Lab 05: Electron Charge to Mass Ratio
Summary
The quantity representing the ratio of an electron's charge to it's mass was experimentally sought after for the first time by J. J. Thomson in 1897 using cathode ray tubes. He is also credited with the discovery of the electron itself. Using a gas sample between two charged plates Thomson was able to observe a current as the electrons interacted with the gas atoms and caused them to ionize. By applying a magnetic field, he could alter the path of the current and observe the change. Using Maxwell's theories and some geometry he was able to calculate the charge to mass ratio of an electron based on measurements of the current's path.
In our lab we used an updated apparatus. In place of the cathode tubes, we had a glass tube full of a very dilute helium gas. This tube is surrounded by a Helmholtz coil that can supply a nearly uniform magnetic field throughout the whole tube. In this tube, we released electrons from a heater plate, focus them into a nearly coherent beam, and apply a varying magnetic field. This varying magnetic field changed the trajectory of the electrons, which emit light from the collisions with the helium, so that it formed a complete circle. We then measured the dimensions of the circular path, and the voltages used to free the electrons to determine the e/m ratio.
- In this lab I worked with Brian P. Josey.
- These are my Detailed Lab Notes.
Analysis
To determine the magnetic field, we used the following formula:
[math]\displaystyle{ B = \frac {\mu R^2 N I}{(R^2+x^2)^{3/2}} }[/math]
where: B - the magnetic field, in Telsa, μ - permeability of free space, 4π * 10-7 weber/ amp-meter2, R - radius of the rings, 0.15 m, N - number of times the wires go around the Helmholtz coil, 130 times, I - current measured in A, x - the axis where the measurement is taken, in this case x=R/2
[math]\displaystyle{ B = (7.8*10^{-4} \frac {weber}{amp-meter^2} )* I }[/math]
In order to find the e/m ratio from the data, we need to find the relationship of the ratio of e/m to our data. Using the follow formulas we can determine the e/m ratio:
[math]\displaystyle{ eV= \frac {mv^2}{2} }[/math]
[math]\displaystyle{ F_B = qvB \, }[/math]
where: e and q - the charges of the electron, V - the acceleration potential, v - the velocity the charges travel at, FB - the force due to the magnetic field, and B - the external magnetic field
Together, these equations give the e/m ratio:
[math]\displaystyle{ \frac {e} {m} = \frac {2V} {r^2 B^2} }[/math]
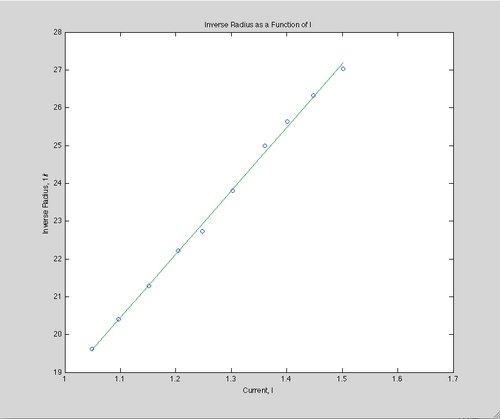
The first method to find the e/m ratio from the data is to plot the inverse of the radius versus the current at a constant accelerator voltage. This gives a simple linear relationship:
[math]\displaystyle{ \frac {1} {r} = \sqrt {\frac {(7.8*10^{-4})^2}{2V} \frac {e}{m}} I }[/math]
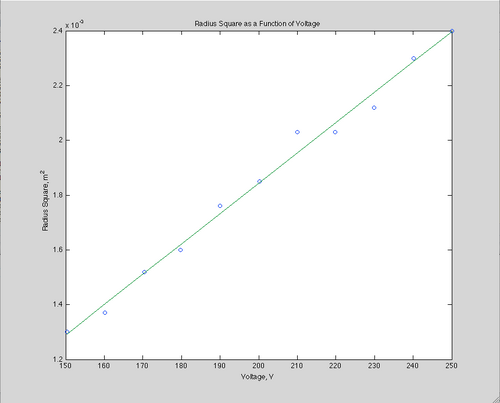
The second method to find the e/m ratio from the data is to graph the radius squared versus the acceleration potential at a constant current. This linear relationship is given by:
[math]\displaystyle{ r^2 = \frac {2V} {(7.8*10^{-4})^2 I} * \frac {m}{e} }[/math]
Using MATLAB for linear regression and the method of least squares, we found the best fit lines for varying current and varying voltage and used their slopes to calculate the e/m ratio from the two methods described above.
The first method gives us a slope of s=16.8179.
[math]\displaystyle{ \frac {e}{m} = \frac {2s^2 V}{(7.8*10^{-4})^2} }[/math]
This produces a ratio of 1.10571*1010 C/kg.
The second method gives a slope of s=1.111*10-5.
[math]\displaystyle{ \frac {e} {m} = \frac {2} {s(7.8*10^{-4} I)} }[/math]
This then gives a ratio of e/m =1.84*108 C/kg.
The currently accepted value is:
- [math]\displaystyle{ \frac{e}{m}=1.76\times10^{11}\frac{C}{kg}\,\! }[/math]
Conclusion
We ended up with a final value of (5.6 ± 0.4)*109 C/kg, this is two orders of magnitude away from the accepted value! SJK 03:53, 21 December 2010 (EST)

Does it make sense to average two values that are so different? In this case, one of the numbers is essentially zero, so all you're doing is dividing the higher number by 2. Clearly they represent different parent means. My guess is that you and Brian had a calculation error.
This implies there were some major sources of error in our experiment. Dr. Gold's Lab Manual indicates that greatest source of error is from the measurements of the radius. This is because the electron beam is enclosed in a round glass envelope which distorts the actual radius of the beam. Systematic error is also present because the electrons don't all achieve their theoretical velocity since the current isn't applied uniformly and since some electrons have to collide with the gas to make the current path visible. In order to get better data, I would do this lab again and try to find some geometric formulas to correct for the curvature of the glass as in the Electron Diffraction Experiment.