Talk:CH391L/S12/Pigments
From OpenWetWare
Jump to navigationJump to search
- Joe Hanson 12:03, 26 March 2012 (EDT):I had the pleasure of talking with Daisy Ginsburg of E. chromi fame when she stopped by the SXSW event, and she offered a couple good insights to consider when using pigment genes. First, how do you determine the density of bacterial growth in order to observe each pigment? We tend to view these reporters as "easy to see", but considering that a typical bacterial colony has 106 to 109 cells inside of it, some would say it's a pretty poor phenotype for sensitive assays. She said the E. chromi poop idea probably would never work because the number of bacteria you'd have to get into the eventual "product" is just silly high. Also, one has to assume that these pigments are deposited inside the cell and not secreted. Is there a toxicity limit or a turnover? One thing that she or I hadn't seen before is the use of these pigments in liquid culture. Anyone got anything?
- Yi Kou 14:05, 26 March 2012 (EDT): For characterization of growth or pigment yield, I think flow cytometry can be applied. For the "number of bacteria" issue, I think there are several possible ways, one is to dilute the sample(/serial dilution) so that the number of bacteria needed is relatively decreased. Also, one can assemble as much as bacteria into a semipermeable membrane cube or micelle or whatever machinery made from chemical engineering field. Another way is to transform the pigment chemically to sth easy or sensitive to detect in order to decrease number of bacteria required. For the pigment toxicity, since there exists no innate catabolism pathway, I think it would definitely pose burden on the new host. However, E.coli is chosen because of its great tolerance for this(similar to its application in production of foreign proteins). I think the extent it can hold production burden is much more efficient than the production of those machinery in it. At least so far so good, otherwise, the colonies would not be lawn like on these projects. I consider building in catabolism/self suppressing circuit would be a messy thing for it, as learned from Dr. Ellington's speech. So for addressing internally accumulated product that cannot leak outside, maybe one way is using membrane permeable reacting reagent from outside(or photolysis, but it may induce mutation). For secreting pigments, I think removal of the product would shift production equilibrium. And certainly, appropriate removal method is the key.
- Jeffrey E. Barrick 13:09, 25 March 2012 (EDT):Please add references to your figure captions.
- Yi Kou 07:43, 26 March 2012 (EDT):Added.
- Jeffrey E. Barrick 13:09, 25 March 2012 (EDT):For the chromobacteria section, many of these pigments seem to have evolved because they are antibacterial agents (like pyocyanin). It doesn't really matter if these are colored or not for their function. Aside from uv protection and harvesting light energy, I guess color is a random side-effect of the molecular structure in many cases. Is there also a connection that colored compounds (such as heme) are often redox-active?
- Yi Kou 07:43, 26 March 2012 (EDT): I did some research, it is common for a pigment to be redox related if the pigment is generated as antibiotic for competing other organisms (and often these are related to either disturbing or maintaining the inside reducing power), famous example is pyocyanin from phenazines[1],other examples can be seen[2][3].
- Jeffrey E. Barrick 13:09, 25 March 2012 (EDT):We've also talked about how sometimes symbiotic bacteria lend their properties to multicellular animals, like the example of luciferase and squid. I wonder if any colors in higher animals are derived from bacterial symbionts in specialized organelles or horizontal transfer of bacterial genes, rather than from eukaryotic .
- Yi Kou 09:01, 26 March 2012 (EDT): I did some research, but not so satisfying: using symbionts to show the color are mainly the marine animals. Besides squid, angler fish is another one. There other two I can find is one carotenoid pigment and one heme proteins. I do not think they are involved in gene transfer.
- Jeffrey E. Barrick 13:09, 25 March 2012 (EDT):Wow, those radiotrophic fungi are crazy.
- Yi Kou 07:54, 26 March 2012 (EDT):Yes, I think they are the "toughest" organisms. A link for this: Radiotrophic fungi. Is it possible to make sth. out of this?
