Dedicated systems/Dedicated translation
Testing a new RBS-ribosome pair
I'm going to construct a new mutant 16srRNA. I'm going to try two of the sequences published by Chin and Rackham[1]. I'm picking two of the mutants that don't require me to mutate the distant bases that the authors also mutate.
Orthogonal ribosome construction
I'm going to make this via 'Round-the-horn site-directed mutagenesis. This seems like the quickest way to make the full 5 base mutation in one go.
Primer design
pCH1497-ASD1 5'-CacaCTTAccttaaagaagcgtactttgtagtgctcacacag attgtctgatagaaagtga-3'
4rRNA-f primer 5'-TTGTGGTAccttaaagaagcgtactttgtagtgctcacacag-3' Tm = 61.8C
10rRNA-f primer 5'-TGGGATTAccttaaagaagcgtactttgtagtgctcacacag-3' Tm = 61.9C
4/10rRNA-r primer 5'-TGATCCAACCGCAGGTTCCCCTAC-3' Tm = 62.8C
Experimental details
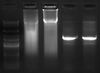
Lane 2 - 4rRNA, phusion
Lane 3 - 10rRNA, phusion
Lane 4 - 4rRNA, pfu ultra
Lane 5 - 10rRNA, pfu ultra
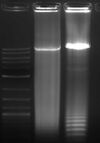
Lane1 - 10kb ladder
Lane 2 - 4rRNA, phusion
Lane 3 - 10rRNA, phusion

Lane 1 - 10kb ladder
Lanes 2-7 - 6 colonies of pCH1497->4rRNA
Lane 8 - positive control PCR on pCH1497
Lane 9 - positive control PCR on I7101
Lane 10 - 10kb ladder
Lane 11-16 - 6 colonies of pCH1497->10rRNA
Lane 17 - negative control, no template

- My plasmid is 10kB so Sean Moore suggested adding fresh polymerase in half-way through. I didn't actually manage to do that on the first attempt.
- Sean also suggested dropping the melting temperature by a few degrees and also the extension temperature. I did 91C for melting and 69C for extension.
- I used Pfu Ultra and Phusion as my polymerases.
- According to the gel I ran of the PCR product (I ran 10μl), phusion worked(?) but pfu ultra did not. Not sure yet why the phusion lanes are smeary. I think I'll redo this and try to add in the fresh polymerase this time.
- Not sure why the PCR product of 10rRNA looks slightly longer than 4rRNA. Maybe just because there is more DNA?
- I did a 10μl ligation rather than the 5 that Sean recommends.
- Got ~50 colonies on each plate of BL21(DE3) with a wide range of sizes(?). I PCR'd 3 big and 3 small on each plate. Only got the right band from the small colonies, not sure why.
- Will start cultures of 4-6 and 10-4, 10-5 and 10-6.
- Sequencing says that 4-6 worked, although it mutated an A to a G just upstream of the ASD. I'll test this anyway to see if it works. I should also check to see if this mutation is due to an error in my reverse primer as it would have been in this region.
- I got no sequence results for 10-6 and 10-5 seems to have had the ASD cut out. I'll try re-prepping 10-6 and see how that sequences.
- From the second gel, start cultures of lanes 3, 6, 11 and 12.
- Colonies 10-1 and 4-2 sequenced correctly. nice.
- In addition, I'm keeping 10-5 from the first sequencing since it has the ASD deleted and 4-5 from the second sequencing as it has a slightly different ASD than expected.
Reporter Devices
I designed six new reporter devices for dedicated translation and six new reporter devices for dedicated transcription and translation. I checked for -
- random start codons
- BB restriction sites
- cryptic SD sequences
- secondary structure
More later.