequences are used to identify species. In this lab, prokaryotes grouped in the Domain Bacteria, consisting of the Proteobacteria, Chlamydiae, Spirochetes, Actinobacteria, Firmicutres, and the photosynthesizing Cyanobacteria, were observed through a compound microscope. In order to differentiate the bacteria, the gram stain was utilized. I believe that Archaea species will grow on the agar plates because Archaea can be found in a variety of environments, due in part to its role in the carbon and nitrogen cycle. I hypothesis that the hay culture’s smell and appearance changes from week to week because the culture’s environment continues to transform and the microorganism evolve to better suit the environment
Materials and Methods
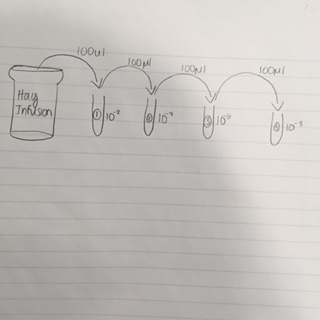
Before starting Procedure 1, observe your hay culture and write any observations and suggest a hypothesis about any changes to the smell or appearance from last week. Procedure I: 1. Observe the growth on the agar plates from Hay Infusion Culture and record in a table (see table 1). 2. Choose four nutrient agar plates, two with tetracycline and two without. Find an area within the plate with substantial bacteria growth and circle it. This is where you will draw your samples for wet mount and gram stain. 3. To make a wet mount, sterilize a loop over a flame and take a small amount from the designated sample area. Place it on the slide and make sure you label the slide with the agar type and dilution. 4. Put a drop of water over the sample and put a cover slip on top. 5. Observe the wet mount using the 10X and 40X objective. 6. Repeat this step for the remaining three agar bacteria samples 7. After you complete the wet mount procedure, begin making the gram stains Gram Stain Procedure II 1. Sterilize a loop over a flame and scrape a small amount of the growth onto the surface of the agar and mix it into a drop of water on the slide. 2. Circle this area underneath the slide as a vantage point 3. Over the staining tray, drench the smear with crystal violet and let it sit for one minute. Then rinse the stain off with water from a wash bottle 4. Cover the sample with Gam’s iodine mordant and let sit for one minute. Then rinse the stain off with a wash bottle. 5. Cover the bacterial smear with 95% alcohol for 10 to 20 seconds and then rinse gently with water. This steps ensures de-colorization 6. Douse the smear with safranin stain for 20 to 30 seconds and then rinse of excess with the wash bottle. 7. Blot the sample and allow to air-dry. 8. Observe the sample under the microscope at the 40X and the 100X immersion objective.
Data and Observations
The Hay Infusion Culture changed from last week. The smell was reminiscent of sewage and only about ½ of the water was present. Additionally, most of the objects and the film on the surface had shrunk. The culture was much clearer than it was last week and soil collected on the bottom.
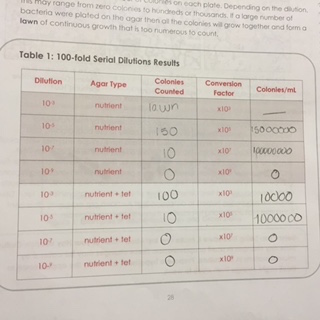
There were recognizable differences between plated colonies where tetracycline was and was not present. The colonies without the antibiotic were more abundant and lighter in color. The colonies where tetracycline existed were yellow/mustard in color and less numerous. These initial results were recorded in Table 1 (below)
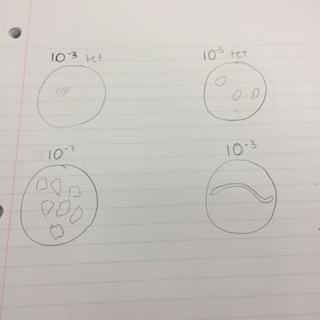
For the bacteria characterization, the following colonies were chosen: 10^-3 with tetracycline, 10^-5 with tetracycline, 10^-7 without tetracycline and 10^-3 without tetracycline The following is a drawing of the wet mount observations under the microscope at 40X.
Table 2 shows the bacteria characterization (below)
Colony A and C were gram positive meaning they have a thick layer of peptidoglycan, a polymer of amino acids and sugars, within their cell walls.
Conclusions
Tetracycline is known for its efficiency against both Gram negative and gram-positive bacteria. Before tetracycline’s wide spread use in the 1950s, most bacteria strands were susceptible to the antibiotic. Nowadays, there is a development of tetracycline resistance bacterial. The antibiotic accomplished through inhibiting the protein synthesis of bacteria cells. Our results show that Tetracycline decreases the amount of bacterial colonies and also darkens the color of the colonies.