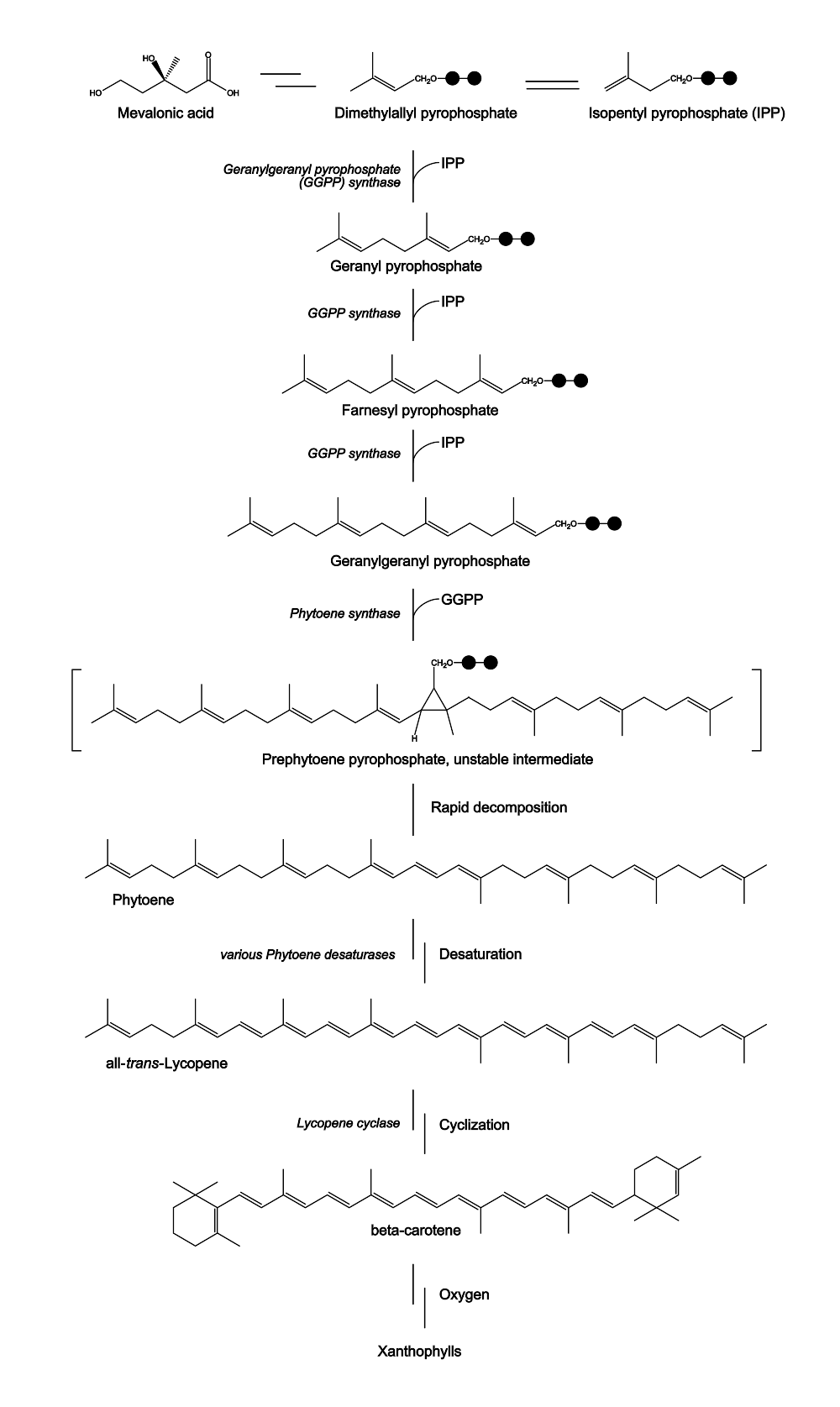
- Ben Slater 16:37, 25 March 2012 (EDT): You mentioned in your presentation that you weren't sure why β-carotene is missing the typical isoprenoid structure in the center. This occurs because the isoprenoid condensation joining the two 20-carbon precursors is a head-to-head condensation instead of head-to-tail as in the previous steps. A good way to spot this is the release of two pyrophosphates instead of one. Another good way indicator is the existence of the characteristic "forked tongues" on both ends instead of just one end.
- Yi Kou 08:43, 26 March 2012 (EDT):I find this needs a little bit more explanation. β-carotene is not missing the typical isoprenoid structure, I was wondering at the time of counting the right 8 isoprene units by counting the double bond (which is usually the method for terpenoid, as is often seen in the steroid synthesis, esp lanosterol). Actually, head to tail or head to head does not have relation to how many double bonds/typical isoprene there are in the structure. Head to tail can also result in right number, and also with release of the two PPis (esp. lanosterol or other steroids I remember). β-carotene has nine double bonds out of 8 isoprene building block because of an cyclopropane containing prephytoene intermediate formed during the synthesis (catalyzed by phytoene synthase using GGPP to make phytoene). Chemically, I think this kind of formation is not favored (though releasing high energy PPis), but may be under phytoene synthase, the cyclization is favored with delta G compensated biochemically or structurally (since head to tail connection easily result in future multi-ring structure).
Ref
<biblio>
- pyocyanin pmid=17526704
- Mutactimycin pmid=15323124
- unknown pmid=20502566