UMSL Genomics:Research
RNA Virus Discovery In Wild Birds of Northeastern Brazil

This research will be performed to help prediction and prevention of outbreak that could affect avifauna. We will use RNA-Seq to identify novel viral agents in wild birds. For this we will use RNA extracted from cloacae and oropharyngeal swabs from different bird species of `Caatinga` and Atlantic Forest biomes of Northeastern Brazil. RNA will be purified, reverse-transcribed into cDNA, and sequenced using the Illumina platform. The goal is to identify putative viral agents by using the Trinity program to assemble transcriptomes de novo from RNA-seq data and command-line BLAST to differentiate between bird and virus contigs.
Groucho
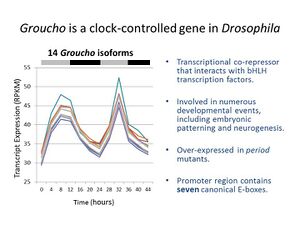
Circadian clocks exist in many organisms and are rhythmically regulating expression of genes responsible for a variety of cellular processes. The Groucho gene is very important in the early stages of development in fruit flies and it is known to be rhythmically expressed in the fly brain. How Groucho is regulating circadian genes is less clear.
Using RNAi, knock down experiments are performed using genes known to regulate circadian rhythms. The long term goal is identification of genes driven by the circadian clock, the mechanisms through which regulation occurs and how these genes influence behavior.
More specifically,
1. Identification of genes that contribute to regulating Groucho
2. Identification of circadian output genes Groucho has a role in regulating.
Computational simulations to determine the best parameters for profiling circadian transcriptional rhythms
RNA-Sequencing (RNA-Seq) is becoming extensively used to systematically profile transcripts’ cycling pattern. However, it consumes a great deal of time, money, and resources, and analysis of the huge amounts of data is not trivial. Meanwhile, it is not always necessary to generate maximum data sizes for each experiment. Our aim is to determine an appropriate balance where we can get enough data for our research with a considerably low cost.
Based on experimental data, we are doing computational simulation to test the right depth of RNA-Seq for circadian expression study. Depth is the time that a nucleotide gets sequenced. It is determined by genome length, average read length, and reads number. We are testing the robustness and accuracy of the post-analysis results under different reads numbers since we are working specifically with Drosophila, Illumina MiSeq. There are other factors, like organism, sample density that also affect the results. We will also evaluate these factors later in our project.
Phylogenetics of Burmeistera (Campanulaceae)

Many tropical groups of plants have ‘explosively radiated’, forming multiple species in a relatively short amount of time. The Muchhala Lab aims to evaluate the importance of different factors in the formation of new species for the diverse genus Burmeistera. In work led by Monica Carlsen, we will begin by inferring a detailed, species-level phylogeny for the group using approximately 100 different nuclear, mitochondrial, and plastid DNA regions. Total genomic DNA for the 115 known species in the genus will be enriched with probes that target these 100 gene regions and sequenced with the Illumina MiSeq. This will provide large amounts of data for phylogenetic inference, which will allow estimation of time since speciation for pairs of Burmeistera species. Future experimental work will test the strength of reproductive barriers between these pairs, including pollinator-mediated, gametic, and postzygotic barriers.
Investigation of Endo-siRNA biogenesis using the Illumina MiSeq Platform
In the past decade, the study of small RNAs (microRNAs, piwiRNAs, siRNAs) has become increasingly critical to our understanding of protein regulation and general cellular function. Many of these small RNAs, such as microRNAs, have been well studied. However, a new class of small interfering endogenous RNAs (endo-siRNAs) has recently been discovered and has yet to be well characterized. To further investigate these endo-siRNAs, small RNA libraries were prepared and sequenced using the Illumina MiSeq. Data from this experiment will allow us to determine what proteins are involved in endo-siRNA biogenesis as well as elucidate the mechanism by which they are produced.
Differential gene expressions and metabolic analysis in Arabidopsis using RNA Seq

Nitrogen (N) is one of the most important nutrients for plant growth. Phospholipase Dε (PLDε) is known involved in the nitrogen utility process. The overexpression of PLDε promotes N uptake and accumulates more biomass than wild type (WT), whereas pldε knockout exhibited worse growth than WT. However, the underlying molecular mechanisms is still in veil. RNA seq technology provides a powerful way for us to further investigate the role of PLDε in nitrogen responses at the whole genome level. Using RNA samples extracted from wild type (WT), pldε knockout and PLDε overexpression (PLDε-OE) seedlings grown in media containing 6 mM N and 0.6 mM N, the RNA seq experiment was performed afterwards. The results offer every single transcript expression level in the whole genome. Differential gene expression patterns and cellular metabolic omics results will be obtained from the RNA seq analysis. It is very interesting and exciting to look for the potential key molecules and/or pathways connecting the function of PLDε and N assimilation.
Identifcation of the number of individuals and cultivation history of Dracaena umbraculifera

Dracaena umbraculifera is plant species that is thought to be native to Mauritius but extinct in the wild. It is currently known from around 20 specimens in botanical gardens around the world, including several specimens in the Climatron at the Missouri Botanical Garden. However, the identification of all of these specimens in botanical gardens around the world is questionable because an inflorescence is needed for identification but the plant does not flower in greenhouses. Furthermore, collection information for all of these specimens is missing. The conservation genetics lab at the Missouri Botanical Garden is using DNA barcoding and genotyping using the Miseq at UMSL to determine the number of specimens that are correctly identified and to track the cultivation history of the species. Our hypothesis is that all the individuals in botanical gardens originated via a single collection in the 1700s that was subsequently distributed to all other botanical gardens across the world.

UMSL Genomics
Department of Biology
University of Missouri, St. Louis