Sources
http://textbookofbacteriology.net/environment_2.html, http://www.ncbi.nlm.nih.gov/pmc/articles/PMC99026/
JM
1/29 Lab 2 Identifying Algae and Protist
Purpose of lab: The two main divisions of all living organisms on the Earth are eukaryotes and prokaryotes. Eukaryotes have membrane bound nucleus and can be both single and multi cellular. Unicellular eukaryotes are categorized into two groups: algae and protists. The purpose of this lab is to identify microscopic and unicellular eukaryotes present in the ecosystem and niches of the Hay Infusion Culture prepared in the preceding lab.
Materials and Methods: 1. Gently move the culture to the work area. 2. Record initial observations including the smell, appearance and any possible life forms. 3. Take 3 samples from two distinct niches (meaning 6 samples) with the culture and note where these samples came from. 4. Place samples on a slide and observe under a compound microscope. 5. Determine if it is a protest or an algae, draw a picture of the organisms and describe its various characteristics with the aid of the Dichotomous key.
Data and Observations: The culture smelled like a combination of rotting vegetables and musky water. There appeared to be a thick and fuzzy film on the top of the culture. The water beneath the film was clouded and brown. Soil collected at the bottom of the jar. The first 3 samples were taken from a rotting brussel sprout. The second three samples were taken from the skin on the top of the culture. As shown in the photo below, one of the three organisms observed near the brussel sprouts could not be identified. Most of the samples taken at the surface of the culture were paramecium. The Paramecium aurelia, taken from the surface of the culture, meets all the needs of life described in the Freeman text. It feeds off of other microscopic organisms and is a P. aurelia is a unicellular protozoa. The organism can replicate in a variety of ways and its reproductive tendencies hint at the organism’s evolution.
Conclusions:
Paramecium was present in both samples. Its abundant presence could be due to the organism's propensity towards the watery environment of the hay culture. If the hay culture was to grow for an additional 2 months, I believe that the amount of organisms present would decrease. The organisms would have to compete for a limited amount of resources and only the fittest of the species would survive and reproduce.
1/26 Lab 1 Observing Evolution and an AU Niche
Purpose of lab, hypothesis and prediction The first purpose of this lab was to observe and determine if evolution moves towards increased complexity by examining three green algae members of the Volvocine Line. The second purpose was identifying biotic and abiotic components of a 20 by 20 meter transect. The purpose of the two procedures in this lab is to observe the diversity to life.
Materials and Methods Procedure I 1. Place Chlamydomonas algae on a slide and observe the organism through a compound microscope. 2. Record the number of cells, colony size, specialization of cells, mechanism of motility, and reproduction (see in Table 1). 3. Repeat steps 1 and 2 for Gonim and Volvox. Procedure II 1. Locate the assigned transect and bring a 50 mL conical tube to collect a sample of the soil/ground vegetation. 2. Upon arriving at the transect, collect the soil and documented the general characteristics of the transect, create an aerial view diagram of it and list abiotic and biotic components in the area. From here, create a hay infusion culture using the transect sample. 3. Place 10 to 12 grams of soil and vegetation sample into a plastic jar filled with 500 mLs of Deerpark water. 4. We added .1 gm of dried milk and gently mix the solution for 10 seconds. 5. Label the jar, remove the top and place in an area where it will be undisturbed.
Data and observations Procedure I Type of cell; number of cells; colony size (um); Specialization of cells?; Mechanisms of Motility; Isogamous or Oogamous Chlamydomonas; 70-100; 1 um; unicellular; flagella; isogomous Gonim; 20; 2 um; multicellular and held together by a gelatinous matrix; cilia; isogomous Volvox; 10; 3.5 um; multicellular; flagella; oogamous
The Volvocine line appears to move toward more evolutionary intricacies. Chlamydomonas, known as the origin of the Volvocine Line, display less complex evolutionary features by being both isogomous and unicellular. The colonial size in Chlamydomonas is smaller than that of Gonium and Volvox. Colonial forms are a genetic change evolving from single cells. Algae that demonstrate colonization are more evolutionary progressive. Additionally, Volvocine is oogamous, meaning it uses a more advance method of sexual reproduction.
Procedure II The transect was located in American University's Community Garden. Within this transect, there were four rectangle plots. Each plot held a different vegetable and was full of dark soil. Much of the plant life was dead but the brussel sprouts were in full bloom. There were irrigation tubes throughout and between the plots. There were dead bushes and sticks towards the southern end of the transect. Five abiotic components: wood, scarecrow, irrigation, woodchips, snow Five biotic components: cucumber, lettuce, brussel sprouts, spinach, bushes
Conclusion In Procedure I, evolution was observed among the three members of the Volvocine Line. The observed evolution moved towards increased complexity as shown by the characteristics of Volvox, the most evolved of the samples. For procedure II, the transect had various abiotic and biotic components. When observing the hay infusion of the Community Garden transect, I expect to see a variety of protists. These protists will be most likely be the fittest of the population since they survived an extreme environmental change (placed in water and stored in a moderate temperature).
JM
1/21 It’s always a good time for a nap. JM