Biomod/2013/Aarhus/Results And Discussion/System In Action
<html> <style> /* ul.menu li.</html>Results_And_Discussion/System_In_Action<html> a {
color: cyan;
}
- /
- toc {
display: none; }
- mytoc {
background: none; width: 200px; }
.toc { border: 0px solid; }
- toc ul ul,.toc ul ul {
margin: 0 0 0 1em; }
table.toc { background-color: #f0f4f4; }
- mytoc a,#mytoc a:visited {
font-size: normal; color: #222222; }
- mytoc a:hover {
font-color: #009ee0; /* text-decoration: underline; */ }
- wiki-toc {
width: 200px; margin-top: 6px; float: left; }
- wiki-body {
margin-left: 200px; padding-left: 12px; padding-right: 35px; }
- toc #toctitle,.toc #toctitle,#toc .toctitle,.toc .toctitle {
text-align: left; }
- toc h2,.toc h2 {
font-weight: normal; font-size: 17px; }
/*
- wiki-contents A {
color: #00aeef; }
- wiki-contents A:HOVER {
color: #00aeef; }
- /
- toctitle span {
display: none; }
/*
- wiki-body p,#wiki-body li,#wiki-body dd,div.thumbcaption {
font-size: medium; }
- /
/* required to avoid jumping */
- tocScrolWrapper {
/* left: 450px; */ position: absolute; /* margin-left: 35px; width: 280px; */ }
- tocScrol {
position: absolute; top: 0; /* just used to show how to include the margin in the effect */ /*margin-top: 20px; */ /* border-top: 1px solid purple; */ /*padding-top: 19px;*/ }
- tocScrol.fixed {
position: fixed; top: 0; }
- editPageTxt {
text-align: left; padding-left: 15px; }
- editPageTxt P {
clear: both; }
- toPageTop {
float: left; position: relative; top: 18px; left: 13px; color: #d13f31; } </style>
<script type="text/javascript"> $(document).ready(function() { var parentTable = $("#toc").parent(); $('#mytoc').append($("#toc").first());
$('#mytoc').find("#toc").attr("id", ""); parentTable.closest('table').remove(); });
$(document).ready( function() { var top = $('#tocScrol').offset().top - parseFloat($('#tocScrol').css('marginTop').replace( /auto/, 0)); var nav = $('#tocScrol'); var max = $('#indexing').offset().top - nav.height();
$(window).scroll(function(event) { // what the y position of the scroll is var y = $(this).scrollTop();
if (y > top) { // && signs are html decoded thus this construction if (y >= max) { nav.removeClass('fixed'); nav.css({ position : 'absolute', top : max - top }); } else { nav.addClass('fixed'); nav.removeAttr('style'); } } else { nav.removeClass('fixed'); nav.removeAttr('style'); } }); }); </script> <html> <html>
<a id="toPageTop" href="#">▲</a>
[<a href="http://openwetware.org/index.php?title=Biomod/2013/Aarhus/</html>Results_And_Discussion/System_In_Action<html>&action=edit">edit this page</a>]
</html>
System in action
Test of photosensitizers and cholesterols
Hybridization of the chemically modified DNA strands
In order to test the ability of the photosensitizers to induce apoptosis in cells, a test system was designed. This system consisted of two complementary DNA strands, of which one was modified with the photosensitizer, In(PPa)Cl, and the other with cholesterol. In order to achive the highest yield of annealing of the modified DNA strands, the annealing of three different designs was tested. The chosen design was design DII (see structure on Figure 42). Gel analysis of this test system showed that the two modified DNA strands had annealed well (see Figure 43 below).
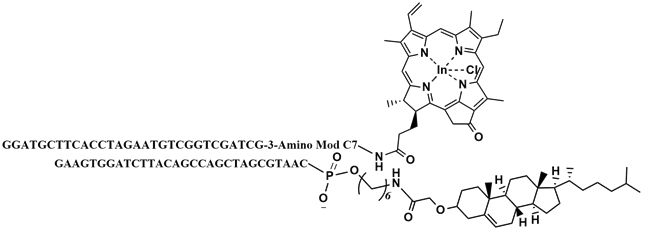
The gel was scanned at 670 nm, where the photosensitizer absorb light, and subsequently stained for the nucleic acids with ethidium bromide and scanned again.
