The BioBricks Foundation:Standards/Technical/Measurement/Promoter characterization experiment (FACS)
From OpenWetWare
Jump to navigationJump to search
FACS protocol V0.1
Materials
- 5 promoter test strains provided as stabs from Endy Lab
- <bbpart>I20259</bbpart>,<bbpart>I20260</bbpart>,<bbpart>I20268</bbpart>,<bbpart>I20269</bbpart>,<bbpart>I20270</bbpart>
- TOP10 background strain (Jason will provide so we're all actually using the same one).
- 100 mM sodium phosphate, 150 mM sodium chloride, pH 7.4 (PBS)
- 5 mL polystyrene round bottom tube with cell strainer cap: BD Falcon #352235
- Jason R. Kelly 13:37, 15 February 2008 (EST):What's the cell strainer cap for? Don't normally use that.
- CAjoF 12:42, 18 February 2008 (EST): The cell strainer cap is make sure there are no big particulates in the sample that may clog the FACS. It is more important for fixed cells, but it certainly never hurts.
- Supplemented M9 Media
- Julius B. Lucks 18:49, 3 April 2008 (EDT): The exact ingredients we used for the minimal media can be found here: Julius_B._Lucks/M9_Supplemented_Minimal_Media
Culturing of the strains
- Streak LB+Kan plates of the 5 test strains and an LB (no antibiotic) for TOP10
- Julius B. Lucks 19:02, 3 April 2008 (EDT): This was done for us by CAjoF. Plates were stored in the 4C refrigerator for 2 weeks.
- For each test construct add 5ml of supplemented M9 medium (w/glycerol, see preparation protocol) and Kanamycin (20ug/ml) to three 17mm test tubes (so 15 tubes total). Add 5ml of supplemented M9 medium (w/glycerol) with no antibiotic for the TOP10 negative control cells to three 17mm test tubes.
- how many replicates do we want to do (X)?
- I would suggest 3 to start. If the replicates are wildly different, we can move to more.CAjoF 12:45, 18 February 2008 (EST)
- Inoculate media by picking single colonies from plates. Grow cultures for 20 hrs at 37°C with spinning at 70 rpm.
- Kim de Mora I had trouble using culture sticks to innoulate the first time I did the experiment. There was a lot of debris that was picked up by the machine. Works much better with tips.
- Dileep D. Monie 13:49, 1 April 2008 (EDT): 70 RPM seems a bit slow. Any reason why I shouldn't shake more vigorously?
- Jason R. Kelly 14:47, 1 April 2008 (EDT): I think you should be OK using whatever RPM you normally use. The variability between shakers/rollers makes me guess we couldn't easily standardize this if we wanted to.
- In the morning dilute 100x into 5ml of pre-warmed (37C) fresh media to get cells back into log phase and let grow for 4hrs under same conditions as overnight
- Does the back-dilution use media containing antibiotic for the 5 test strains? CAjoF 16:11, 29 February 2008 (EST)
- Jason R. Kelly:Yeah, they should contain Kan.
- Does the back-dilution use media containing antibiotic for the 5 test strains? CAjoF 16:11, 29 February 2008 (EST)
- The cultures should be at an OD of 0.2 - 0.4 after 4hrs.
Live Cell FACS Procedure
- When your 5 mL culture reaches OD600 ~ 0.300, pellet the cells by centrifuging 1ml of each culture in an eppendorf at 3000xg at room temperature for 5 minutes.
- Kim de Mora: I did this with an OD of 0.1 and with 250 μL of washed cells, still had 90 000 events/sec on our LSR-II.-
- Remove the supernatant.
- Resuspend the cell pellet in 1 mL PBS.
- Add 250 uL washed cells through the cell strainer lid into a 5 mL polystyrene tube. To get the solution to pass through the strainer, apply slight pressure to the strainer lid with the pipet tip as dispensing the cell solution.
- Add 250 uL PBS through the cell strainer lid into the 5 mL polystyrene tube.
- Place cells on ice.
- Analyze cells by flow cytometry using 488 nm excitation as quickly as possible.
Analysis of FACS Data
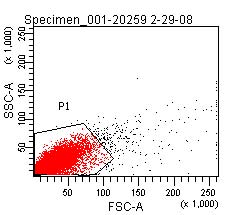
- Using the signal from the TOP10 cells, create a gate (P1, in image below) in the forward scatter-side scatter plot that captures the core of the scattering events.
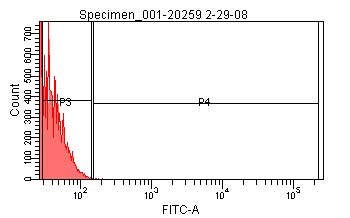
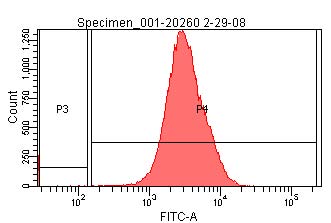
- Using the signal from the TOP10 cells, create a second gate (P3, in image below) on the fluorescence channel which includes 99% of the total population and a third gate (P4, in images below) which encompasses all greater fluorescence signals.
- Copy these gates to all other samples.
- Determine the geometric mean of fluorescence intensity of the P4-gated population in each sample, <Isample,P4>.
- Dileep D. Monie 18:38, 3 April 2008 (EDT):The geometric mean of I20259 should be calculated using the P3-gated population.
- Determine the geometric mean of the P3-gated population of the TOP10 cells, <ITOP10,P3>. This corresponds to the mean background fluorescence intensity of the cells.
- Calculate the fluorescence intensity in consensus Biobrick GFP units = I/II20260 = <Isample,P4>-<ITOP10,P3>/(<II20260,P4>-<ITOP10,P3>).