IGEM:IMPERIAL/2007/CFS/Design
Cell-Free Systems
Design
To get around the problem of having bacteria in contact with food or medical devices in the applications of Cell by Date and Infector Detector respectively, we aimed to express reporter DNA constructs within a CFS consisting of cell extract encapsulated in selectively permeable vesicles. Our plan was to first prepare the cell extract and perform in-vitro expression in bulk solution. In parallel, we would investigate and optimize conditions for phospholipid vesicle formation. The final stage of our project would involve combining the two, so that protein expression is achieved inside the vesicles.
In-Vitro Expression
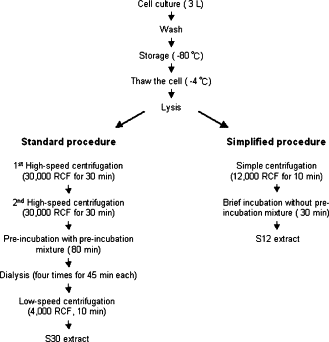
Since we are using E. coli plasmid DNA constructs, we chose to use the compatible E. coli cell extract for in-vitro expression. We tried out four options so as to find out the system most suitable for our two applications.
|
|
| ||
 |
  | ||
Vesicle Formation
For the formation of phospholipid bilayer vesicles, we chose to use the mineral oil method. The protocols are based on Engineering Asymmetric Vesicles by Sophie Pautot, BJ Frisken, and DA Weitz. (2003) and Toward an artificial cell based on gene expression in vesicles by Vincent Noireaux et al. (2005).
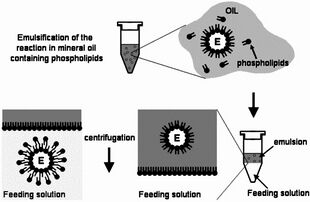
Summary of the mineral oil method
The mineral oil method consists of three stages, regardless of composition or equipment:
- Preparation of lipid-oil suspension
- Emulsification of vesicle contents
- Bilayer formation through sedimentation