BME100 s2015:Group8 12pmL6
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
OUR COMPANY
LAB 6 WRITE-UPBayesian StatisticsOverview of the Original Diagnosis System Samples of DNA can be amplified and later analyzed through the use of PCR (polymerase chain reaction) and some imaging software, respectively, to see whether or not the patients have the SNP associated with the disease. The students in class were divided into 34 teams with 6 students in each team and instructed to ascertain whether the two patients given to each team out of 68 patients were negative, positive, or inconclusive for the disease. Each team was only assigned 2 patients who each had 3 separate DNA samples which were tested separately in the effort to minimize error. Along with this, the software which was used to analyze the DNA drops, ImageJ, was amended in such a way that its measurements were the most accurate when viewing the color to minimize error. Due to errors in previous labs only data from 32 teams were used to calculate the overall class statistics. Averaging the three separate DNA tests for each patient out of 32 diagnoses, 14 had positive conclusions and 18 had negative conclusions.
. Calculation 1- The first calculation determines whether or not and to what degree the test is accurate at detecting a positive result for SNP. Using Bayesian statistics, this calculation determines the probability of a positive test conclusion given a positive PCR reaction for the data. The value that was calculated for this section was very close to 1.00 or 100% so it can be assumed that the test is fairly reliable. Calculation 2- This second calculation determines whether or not and to what degree the test is accurate at detecting a negative result for SNP. Using Bayesian statistics, this calculation determines the probability of a negative final test conclusion given a negative PCR reaction. The value was once again close to 1.00/ 100% so the test was fairly reliable. Calculation 3- This third calculation determines how effective the PCR was at predicting whether or not the patient would actually develop the disease in question. The Bayesian statistics determined the probability of the patient having yes for the disease given a positive test. The value was a little over half way to 1.00 so it wasn’t as reliable as the first two calculations. Calculation 4- This last calculation determines how effective the PCR was at predicting whether or not the patient would not develop the disease. The Bayesian statistics for this section determined the probability of the patient not having the disease given a negative test result. The value was about 3/4ths close to 1.00 so it was pretty reliable as well. There were a