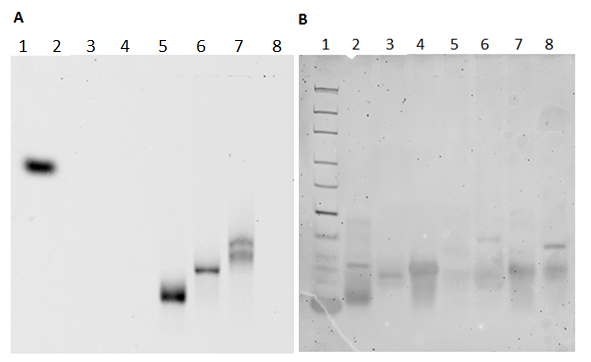
On the gel in Figure 43, the single stranded DNA modified with In(PPa)Cl (lane 5) gives rise to a band below the band for the two modified DNA strands together (lanes 6 and 7). This band shift is due to the annealing of the two strands. The gel, both stained and unstained, confirms that the modified DNA strands of DII anneal. On this gel it is also possible to see the two unmodified DNA strands (lane 8), which also anneal, although the annealing is not complete.
Cell experiments with DII
As the gel in Figure 43 showed a sufficient annealing of the two strands in DII, the system could consequently be tested on cells, to see if the cholesterols were able to bring the photosensitizers close enough to the cells to mediate photoinduced cellular death. First, the localization of the cholesterols to the membrane was assayed. HeLa cells were incubated with DII in a concentration of 5µM for 30 min.
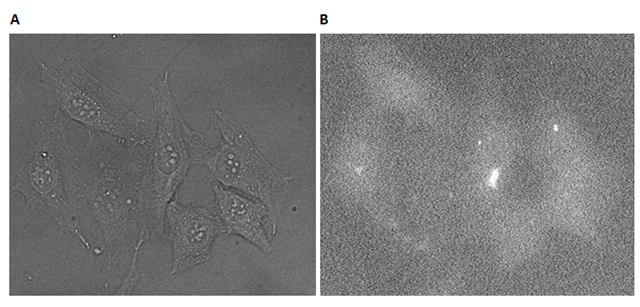
As seen from Figure 44A, no bubble formation or flattening of the cells was observed, indicating that the cells were still viable after the treatment. This indicates that no dark toxicity was induced with the DII system. In Figure 44B, a very weak fluorescence is observed. The cellular debris is seen as a bright spot at the center of the image. In order to obtain a higher degree of cellular interactions, the experiment was repeated with DII in a 5 µM concentration for a period of 2 hours. An increase in cellular interactions would also yield a higher fluorescence signal, that might help locating the position of the DII testsystem on the cell. The cells were again visualized with brightfield microscopy and fluorescence microscopy (Figure 45).
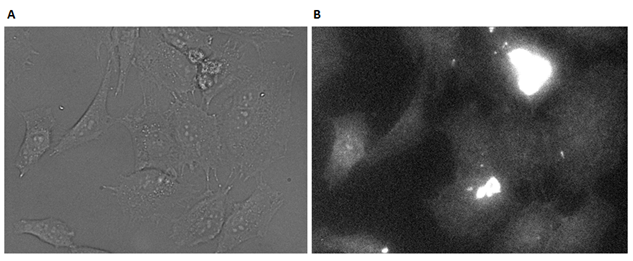
The cells were examined after 2 hours of incubation with DII (Figure 45A), and appeared healthy. In order to locate the DII system, the flourescence was imaged (Figure 45B). All parts of the cells seems stained, and therefore it was not possible to conclude anything about the localization of DII. The flourescens is nonetheless better than after 30 minutes incubation, thus higher incubation time has lead to higher cell interaction.
