Kubke Lab:Research/CND/Records2010-2011Summer/MH007
| Cranial Nerve Development | Experiment |
Embryo details

Species: Gallus gallus domesticus
Embryo Name: MH007
Embryo stage: ST25 Confirmed by Fabiana (supervisor).
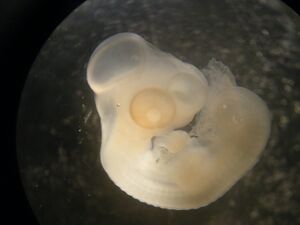
Staging description:
Fixation: PFA
Cryoprotection: None
Material label and storage: Slides are contained within the slide folder labeled MH007 in the lab.
Experiment details
Objective:To perform serial Nissl sections of the embryo for histological analysis.
Procedure:
- (Note: Please check whether these are notes for MH008 or MH007 --MF Kubke 18:37, 7 February 2011 (EST))
- (Note: MH007. I copied the text from MH008 to save time, then went through and made the appropriate changes.)
- The embryo MH007 was staged according to the Hamburger and Hamilton (1951) staging system.
- 10:00am Using dissection scissors and under a dissection microscope I cut the embryo just rostral to the wing bud, see Notebook entry.
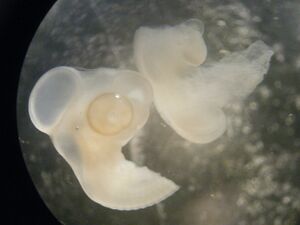
- The head region of the embryo was then gently replaced into a vial filled with PFA using forceps.
- A large metallic mould was half filled with OCT and then, using a plastic spoon, the embryo was very carefully laid on top of the OCT from the vial containing PFA.
- Using forceps the embryo was oriented such that the hindbrain ran parallel to the sides of the mould so that coronal sections could be made.
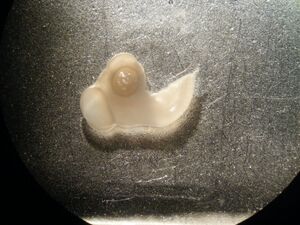
- Bubbles in the OCT were removed with the forceps.
- (Note: This was not satisfactorily achieved. I used an OCT bottle with very limited OCT left which caused lots of bubles to become assimilated into the block. This caused the tissue to scrunch during cutting.)
- 10:45am Incubated the mould at -19°C in the cryostat chamber.
- (Note: The larger mould took 45 minutes to completely harden, significantly longer than the smaller moulds which take about 15 minutes.)

- 11:30pm The block was oriented 90° to a chuck and stuck on by freezing OCT between the two contacting surfaces.
- OCT was slowly built up either side of the block inside the cryostat chamber.
- (Note: The OCT had to be added very slowly to prevent the warm OCT from melting the frozen OCT)
- The block had to be trimmed by hand with a razor blade so that the block could fit between the metallic chuck holder and the knife.
- (Note: This was not necessary with the smaller moulds.)

- The chuck was inserted into the metallic chuck holder and incubated at -19°C for 30 minutes.
- 1:00pm Began trimming the block using the cryostat.

