Multilayer Paper Microfluidics - Madyson Redder
Overview
Multilayer paper microfluidics (MLPM) are a type of microfluidic system that uses multiple pieces of paper stacked together that can test multiple fluids for multiple different things at once, without mixing. MLPM devices can run more complex tests than single layer paper microfluidic devices. The results are displayed side by side, which can be used for easy comparison, and the devices are cheaper and more efficient than both 3D polymeric/glass and single-layer paper microfluidic systems. [1]
Advantages
MLPM devices rival other devices, such as 3D polymeric, glass, or single layer paper microfluidic devices in the sense that they do more. 3D polymeric or glass microfluidic devices were created to run tests on small amounts of liquid and receive results in a timely manner. However, these devices are costly and time consuming to produce. A solution to this problem was single-layer paper microfluidic devices. The most common known examples of single-layer paper microfluidic devices are pregnancy tests, COVID-19 antigen tests, and glucose test strips. While these devices are beneficial in that they are low cost, easily accessible, and provide results in a timely manner, they typically only conduct one test at a time. What if a paper microfluidic device could run multiple tests simultaneously with a number of different samples? What if it could run a multistep reaction at once?
MLPM devices can conduct multiple tests of different samples at once and show side-by-side comparison.[1] They can also conduct more complex tests than single layer paper microfluidic devices. An example of a single-layer paper microfluidic device is a pregnancy test. A pregnancy test has a test and control area, and it uses the HCG hormone in urine to detect pregnancy. If it is positive, a line appears in both the test and the control area, and if it is negative, only one line appears in the control area. However, it does not indicate the concentration of HCG, which is an indicator to how far along someone is in their pregnancy.
MLPM devices have the ability to determine concentrations of the desired substance as well as changes in the structure of the desired substance. Changes in concentration of protein biomarkers can be indicators of infectious or non-infectious diseases; however, detecting those changes is not as simple as reading one or two lines on a urine test stick. To determine the concentration of proteins in a sample, enzyme-linked immunosorbent assays (ELISA) are used, which are single layer paper microfluidic devices.[2] The determination of the concentration of proteins requires multiple steps with several different single layer paper microfluidic devices. By creating a ELISA with multiple layers, these steps could be performed sequentially, reducing the risk of human error moving the sample between different devices, saving time and money, and reducing waste.[2]
Fabrication Methods
3D Device Fabrication
Paper Stacking
In a study done to observe rapid flow in MLPM devices, hydrophobic wax barriers were created on cellulose paper to form channels for fluids to flow through.[3]. Pieces of paper were stacked on each other, with pieces of double-sided tape placed in between each layer of paper. Holes were cut into the double-sided tape to ensure each channel could travel between layers both vertically and laterally without mixing with other channels. Packing tape was used to seal the outside of the µPADs.[3] The gaps between each layer of paper, due to the packing tape barrier, showed increased flow rate of the fluid throughout the channels. The gap height affected the flow rate of the fluid, with an increased gap height correlating to an increased flow rate. The maximum flow rate was achieved with a 234 µm channel height; any height higher showed a decrease in flow rate.[3]
Origami Folding
When assembling MLPM devices, double-sided tape is used to secure the pieces of paper together. However, only one layer of the paper can be used for colorimetric detection as the device cannot be dismantled, and it requires additional tools to cut the holes into the tape to ensure vertical wicking. Another study on MLPM devices uses the Japanese art of origami to assemble a MLPM device using a single sheet of paper folded into multiple layers, called oPADs, leading to easy assembly and dismantling.[4] The channel designs were created using photolithography, and the piece of paper was folded into nine layers with four inlet reservoirs. The layers could be dismantled back into a singular sheet of paper to show multiple layers with colorimetric detection without compromising the device.[4] Origami assembly, as stated earlier, eliminates the need for tape, which could potentially lead to contamination and unwanted adsorption.[4] The oPAD could also be assembled in under a minute because it did not require additional tools for assembly.
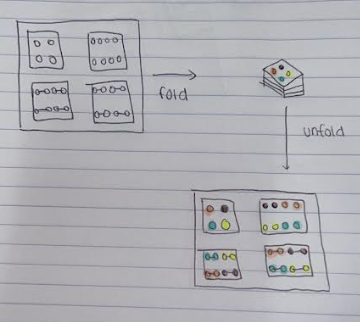
Channel Fabrication
While there are several different kinds of channel fabrication methods, this section will discuss the main channel fabrication methods for MLPM devices.