The DII system was then tested for the ability the photoinduce death in cells. Cells were incubated with 5 µM DII, and subsequently irradiated at 670 nm for 15 minutes.
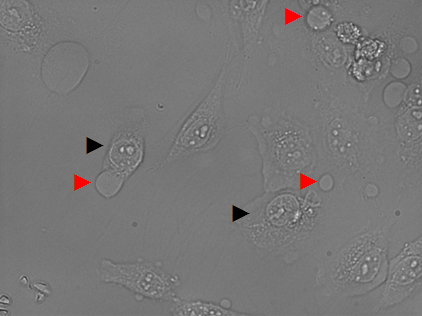
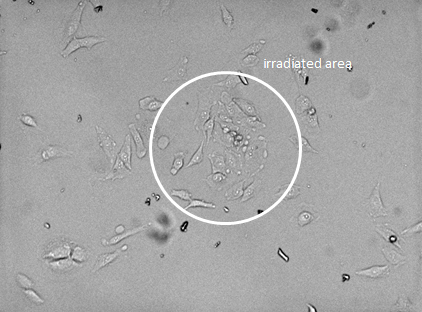
After irradiation, the formation of vacuoles (seen as bubbles, and marked as red arrows) was observed, as well as flattened cell bodies and pronounced nuclei (black arrows). As the image in Figure 46, show clear signs of cell death, it can be concluded that the photosensitizer was able to emit singlet oxygen upon light exposure, and induce cell death. The drug delivery system designed in this project and also the designed test system DII was aiming for the tight controlled self-induced apoptosis pathway for cell death. In many cases photodynamic therapy (PDT) is highly efficient in inducing apoptosis, though high doses have been shown to result in the target cells becoming necrotic, while the surrounding tissue become apoptotic. [1] Thus DII might still induce apoptosis, but the applied dose is too high, and the cells have become necrotic. The brightfield image in Figure 47 shows that only cells in the irradiated area are affected, and thereby demonstrate the selective cytotoxicity of the system.
Since DII showed a very weak fluorescence signals, a new design, DII*, was made. In this system, In(PPa)Cl was replaced with PPa, which is known to be a very good flourophor. This system was then tested in cells, as described above.
Conjugation of 3’amine modified DNA strand with PPa-NHS ester (design DII*)
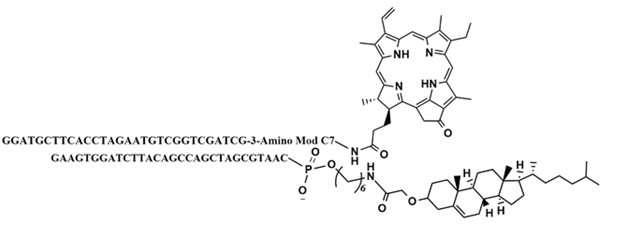
Design DII* was made using, the automated oligonucleotide synthesizer. The product was obtained as a black solid (12 nmol, 52 %, 239.6 μM). ε=287300 for the DNA strand (found on [1]) MALDI-TOF linear possitive mode (appendix): m/z calcd. for 5’- GGATGCTTCACCTAGAATGTCGGTCGATCG-3-Amino Mod C7-C33H33N4O2 [M+H]+: 9967.47 found 9962.36.
Cell experiments with DII*
In the experiments with DII, it was not possible to localize DII on the cells. To further test, if it is the cholesterol part of the construct that is inserted into the cell membrane, HELA cells were exposed to only PPa-DNA. This was done to verify, that the photosensitizer itself was note able to attach itself to the membrane, thereby indirectly establishing the role of the cholesterols in the attachment. The cells were incubated with a 1µM PPa-DNA for 30 min.
