Objective
Today's objective included the following:
- Try to create more clay-PVA beads.
- Make a calibration curve for our malachite green (MG).
- Prepare an additional PVOH film using clay solution instead of clay solid.
- Finish cross-linking films
- Dry film sample in preparation for XRD analysis
Protocol
1. Synthesis of PVOH-clay beads
The full procedure for making the PVOH beads can be found here, instead using 20% clay. The procedure we used, with our altered reagent amounts, can be found below:
- 100 mL of ethyl acetate and 2mL of glutaraldehyde was added to a large dish
- 0.500 g of bentonite clay was combined with 30mL of 10% HCl and was mixed, while heating, until all PVA crystals have dissolved.
- The heated PVOH-clay solution was then added, dropwise, to the ethyl acetate and glutaraldehyde solution using a burette. The solution needs to be vigorously stirring during this step.
We did not continue the rest of the procedure because the beads did not correctly form....again.
2. MG calibration curve
We first diluted our 200ppm stock solution to the following dilutions: 20ppm, 10ppm, 2ppm, 1ppm, and 0.2ppm. However, our 20ppm and 10ppm dilutions gave us an absorbance greater than 1, so instead we substituted those two data points for 8ppm and 5ppm dilutions. Our UV Vis spectra and calibration curve can be seen found below.
3. Preparing PVOH films
Films of polyvinyl alcohol (PVOH, or PVA) were created using the following protocol:
- 1 g of PVOH, 3.33mL of 3% weight bentonite clay solution, 0.5mL of 8% glutaraldehyde, and 8.67 mL of DI water was heated, while stirring and covered with a watchglass, until al l PVOH crystals have been dissolved.
- After the crystals were completely dissolved the solutions were transferred to a Teflon dishes and placed in the fume hood to dry.
4. Cross-linking PVOH films
Film cross-linking from 9/07/2016 was only performed on two of the three films, so today we finished cross linking the third film. The procedure used can be found here.
5. Drying film sample
The 0.5mL 8% glutaraldehyde film was weighed and placed in an over at 100°C for 2 hours. After it dried, it was reweighed to analyze the swelling of the film. The following weights are below:
- Patted dry film: 0.3553 g
- Oven dried film: 0.1111 g
Observations
Our beads did not form, again. The solution was still very lumpy. We realized this is likely because we added the glutaraldehyde too early, it needed to be added AFTER the beads were made, not before. Because of this, they were probably cross linking too soon and that's why large lumps instead of beads kept forming.
Figure 1: UV Absorption of Malachite Green

Figure 2: Calibration Curve for Malachite Green
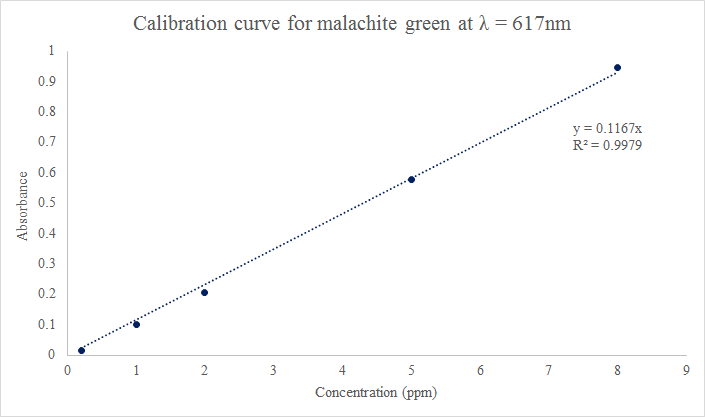
Based on the data in Figure 2, the extinction coefficient will be 1.167*10-3. Since the R2 value for our line of best fit is 0.9979, or a 99.79% fit, we don't necessarily need more data points since this number is extremely high.
|

