BME100 s2016:Group13 W1030AM L6
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
OUR COMPANY
LAB 6 WRITE-UPBayesian StatisticsOverview of the Original Diagnosis System In BME 100, 17 groups of 4-6 students per group each diagnosed two patients resulting in 34 patients total. The patients were screened for the SNP strand of DNA which was the gene that coded for non-Hodgkin Lymphoma. In order to screen the patients for the SNP gene disease marker the PCR machine replicated the disease forward and reverse DNA strand. After the experiment was completed in the PCR machine the PCR products from each test tube was added into its respected 500 micro-liter buffer solution. The image J portion of the lab consisted of a Calf thymus DNA solution of 0, 0.25, 0.5, 1, 2, and 5 micrograms per milliliter concentration. Also the eight test tubes of the PCR reaction which consisted of the positive, negative, three samples from patient one, and three samples form patient two. Eighty micro-liters of the DNA solution was pipetted onto a glass sheet, then 80 micro-liters of SYBR Green I was pipetted onto the bubble of PCR reaction in order to bind to the SNP genes and glow when a light is shined through the bubble forming a 160 micro-liter bubble. The phone camera was then placed 4cm away from the bubble and three pictures of each bubble formed with the pipette were taken in order to ensure a quality picture was taken, resulting in 42 photos total. After each pipetting the old tip was disposed of and a new tip was put on in order to prevent cross contamination. Also the glass plate was moved over in order to get a new gap in order to prevent cross contamination. This was important because both processes were used to prevent inaccurate results. However the main problem with this lab was with keeping the SYBR Green I in the dark when using it, which was an important task to do because SYBR Green I was less reactive the longer it was exposed to light. Based off the final results: .889 frequency for positive results, 0.875 frequency for negative results, 0.500 frequency for positive results correlated with the doctors diagnosis, and 0.889 frequency for negative results that correlated with the doctors diagnosis.
What Bayes Statistics Imply about This Diagnostic Approach
For Calculation one the question, what is the probability of a pos final test given a pos PCR? was analysed. P(A) means the probability of a pos final test, which resulted in a frequency of 0.380. P(B) means the probability of a positive PCR test, which resulted in 0.380. P(B/A) means the probability of a pos PCR test given a pos final test, which resulted in 0.830. P(A/B) means the probability of a pos final test resulting in a pos PCR test, which resulted in 0.830 based off the Bayesian equation. For Calculation two the question, what is the probability that a patient will get a negative final test conclusion, given a negative PCR Reaction? was analysed. P(A) means the probability of a negative final result, which resulted in a frequency of 0.560. P(B) means the probability of a negative PCR reaction, which resulted in 0.580. P(B/A) means the probability that a neg PCR reaction will result in a negative final