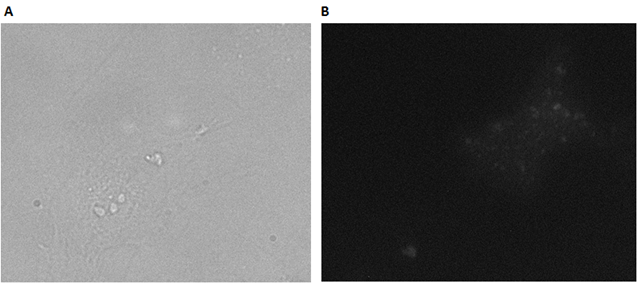
The cells in Figure 49A appear healthy, thereby indicating that the PPa-DNA strands alone have no dark toxicity. Subsequently, the cells were irradiated to examine the flourescence and the cellular interactions. From Figure 49B it can be seen that the cells are only weakly fluorescent. This indicates that the cellular interactions of the PPa-DNA strand alone is very low. This could be explained by the lack of ability of PPa to bind to the cells without the cholesterols. Figure 50 shows the comparison between untreated cells and irradiated cells, incubated with 1µM PPa-DNA for 30 min.
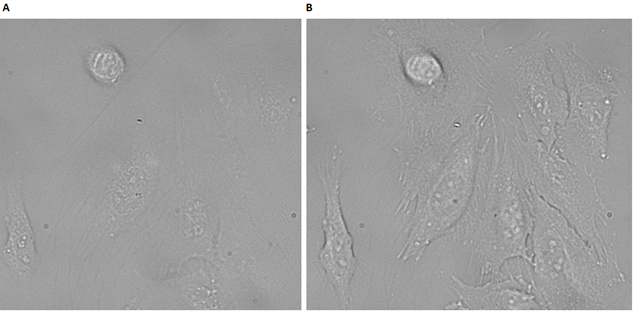
From Figure 50, it is further demonstrated that no cells had died upon irradiation with PPa-DNA alone. Thus PPa-DNA has no effect on the cells alone.
Test of PPa-DNA along with cholesterol
Cells were then incubated with a 1µM sample of PPa-DNA annealed with DNA-cholesterol strand (Figure 51).
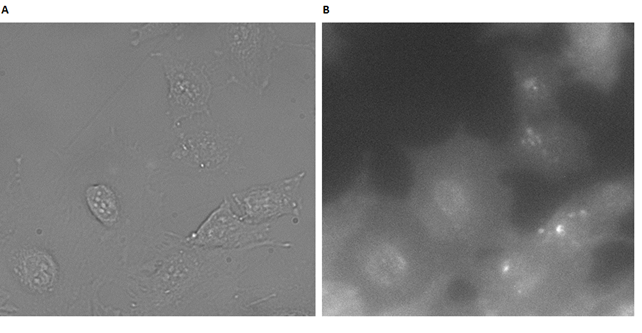
As seen from Figure 51A, the cells were healthy before the irradiation, demonstrating that DII* has no dark toxicity. After irradiation, an enhanced fluorescence signal is observed, compared to cells with PPa-DNA alone, (Figure 49B), showing a higher degree of cellular interactions. This establishes the role of the cholesterols in the construct, to mediate a contact with the cell membrane and enable the cytotoxic function of the photosensitizer. To test the combined ability of the PPa-cholesterol duplex to induce apoptosis, cells were again treated with a 1µM sample of PPa-DNA annealed with DNA-cholesterol strand.
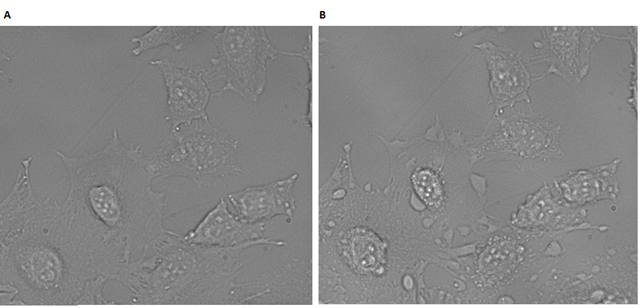
In Figure 52B a prominent vacuole formation is prominent, which indicated that the cells were necrotic. Due to the high dose, that leads to necrosis, it is not possible from these experiments to determine if this construct primarily causes apoptosis or necrosis.
