Cro
Background
The lambda phage’s Cro is a 66 amino acid long protein that exists primarily as dimmers in the cell. Cro and lambda repressor cI can both potentially bind to three operator sites, OR1, OR2, and OR3.
Potential Binding Combinations
When cI repressor binds to OR2 [only], RNA polymerase bind to PRM and prevents its binding to the PR region. leading to transcription of cI tenfold (Positive Control) and turning off the cro gene (Negative Control).
When cI repressor binds to OR1, RNA polymerase favors neither PRM nor PR. Polymerase access to PR is blocked, but the absence of repressor fails to attract polymerase to the PRM region, leading to a very low level of PRM.
A cI repressor bound to OR3 blocks PRM transcription, but polymerase can still easily reach PR.
Role in Lambda Lifecycle
In a lysogenic cell, the repressor is usually bound to OR1 and OR2. Cooperativity between adjacent repressor molecules raises the operators’ affinities for repressor. A normal [lysogenic] repressor concentration causes transcription of cI and prevents cro synthesis. As repressor concentration increases further, repressor binds to OR3 and turns off the cl gene, allowing a constant level of repressor despite changes in growth rate.
The switch to lysis is brought on by stimuli [such as ultraviolet light] that damages the bacteria’s DNA. This activates a bacterial protein (RecA) which cleaves the cI repressor molecules. As the concentration of cI drops, OR1 and OR2 become unoccupied. cI transcription ceases, and the RNA polymerase binds to PR to transcribe cro. Cro binds independently to OR1, OR2, and OR3, preventing PRM transcription. The genes to the right of cro are also transcribed, which leads to the lytic phase.
As parts of a bistable switch, Cro and cI repressor protein levels in the cell directs the choice between lysis and lysogeny :
- When no cI is present, the cro gene produces Cro (leading to lysis).
- With higher cI repressor levels in the cell, only the cI gene is active, resulting in lysogeny.
- At very high cI concentrations, both cI and cro transcription is repressed.
Original Goal
First, we tried to verify the lytic activity of the Cro protein. The cro gene was placed behind a tetracycline-inducible promoter. By varying the amounts of anhydrotetracycline (aTc) in the media, we can produce a range of Cro concentrations in the bacteria. (Further explanation of the regulation involved in this construct mentioned here). Bacteria containing this construct were infected with phage carrying amber mutation in cro, so the phage could only be lytic if the construct produced a sufficient level of Cro in the host cell. By measuring percent lysis at each concentration, we could determine how much aTc was required to complement the mutant phage. Comparing these values to a similar tet-inducible YFP, we could choose an appropriate riboregulator to control Cro production in the final engineered phage.
Status and Future Plans
The cro construct plasmid (with the stronger constitutive promoter, J23100) was successfully cloned and entered into the registry (S03749). Titering was done with this construct in the D1210 bacterial strain.
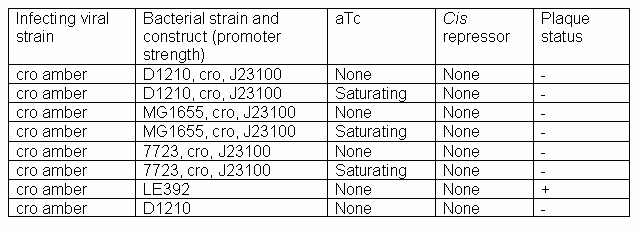
Titering results for cro Amber phage
Even in saturating levels of aTc, no plaques appeared. The construct was resequenced (and reverified), and transformed into non-suppressor strains MG1655 and 7723 to verify that the problem didn't lie with the D1210 strain itself. The results suggest that either a very high level of Cro protein (a level beyond the capacity of the cloned construct to produce) is required for the switch to lysis, or the timing of Cro needed by the virus is somehow affecting results. For now, cro is abandoned in favor of N and Q, which contain more promising results.
|