HEPES: Difference between revisions
From OpenWetWare
Jump to navigationJump to search
illustration |
No edit summary |
||
| Line 1: | Line 1: | ||
[[Image:HEPES structure.png|right|thumb|Chemical structure of HEPES = N-(2-'''h'''ydroxy'''e'''thyl)<br>'''p'''iperazine-N’-<br>(2-'''e'''thane'''s'''ulfonic acid);<br>aka 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid; CAS number: 7365-45-9]] | [[Image:HEPES structure.png|right|thumb|Chemical structure of HEPES = N-(2-'''h'''ydroxy'''e'''thyl)<br>'''p'''iperazine-N’-<br>(2-'''e'''thane'''s'''ulfonic acid);<br>aka 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid; CAS number: 7365-45-9]] | ||
HEPES is a common buffering chemical similar to Tris in Tris-HCl buffers and phosphates in PBS. Buffers are used to keep the pH at a certain value and can "buffer" the addition of small amounts of acids/bases. | |||
A ''Good'' buffer [Good'66 PMID 5942950, Good'74 PMID 4206745, Blanchard'84 PMID 6717292]. | A ''Good'' buffer [Good'66 PMID 5942950, Good'74 PMID 4206745, Blanchard'84 PMID 6717292]. | ||
== Property of HEPES == | |||
Stable pH vs. temperature, no primary amine groups, no metal chelation, near physiologic pH range. HEPES is often used to maintain protein solubility in biochemical experiments. | Stable pH vs. temperature, no primary amine groups, no metal chelation, near physiologic pH range. HEPES is often used to maintain protein solubility in biochemical experiments. | ||
| Line 11: | Line 16: | ||
* ΔpK<sub>a</sub>/ΔT = -.014 | * ΔpK<sub>a</sub>/ΔT = -.014 | ||
* HEPES contains tertiary amines, which are reactive under certain conditions. | * HEPES contains tertiary amines, which are reactive under certain conditions. | ||
* Chemical formula: C<sub>8</sub>H<sub>18</sub>N<sub>2</sub>O<sub>4</sub>S | * Chemical formula: C<sub>8</sub>H<sub>18</sub>N<sub>2</sub>O<sub>4</sub>S | ||
Revision as of 15:46, 2 November 2011
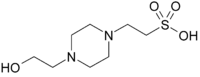
piperazine-N’-
(2-ethanesulfonic acid);
aka 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid; CAS number: 7365-45-9
HEPES is a common buffering chemical similar to Tris in Tris-HCl buffers and phosphates in PBS. Buffers are used to keep the pH at a certain value and can "buffer" the addition of small amounts of acids/bases.
A Good buffer [Good'66 PMID 5942950, Good'74 PMID 4206745, Blanchard'84 PMID 6717292].
Property of HEPES
Stable pH vs. temperature, no primary amine groups, no metal chelation, near physiologic pH range. HEPES is often used to maintain protein solubility in biochemical experiments.
- pKa at 25C of 7.55 (7.31 at 37C)
- a second pKa at pH 3 is not of interest
- usable buffering range of 6.8 to 8.2
- molecular weight 238.3
- ΔpKa/ΔT = -.014
- HEPES contains tertiary amines, which are reactive under certain conditions.
- Chemical formula: C8H18N2O4S
Buffers are typically 1 M, prepared by neutralizing HEPES with sodium hydroxide. HEPES is essentially insoluble until it is neutralized. and