Attachment of the chemical modifications to the plate
Attachment of photosensitizers to the origami plate
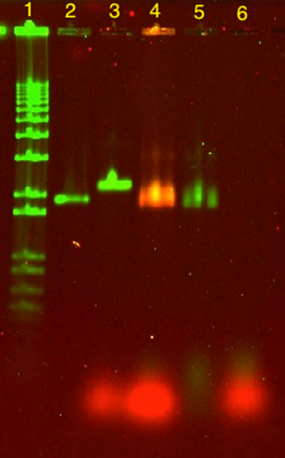
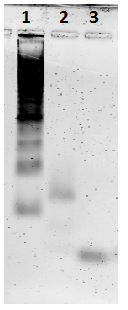
A plate was folded with photosensitizers linked to a staple module through an amide modification. In order to confirm its attachment, a denaturing gel was run. Three different origami samples were made. The first reaction had photosensitizer-modified staples mixed in, but was not processed with an annealing ramp (negative control). The second reaction had the photosensitizer module and was processed with a non-linear annealing ramp. The third reaction had ModLeft (staple strands without photosensitizers) and was also processed with a non-linear annealing ramp (positive control) In the gel in Figure 56, an orange band is seen in lane 4. This indicates that the photosensitizer had been successfully attached to the plate. In lane 3 and 5 green bands are visible which indicates that these plates do not have photosensitizers on them. The lower red bands which are staples with the photosensitizer module, indicate the same thing.
References
-
Oleinick, N. L. et al. The role of apoptosis in response to photodynamic therapy: what, where, why, and how. Photochem. Photobiol. Sci. 1, 1-21 (2002).[1]
</html>
<html> <head> <style>
- indexing {
/* float: left; position: center; */ background-color: #222; border-top: 2px solid #d13f31; color: #006e9c; margin: 0px; padding: 0px 0px 10px 0px; width: 100%; text-align: center; }
.footer-section { padding: 10px; display: table-cell; text-align: left; }
.footer-section-title { font-size: 20px; }
- footer-contents {
color: #006e9c; display: inline-table; }
.footer-section A { color: #006e9c; text-decoration: none; }
.footer-section A:HOVER { color: #00aeef; }
.footer-section ul { list-style-type: square; }
- sitemapTitle {
margin-top: 20px; font-size: 24px; }
- editFooter {
float: right; margin-top: -28px; margin-right: 5px; }
- editFooter A {
color: #006e9c; text-decoration: none; }
.cf:before,.cf:after { content: " "; /* 1 */ display: table; /* 2 */ }
.cf:after { clear: both; }
- bodyContent a[href^="mailto:"], .link-mailto {
background: url() no-repeat scroll right center transparent; padding-right: 0px; color: #006e9c;
}
</style> </head> <body>
SITEMAP | BIOMOD 2013 NANO CREATORS | Aarhus University
Copyright (C) 2013 | BIOMOD Team Nano Creators @ Aarhus University | Programming by: <a href="mailto:pvskaarup@gmail.com?Subject=BIOMOD 2013:">Peter Vium Skaarup</a>.
<img alt="Sigma - Aldrich" src="http://openwetware.org/images/3/39/Sigmaaldrich-logo%28transparant%29.png" width="300px" height="154px"> <img alt="VWR International" src="http://openwetware.org/images/2/28/Vwr_logo.png" width="300px" height="61px"> <img alt="Promega" src="http://openwetware.org/images/7/72/Promega.png" width="175px" height="105px" style="padding-right: 5px; padding-left: 5px;"> <img alt="kem-en-tec" src="http://openwetware.org/images/3/3a/Kementec.png" width="130px" height="129px"> <img alt="Centre For Dna Nanotechnology" src="http://openwetware.org/images/4/4f/CDNA_logo.png" width="420px" height="90px"> <img alt="Dansk Tennis Fond" src="http://openwetware.org/images/9/9a/Dansk_tennis.png" width="250px" height="53px">
</body> </html>