variety of errors, both human and device, that occurred for each group in the class, giving rise to some slight error in the Bayesian values. Examples of device errors include every group having the machine run a different amount of cycles due to their procedure, and machines in general not working properly. Some human forms of error include some of the tubes getting cracked, mislabeling of tubes, and inaccurate disposal of pipets which could lead to cross contamination. Computer-Aided DesignTinkerCAD Our Design
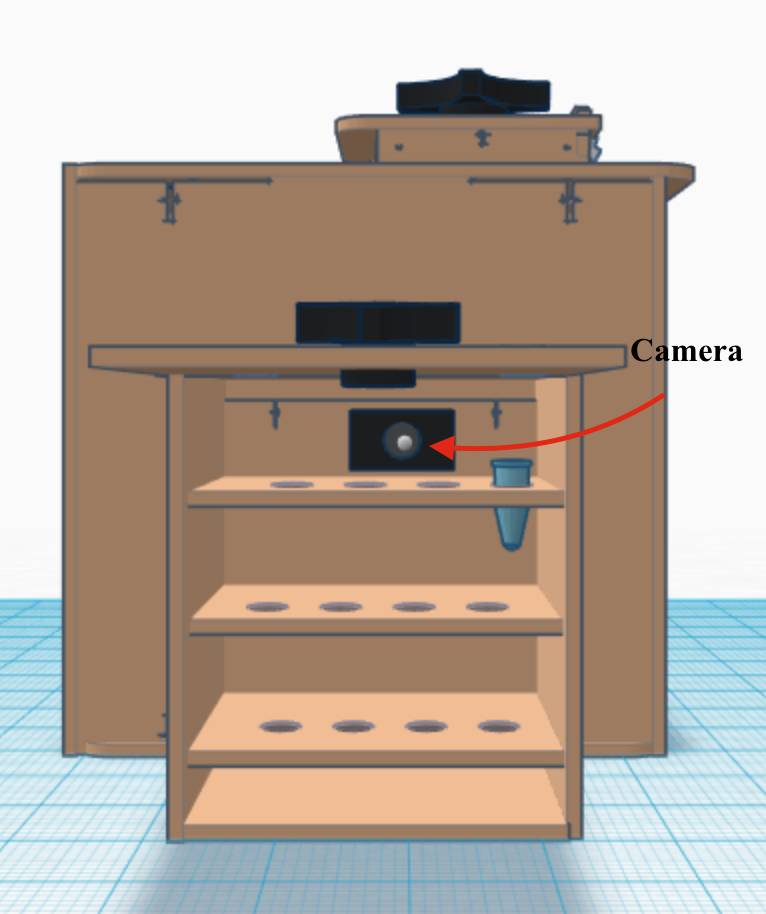
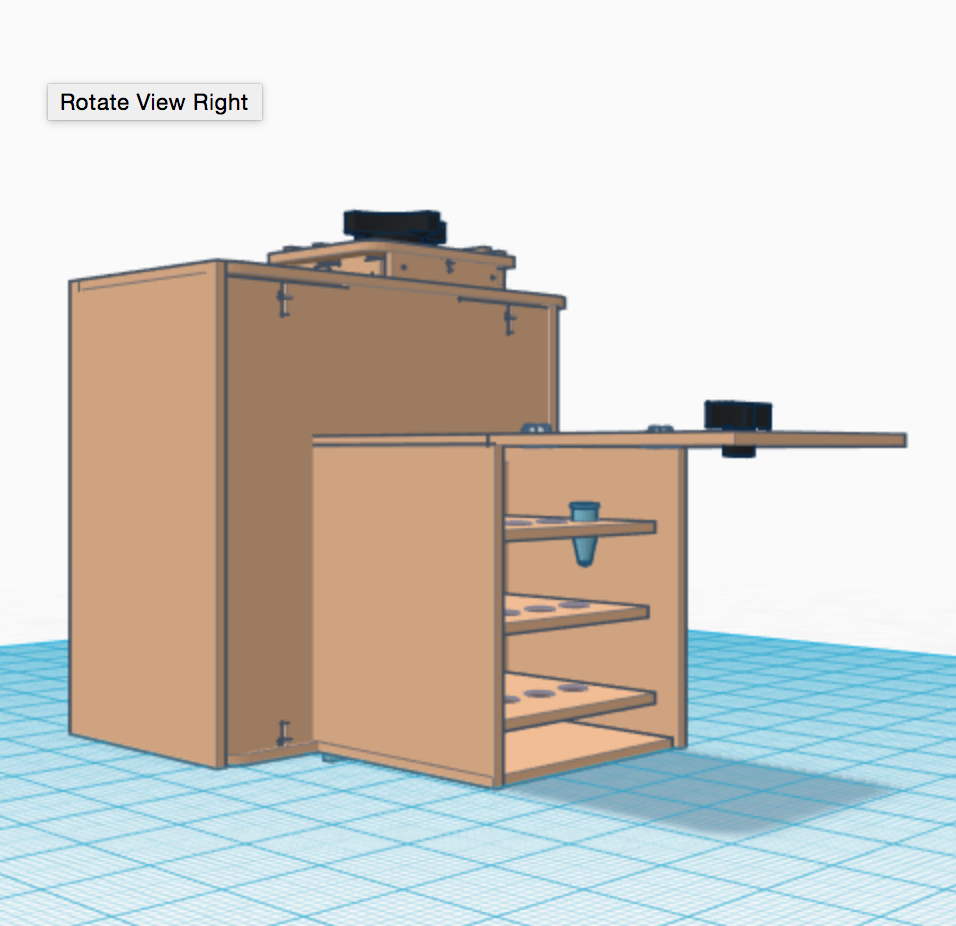
In this design, we added a small box using pre-existing designs, such as the sides or the back of the box which are available in thingiverse.com. When we started on the previous lab, we had to take pictures of the SYBR Green samples with one of our phones. We had some trouble getting clear pictures and it was time consuming. Our Open PCR machine can take pictures of the sample without having the trouble of taking pictures with our phones. Furthermore, the camera inside the small box moves in a fixed pattern in order to capture multiple samples without changing them, which saves time. The tubes for PCR will be designed so that labeling will not obscure 50 microliters of PCR mix with room for SYBR green as well. The tubes would be tested to ensure that allow emission light energy to be passed through in a way that its effect can be accounted for in the final data. Also that the compounds absorption can occur and be detected. Enhancement of fluorescence for qualitative comparison. The PCR machine would have an attached fluorometer running on the same power source and a USB cord. By attaching the machine to the computer the thermal cycler can be programmed through the computer and data/pictures can be sent straight to the computer for analysis. The camera would be mounted on the back in such a way that it can be programmed to move along the back, take a picture of each tube and then pass an LED light through the tube. On the other side adjacent the camera and LED light will be an interference filter to reduce noise, a photo-multiplier tube to amplify the current, and a photodiode to convert light to electricity so that it can be be sent as a digital readout through a USB cord with the picture straight to the computer. These last two components were not easily added By doing this an enormous amount of the researchers time is saved. PCR tubes can be removed, snapped apart, have SYBR green added straight to the tube and put in the fluorometer. This would also decrease the chance of mismatching data, and other user related errors. There would be no need for a smartphone or an irritating stand either.
