Foams In Microfluidic Devices - Elijah Taylor
Introduction and Motivations
A Foam is a two phase colloidal system consisting of bubbles of gas suspended in a liquid or in a solid. Liquid foams (foams consisting of a liquid and gas phase) are commonly found in foods, hygiene products, or intermediary states in chemical reactions, and are notable for the increased surface-area to volume ratio between the gas and liquid phases and for their ability to expand or contract under pressure. Whipped cream, carbonated drinks, and yogurt are all types of liquid foams that use trapped gases as methods for creating textures and flavors, while some hand and dish soaps use the gas as a carrier to trap and remove dirt or grime. Other liquid foams, like the ones found in fire extinguishers, expand to many times their original volume in order to cover a large area with a foam contained in an easier to transport vessel. Solid foams (foams consisting of a solid and gas phase) lack the capacity for volume expansion to the same degree as liquid foams, but retain the large surface-area to volume ratio between the two phases. Solid foams, like sponges or yoga mats, are often very flexible, the trapped gases adding a component of malleability to the solid they are containing within, though rigid foams are also common. These rigid foams are typically used as insulation with the gas pockets acting as added resistance within the solid to inhibit heat transfer. Some foams, such as wall insulation and sealants, exhibit properties of both liquid and solid foams. They are stored as liquid foams to conserve space before they are sprayed onto a surface where they rapidly expand and harden into solid foams. The many applications of foams have promoted interest in creating economically favorable commercial and industrial materials, which require cheap and accurate methods to test the mechanical and chemical properties of these new foams before they are put into production.
Microfluidics allows for rapid testing of various types of foams at a low material cost, as the smaller scale requires less material to be produced for analysis. The small scale also prevents large or costly accidents, with mistakes being very localized and easier to contain. The cheaper costs and greater accuracy of microfluidic devices has generated interest in the use of such platforms for the study of foams in fields such as food or materials science.[1] The large surface area to volume ratio that is present in many foams also allows for higher reaction rates in systems with a low degree of mixing.[2] This helps alleviate the common difficulty of a low degree of mixing present on microfluidic devices due to their scale promoting laminar flow. The gas-liquid interface of liquid foams allows for a greater number of boundary reactions per volume than flowing the gas and liquid phases side-by-side, which provides an additional use for microfluidic foams as agents in the study of gas-liquid reactions on the microscale.
Micro-Bubble Creation Techniques
While macro-scale foams are typically created by injecting a large volume of gas into the liquid or solid medium and allowing the characteristic bubbles to be generated in bulk, micro-scale foams must be generated on a bubble-by-bubble basis.[3] Additionally, solid-gas foams are difficult to generate on microfluidic devices due to the inability for the solid to flow through the chip. Solid-gas foams are instead generated by flowing the solid phase as a liquid, generating the foam, and then solidifying the foam at a later point. There are several methods for gas bubble generation, each varying based on the desired properties (i.e.: bubble size or density of gas pockets) and flow rates of the foam. These methods are similar if not identical to many methods for droplet formation on microfluidic devices.
T-Junction
A T-junction is one of the most commonly applied methods for bubble and droplet formation in microfluidics due to its ease of use and simple geometry. The dispersed (gas) phase is pumped in orthogonal to the flowing continuous (liquid) phase and the main direction of flow of the foam. The continuous phase is commonly flowed at a higher rate than the dispersed phase. Bubbles are generated as shear forces from the continuous phase drag a segment of the dispersed phase into the flow stream, generating a spherical or obloid bubble.[3] The size and shape of the bubble are controlled by the flow rates of the dispersed and continuous phases as well as the geometry of the microfluidic channel.[4][5] Variations of the T-junction, such as the Y-junction and cross-junction, also rely on using sheer forces of intersecting streams for bubble generation.
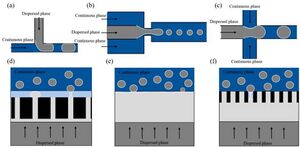
Flow Focusing and Co-Flow
Flow focusing models also employ shear forces to generate bubbles, as the T-junction model does, however the dispersed phase is flowed parallel to the flow direction of the foam while the continuous phase is pumped in orthogonal to the dispersed phase. Bubbles are "pinched off" the main dispersed phase stream by the continuous phase and pushed in the direction of flow to generate the foam.[5]
Co-flow models are a form of flow focusing where the continuous phase is flowed parallel to the dispersed phase before both phases are forced through a narrow channel, creating a volume gradient in the dispersed phase that causes a spontaneous generation of bubbles.[4]
Edge-Based Droplet Generation
Edge-Based Droplet Generation (EDGE) is a method of micro-bubble generation that uses principles of macro-scale foam generation to create bubbles at a faster rate than is often achieved in flow focusing or T-junction models. Unlike previously discussed methods, EDGE does not rely on sheer forces to generate bubbles. Instead, the dispersed and continuous phases are flowed parallel to one another, separated by a raised, shallow plateau. The dispersed phase is pumped at a higher pressure than the continuous phase and flows over the plateau until it contacts the continuous phase in a similar orientation to a T-junction. At significant enough pressures (varying based on the chemical properties of the phases) the dispersed phase will enter the continuous phase and spontaneously generate bubbles.[6] This method often generates bubbles of random size, akin to many common macro-scale foams, however should a specific size of bubble be desired, partitions are set up along the dispersed-continuous phase interface to control bubble formation.[4]
Foam Stability
Stability is a major concern when considering the production of foams. Most commonly occurring in liquid-gas foams, destabilization occurs naturally over time as the phases begin to separate and the system moves toward an equilibrium state where there are no longer gas particles dispersed in the liquid. Microfluidics allows for small scale testing of the stability of various types of foams based on the properties of the foam, such as surface tension, bubble size, and density of bubbles in the fluid. Microfluidics also allows for the study of foam stabilizing techniques to improve the longevity of the foam while conserving resources and costs before being applied to large scale production.