result, which resulted in a frequency of 0.880. P(A/B) means the probability that a patient with a negative final result will result in a negative PCR reaction, which resulted in a frequency of 0.840 based off Bayes formula. For calculation three the question was, what is the probability that a patient develops the disease given a positive test? was analysed. The P(A) wants the frequency that people will develop the disease, which resulted in 0.28. The P(B) means the frequency of a positive test result, which resulted in 0.38. The P(B/A) means the probability given a positive test means that the patient will develop the disease. which resulted in 0.50. The P(A/B) means the probability that a positive test result will result in the person developing the disease, which resulted in 0.37 based off the Bayesian equation. For Calculation four the question was, what is the probability of a person with no disease given a negative final test? P(A) means the probability of no disease, which resulted in 0.720. P(B) means the probability of a negative test, which resulted in a 0.56 frequency. P(B/A) means the probability of a negative test given that the patient does not have the disease, which resulted in a 0.6950. P(A/B) means the diagnoses of disease given a negative test result, which resulted in 1.14 based off the Bayesian equation. The three main source of error in this experiment were from not placing the camera in the same spot for each droplet causing the pixels to be inconsistent. The PCR machine might of not been closed all the way, it was hard to tell since it was a screw on lid. If the machine is not closed correctly then it could affect the temperature causing an incomplete PCR reaction resulting in faulty data. The third source of error is mainly human error that occurred with pipetting, there was a high possibility of cross contamination due to the necessity for every solution to be kept separate. Intro to Computer-Aided DesignTinkerCAD
Our Design
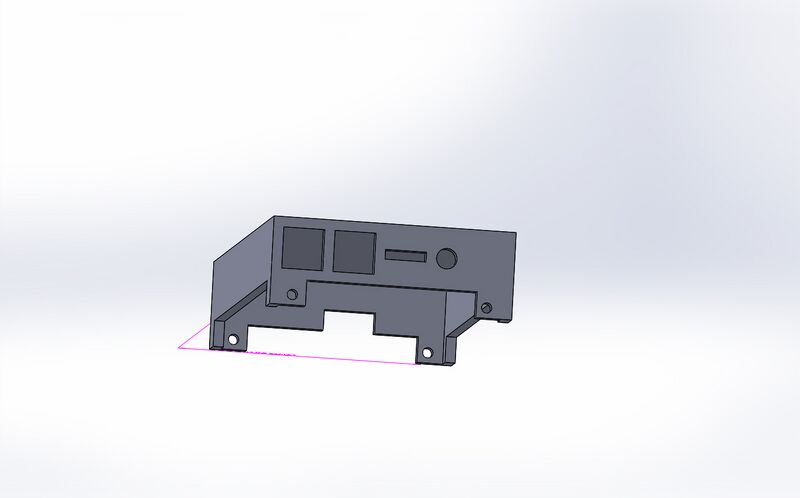
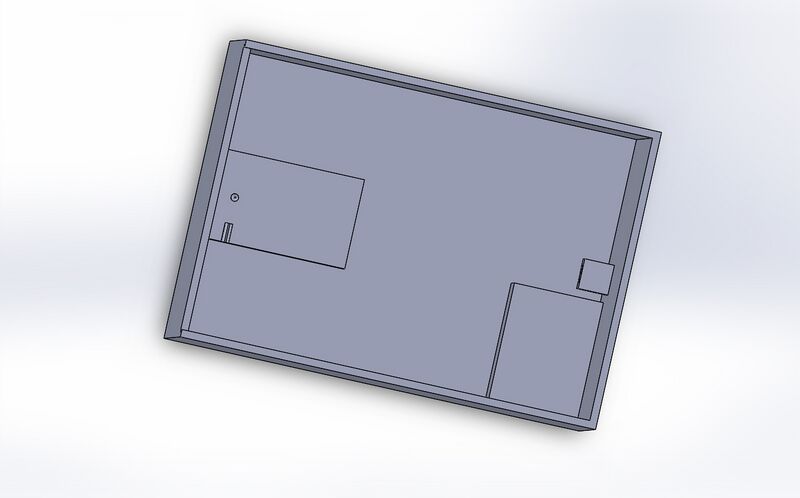
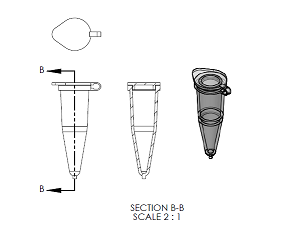
Fluorimeter Design
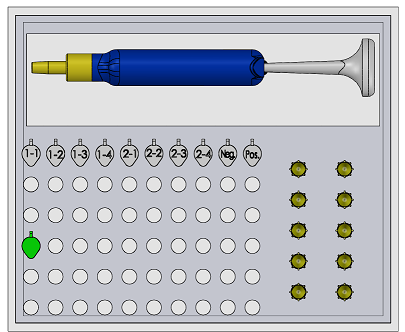
Feature 1: ConsumablesConsumables Kit Our consumables kit will include: A pipettor is included in the kit and will be able to use any standard size disposable tips. There are disposable tips included in our kit and can also be used with any standard size pipette. Our plastic tubes will be pre-labeled and will fit in any plastic tray due to it's universal design. They will open and close with ease for the convenience of saving time and avoiding accidents. Our reagents ( SYBR Green Solution, Buffer) will be prepackaged in all the right measurements all in one container so that mixing will be no problem resulting in a quicker process.
Feature 2: Hardware - PCR Machine & FluorimeterAdjustments to flourimiter: We added a low power fixed view camera that can be set to take pictures at certain intervals when triggered, the camera is made to save the pictures to an SD card inserted in a data port in the back of the unit. Additionally the unit is equipped with a slide lid to prevent any outside light sources from effecting the results.
| |||||||