Feature 1: Consumables KitBelow are lists for the strengths and weaknesses of the kit: Strengths
Weaknesses
Since micropipettors won't necessarily be reordered every time, a first time buyer will have the choice to add a micropipettor with tips to the order. All subsequent orders can be of only the consumables themselves. There will be an option to add more tips for convenience and ease of shopping. This packaging system will allow for effective use of time, since the all the items will be layered in one kit. Each item will be cushioned with bubble wrap. As there is no glass needed in either the fluorometer or the PCR with the new design it would be very unlikely for anything to break while being shipped. The box will have two compartments, one for temperature sensitive enzymes that is enclosed and insulated. The other will contain the stand, tubes, and if necessary micropipettor and tips. This kit will be available online on the company website with free standard shipping for customers ordering only tips or tubes, an added fee will exist due to the temperature dependent nature of the kit. It will be easily ordered online with a choice of often used primers and the option to design and order refill of primers and other components. Feature 2: Hardware - PCR Machine & FluorometerA Fluorometer is an instrument that detects fluorescence; quantity is related to the amount of florescent material and, less directly, proportional to the amount of the molecule being detected. By using the Fluorometer method, the ease of having an iPhone to collect data allows for convince. Also it is low cost, because it incorporates a special type of optics, known as an optical caustic, which is used. This system is very simple and low cost because there is no need for lasers, mirrors, or lenses. The procedure and instrumentation you used in Lab C is based on the properties of a superhydrophobic surface. The slide is coated with a rough layer of Teflon but has circles of bare glass. This allows the drops to cling to the glass but be repelled by the surface and creates the right environment for two things to happen. Firstly the light from the Blue LED is focused in the drop causing an increase in intensity which helps kick up the fluorescence event, and the SYBR Green I was added with a solvent that helps to form a complex with DNA that prefers to go to the surface. The current set up used requires the user to have a smart phone, use the stand to take pictures all while making sure that the phone is level to the light passed through the drop. This also requires a glass slide fixed into another stand. If only a little DNA was in possession and the drop were to slide of, which can very easily happen, this would be devastating. Incorporating both the LED Blue light and SYBR green which are not soluble in water provide the glow in the Fluorometer box that will be recorded with iPhone. PCR machine have thermal cyclers that regulate temperature during cyclical programs. PCR machines include things such as lid security, heat block reliability, and Interchangeable blocks to accommodate multi-well plates or tube strips, and programmable scripts. All of these advancements insure more accurate results. The thermal block holds tubes at the exact temperature needed for each cycle and programming controls the length and temperature of various PCR steps.
The weaknesses for the Fluorometer method was that it was inconvenient having to take on picture at a time, also the stand had to be perfectly level which was a hassle and provided inaccurate results. Also having to transport the images into image J and knowing how to keep track which image is what leaves a lot of room for user error. If a user did not have a smart phone with camera results will be skewed, if the user had no smart phone the experiment could not be conducted period. By streamlining the Flourometer and the thermal cycler into one machine this will greatly simplify the process of DNA detection. Tubes for the PCR will be specially designed tubes with emission filters to reduce noise. On the other side of these tubes are specifically designed silicon photodiodes which convert light into electrical current. This light intensity which is directly proportional to the concentration and can then be sent as data to a computer through a digital readout. There would also be emission filters and photomultiplier tubes to improve quality for data received by amplifying the current and reducing noise. In this way glass slides are avoided and all PCR reactions can be moved to the Flourometer at once. A camera will be mounted on a stand that moves around the back of the Flourometer. This will be programmed to automatically stop at each tube take a picture, and transmit the picture directly to a computer through a USB cord as well as send the necessary photons through for concentration detection. By doing this data can be described quantitatively and qualitatively automatically. Weakness of the PCR machine are the fact that it can only do one thing, and that is is to heat up the DNA and cycle it. Although it is effective at what it does, one goal of the new device is to add more uses to PCR machine to make it worth the high cost of it. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||