Lidstrom:Colony PCR
From OpenWetWare
Jump to navigationJump to search
Back to Protocols
One Recipe:
- make sure the boiling and extension temperatures listed below match those of your kit. Most inexpensive polymerases are Taq based, so this Taq protocol should workThis is a Taq based protocol.
- The Lidstrom Lab likes OneTaq 2X master mix
- for 25 uL: 12.5 uL 2X master mix, 0.5 uL 10 uM forward primer, 0.5 uL 10 uM reverse primer, 11.5 uL H2O
- for 20 uL: 10 uL 2X master mix, 0.4 uL 10 uM forward primer, 0.4 uL 10 uM reverse primer, 9.2 uL H2O
cycling:
- 5 min at 95oC (initial lysing of cells & melting of DNA)
- 1 min at 95oC (melting)
- 1.5 min at 55 oC (annealing)
- 1 min/kb at 68oC (OneTaq extension temp: use the one for your polymerase)
- repeat steps 2-5 30-40 times
- 5 min at 72°C (once)
- 4°C forever
note: the times for melting and annealing can be cut down to speed up the process if desired.
General:
- Use Taq if you aren't going to use the DNA you amplify for a construct. Taq is cheap. Do not use Phusion (~$1/50uL rxn). Taq is less accurate & less fast but that's fine.
- The thermocycling denaturation & extension temperature should be that recommended by your particular PCR kit. I have seen 68oC & 72oC.
- The primer concentration varies in different people's recipes. Janet hasn't tested variations.
- One fatal flaw: overloading the PCR. Just a tiny piece of a colony should be used.
- If they aren't working, you can put them in tubes, put them at 95oC for a while, then centrifuge they lysed cells so all the debris falls out. Run PCR from on the aqueous portion.
- Janet is using NEB's OneTaq quick-load (not hot start) & their recommended protocol
- manual: Annealing temperatures can be optimized by doing a temperature gradient PCR starting 5°C below the calculated Tm
Parameters:
- T_anneal: up to you. For VF2 & VR 54-56oC works well.
- t_anneal: use 30s/kb and round up.
- rxn volume: 10-25 uL. Too small of a volume can result in overloading without noticing.
Comments
Tips
- Make a master mix of water + primer in the correct ratio so you can do repeat colony PCRs with ease. These mixes can be stored in the fridge for months without trouble.
- OneTaq 2X Quick-Load lasts weeks/months in the fridge; no need to freeze/thaw for each use.
- Don't overload the mix with cells. The cell debris can overload the polymerase.
- Liquid culture can inhibit the reaction
- This experiment shows that 2uL of [[Lidstrom:Autoinduction_Media|autoinduced culture] inhibits the ability of OneTaq 2X Quickload to produce DNA.
- *
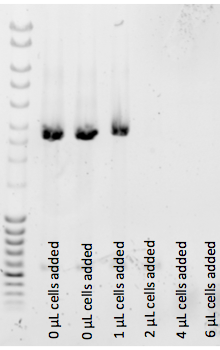
All wells contain miniprepped plasmid template (<<1ng/well) and varying amounts of 2-day autoinduced BL21(DE3) culture containing that same plasmid was added in varying amounts. 1uL of that culture still allowed the 12uL PCR to work, but 2uL and above inhibited badly.
Old stuff
- It might be tempting to make a 1X soltuion with primers included, store it in the freezer, and thaw it as you need to use it. The problem with this: primer dimers may amplify. Obviously the degree to which this is an issue will depend on your primers, but it is likely worth avoiding by sticking with the 2X freezer stock. Also, there may be issues with the protein's stability upon freezing in a lower buffer/stabilizer concentration.
- From Justin Siegel 2/6/2012: "The problem isn't the associate primers, it that when they associate and the polymerase extends them. Then your reaction can get overrun since now extended primer dimers can act a perfect template in future amplifications and will preferably amplify over larger pieces. So if there is primers+enzyme but no template you run a really high risk of getting primer dimers."
- For what it is worth, my mix that includes the primers VF2 & VR works great. -Janet 2/7/2012