- Serial sections were then cut and mounted onto gelatin-subbed glass microscope slides.
- The object temperature was changed to -20°C and the chamber temperature was set to -22°C in an attempt to counteract the effects of the hot and humid envionment.
- (Note: This was not successful and the temperature inside the chamber still was dropping to -16°C during cutting. If I cut slowly and took my time between cuts I could lower the temperature to -18°C but was not able to achieve the desired -19°C.)
- Bubbles were removed from the block and re-filled with OCT before being allowed to harden for 20 minute prior to resuming cutting.
- The sections were placed in a staining rack and allowed to dry overnight in Satya's Histology Lab's fume hood.
- The sections were then stained using Cresyl Violet solution. See [[Kubke Lab:/Notebook/Cranial nerve development/2011/02/04
|Notebook entry]]. Only the staining and differentiation step was used.
- The stained sections were then coverslipped with DPX mountant and allowed to dry on the benchtop.
Comments:There were two problematic factors I could identify during the cutting of MH007.
- The first is that I used the end of a bottle of OCT. Squeezing the OCT out produced a lot of bubbles which I found difficult to remove once it had hardened. Bubble were incorporated into the block whilst I was building up the block in preparation for cutting whilst a small few were incorporated during the initial formaiton of the block although the majority of those bubbles could be removed with forceps. There were between 10-20 sections lost following the mounting of the first slide. The bubbles were changing the way the sections were being fed under the glass cover, causing crinkling and rotation due to uneven resistance against the knife caused by the bubbles. I had to cut out one of the large bubbles and refill the hole with OCT and wait for it to harden.
- Secondly Satya's Histology Lab was immensely hot and humid. For some reason it was far hotter than even the hallway next to it. This could be due to the many freezers and machines surrounding the lab. The chamber was warming up considerably and often reaching -16°C. At -17°C The sections began to stick to the glass cover which made it very difficult to mount. For about 50% of the sections I had to mount the section onto my slide form the glass cover. My technique for cutting became aimed at attempting to intentionally have a small region of the OCT stick to the glass which would flip upside down when I lifted the glass cover up allowing me to mount the section of the glass. The humidity caused a lot of moisture build up which was adding to the crinkling the OCT and adhesion to the glass cover. It also meant that I had to remove the misture build up with the paintbrush following each mounting which added to the time taken to complete the experiment.
Cryostat Sectioning Cryostat settings (for a more detailed protocol visit Kubke_Lab:Cryomicrotomy)
| Cryostat | Leica – CM3050S |
| Knife | MX35 Premier +, 34 degrees, 80mm Thermo Scientific |
| Day Cut | 3rd Febuary 2011 see Notebook entry. |
| Knife Angle | 1.5° |
| Chamber Temp | -19°C Initially, then re-set to -22°C |
| Object Temp | -19°C Initially, then re-set to -20 |
| Glass Slides | Gelatin-subbed Original Menzel-Glaser microscope slides with cut edges and frosted Ends. Slides were subbed using the Cold gelatin subbing protocol including the pre-wash procedure, see Notebook entry 24/1/11. |
| Plane of section | Coronal |
| Number of slides | 17 (Slides 1-4,6-18). 156 Sections. |
| Observations | *Slide 1 was problematic. There was lots of damage to the sections during cutting and mounting, the OCT was rotating, crinkling and folding mostly due to air bubbles in the block.
|
(Include in your observations, eg ,were the sections serial, was any section lost, was quality assessed, etc)
Cresyl Violet staining
For more informattion see Kubke_Lab:Nissl_Stain_Protocol
| Date | 4th Febuary See notebook entry | |||||||||||||||||||||||||||||||||||||||
| Defatting and rehydration step | ||||||||||||||||||||||||||||||||||||||||
| This step was ommitted. | ||||||||||||||||||||||||||||||||||||||||
| Staining and differentiation step | ||||||||||||||||||||||||||||||||||||||||
| Solution | Time | Comments | ||||||||||||||||||||||||||||||||||||||
| Water | 5 sec | |||||||||||||||||||||||||||||||||||||||
| Cresyl Violet | 8 mins | |||||||||||||||||||||||||||||||||||||||
| 50% alcohol | 2mins | |||||||||||||||||||||||||||||||||||||||
| 70% alcohol acetic acid | 2mins | |||||||||||||||||||||||||||||||||||||||
| 95% alcohol | 2mins | |||||||||||||||||||||||||||||||||||||||
| 100% alcohol 1 | 2mins | |||||||||||||||||||||||||||||||||||||||
| 100% alcohol 2 | 2mins | |||||||||||||||||||||||||||||||||||||||
| Xylene 1 | 5mins | |||||||||||||||||||||||||||||||||||||||
| Xylene 2 | 5mins | |||||||||||||||||||||||||||||||||||||||
| Xylene 3 | 5-45minutes |
| ||||||||||||||||||||||||||||||||||||||
| Coverslip | Ind. | |||||||||||||||||||||||||||||||||||||||
Results
The quality of the tissue sections will be assessed by Malisha and Reuben.