Principles of Destabilization
Foam destabilization primarily occurs through three physical processes in liquid-gas systems.
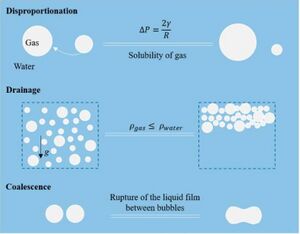
Disproportionation
Disproportionation is the predominant method for destabilization in most liquid-gas foams. Resulting from pressure differences in the bubbles generated by variations in size, gas from smaller bubbles diffuses through the liquid phase. The pressure difference in the bubbles is governed by the Laplace Pressure: [math]\displaystyle{ ΔP = \frac{2γ}{R} }[/math]
From this, it can be seen that for a bubble of smaller radius R, the pressure would be greater than a bubble of larger radius R. Because of this, the microfluidic methods that generate more mono-disperse bubbles result in more stable foams.[4]
Drainage
Drainage is a method of foam destabilization driven by gravity. The gas bubbles in the liquid have a lower density (ρ) than the liquid, resulting in a gradual rise of the bubbles to the surface of the liquid.[4] As the bubbles breach the surface of the liquid, the gas escapes into the atmosphere for an open system or creates a new gas phase above the liquid in a closed system, such as on a microfluidic chip.
Coalescence
During coalescence, two bubbles come into contact and merge to form a single larger bubble in the liquid. This process repeats as more bubbles coalesce, until a single large bubble is present in the fluid and the gas and liquid phases are fully distinct. Similar to drainage, the single large bubble effectively removes the presence of gasses in the liquid phase and without the intermixed phases the foam loses its colloidal properties. This presents a major problem, as the material no longer retains the same characteristics of the initial foam and is unable to be properly used or studied. As such, coalescence reduction is of major interest during testing of microfluidic devices, as although most liquid foams will eventually destabilize, decreasing coalescence can greatly increase the longevity of the foam.[3]
Surfactants
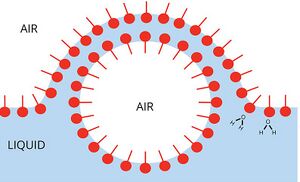
The most commonly applied method for increasing foam stability is by application of surfactants or similar surface-active particles. Surfactants are molecules that consist of a hydrophilic head attached to a hydrophobic tail and are dispersed throughout the liquid phase of the foam before the creation of the gas bubbles. Surfactants lower the initial energy required to produce a bubble in the liquid phase by reducing the surface tension between the gas and liquid.[7] As a bubble forms, the surfactant compound aligns itself along the gas-liquid boundary, the head of the surfactant contacting the liquid while the tails extend into the gas bubble. Surfactants have the additional benefit of inhibiting bubble coalescence, as two bubbles in contact with surfactants on their surfaces must overcome the additional surfactant layer before merging.[4][7]
References
[1 ]P. Marmottant and J. Raven, Microfluidics with foams. Soft Matter, 2009, 5, 3385
[2] K. Prabhakaran, S. Vijayan, and P. Wilson, Alumina foam microspheres by emulsion drop-casting in aqueous ammonium chloride solution. Ceramics International, 2018, 44,12547-12554,
[3] K. Muijlwijk, Microfluidic methods to study emulsion formation. Wageningen University, 2017, Disseration
[4] B. Deng, J. de Ruiter, K. Schroën, Applications of Microfluidics in the Production and Analysis of Food Foams. Foods, 2019, 8(10), 476
[5] A. Huerre, V. Miralles and M. Jullien, Bubbles and foams in microfluidics. Soft Matter, 2014, 10, 6888
[6] K. van Dijke, G. Veldhuis, K. Schroën and R. Boom, Parallelized edge-based droplet generation (EDGE) devices. Lab Chip, 2009, 9, 2824
[7] J. Baret, Surfactants in droplet-based microfluidics. Lab Chip, 2012, 12, 422
