Glycerol
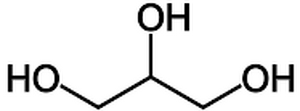
Glycerol is an alcohol often used in the lab to avoid freezing and increase the density of liquids. Its standardised IUPAC name is propane-1,2,3-triol. It is also often referred to as glycerine or glycerin.
Uses of glycerol
Density
Glycerol is used in many loading buffers to increase the density of the solution which enables good loading into already liquid filled wells of agarose and polyacrylamide gels. See agarose gel loading buffer and SDS sample buffer.
Freezing
Glycerol is also used to decrease the freezing point of protein solution which are easily degraded by repeated freeze/thaw cycles. A 50% (w/v) glycerol/water solution freezes at -25ºC and can thus be stored in a -20ºC freezer. Note that glycerol with 1.26g/cm3 is much denser than water and, therefore, a 50% (v/v) solution will be very different from a 50% (w/v) solution. For example, restriction enzymes are typically stored in a glycerol/water solution.
Glycerol solution recipes
- 50% glycerol solution recipe from the Silver lab
- 80% glycerol solution recipe from the Griffitts lab