Cutting
The simplest way to create channels is by cutting. Either a knife cutter or a CO2 laser cutter is typically used, but scissors or razor blades can also suffice. Cutting, however, is not the most reliable way to create channels as the channels often require tape or glass slides to make the device more stable.[2]
Wax Printing
Wax printing uses wax instead of ink in an ink printer and prints the wax onto the surface of paper. The paper is then heated to melt the wax and allow it to penetrate through the surface of the paper. It is an efficient and low-cost method for creating channels on paper.[2]
Photolithography
A piece of paper is covered with "a negative photoresist [and] expos[ed] to UV light through a photomask to crosslink the photoresist in the desired pattern."[2] The paper is then developed in a solvent to remove any extra photoresist. Photolithography, while one of the most laborious techniques for creating channels, is known for its ability to create precise and high resolution channels.
Uses
Developing Countries and Travel
Access to quick and accurate healthcare is not readily available in certain situations. MLPM devices are useful in that they are small, lightweight, and can be easily stacked, transported, and stored.[1] Situations in which multilayer paper microfluidic devices would be useful are in developing countries in which access to healthcare is scarce and for people who are traveling or in the military.
Additionally, MLPM devices can be used to perform more complex tests that require multiple reactions. An example of a device created for pharmaceutical purposes was a MLPM electrochemical device that can detect both glucose levels and beta-hydroxybutyrate (BHB) in diabetics.[5] BHB is a ketone body, which is produced from uncontrolled diabetes. Too much BHB and other ketone bodies can lead to diabetic ketoacidosis, which has severe side effects and is often fatal if not detected soon enough.[5] While there have been developments for BHB at-home detectors, they are not available worldwide and are costly. BHB requires a multistep reaction to detect it, so the solution to this problem was a MLPM electrochemical device that takes the shape of a greeting card, which can be inserted into a glucometer. Using origami, folding the paper into the greeting card shape allowed for the enzyme reaction to occur on the top sheet while the card was open, and when the card was closed the sample could then travel through the electrode detection zones to detect the BHB level. While the card is open, there is no fluid flow to the electrodes on the bottom half, ensuring there was no premature flow to the electrodes.[5] Not only would this device cost less than traditional BHB tests, it is easy to assemble and use due to the simplicity of only needing to open and close it.
Pharmaceutical Industry
The pharmaceutical industry also benefits from MLPM devices; instead of testing 4 different samples on 4 different devices, each sample can be tested on the same device. Using one device over multiple is beneficial in reducing waste, saving money, and being able to compare results to each other easier.
By utilizing a MLPM device, one study was able to create a device that detected four different cancer biomarkers from one sample. Using two pieces of paper stacked on each other and different electrode zones, the device was able to detect the different biomarkers at once and at different concentrations.[6]
Wildlife and Habitat Conservation
Rather than removing animals and plants from their natural habitats, MLPM devices are also beneficial for running multiple test on animals or plants in the wild where advanced medical equipment cannot be accessed .
References
- ↑ 1.0 1.1 1.2 Martinez, A. W.; Phillips, S. T.; Whitesides, G. M. Three-Dimensional Microfluidic Devices Fabricated in Layered Paper and Tape. Proceedings of the National Academy of Sciences 2008, 105 (50), 19606–19611. https://doi.org/10.1073/pnas.0810903105.
- ↑ 2.0 2.1 2.2 2.3 2.4 Nishat, S.; Jafry, A. T.; Martinez, A. W.; Awan, F. R. Paper-Based Microfluidics: Simplified Fabrication and Assay Methods. Sensors and Actuators B: Chemical 2021, 336, 129681. https://doi.org/10.1016/j.snb.2021.129681.
- ↑ 3.0 3.1 3.2 Channon, R. B.; Nguyen, M. P.; Scorzelli, A. G.; Henry, E. M.; Volckens, J.; Dandy, D. S.; Henry, C. S. Rapid Flow in Multilayer Microfluidic Paper-Based Analytical Devices. Lab on a Chip 2018, 18 (5), 793–802. https://doi.org/10.1039/C7LC01300K.
- ↑ 4.0 4.1 4.2 Liu, H.; Crooks, R. M. Three-Dimensional Paper Microfluidic Devices Assembled Using the Principles of Origami. Journal of the American Chemical Society 2011, 133 (44), 17564–17566. https://doi.org/10.1021/ja2071779.
- ↑ 5.0 5.1 5.2 Wang, C.-C.; Hennek, J. W.; Ainla, A.; Kumar, A. A.; Lan, W.-J.; Im, J.; Smith, B. S.; Zhao, M.; Whitesides, G. M. A Paper-Based “Pop-Up” Electrochemical Device for Analysis of Beta-Hydroxybutyrate. Analytical Chemistry 2016, 88 (12), 6326–6333. https://doi.org/10.1021/acs.analchem.6b00568.
- ↑ Wu, Y.; Xue, P.; Kang, Y.; Hui, K. M. Paper-Based Microfluidic Electrochemical Immunodevice Integrated with Nanobioprobes onto Graphene Film for Ultrasensitive Multiplexed Detection of Cancer Biomarkers. Analytical Chemistry 2013, 85 (18), 8661–8668. https://doi.org/10.1021/ac401445a.
