BME100 s2015:Group17 12pmL6
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
|
OUR COMPANY
LAB 6 WRITE-UPBayesian StatisticsOverview of the Original Diagnosis System This laboratory was approached in a group setting, there were 18 groups (5-6 students each), each analyzing two patients. This gave us a total of 36 patients. Nevertheless, the results calculated in the laboratory took error into consideration, and the groups with skewed results were taken out of the final results. These errors can be a results of contamination of PCR reaction, or any of the primers used in the non-sterile environment. Furthermore, the positive and negative controls could have been a source of error because of the quality of these controls. All these errors were taken into consideration when the final results were tabulated. Therefore, there were multiple calculations taken for each PCR reaction, therefore there were three different ImageJ images that were taken into account in the calculations. Also, during the calibration of the controls, there were three trial taken to taken in consideration the error of this laboratory. For diagnosis, this laboratory explored a wide array of probabilities and results. Based off our individual team data, it was concluded that both patients did not have the disease-associated SNP, because their data came out negative. As a whole, the class found different results. Based off the probability that a patient will get a positive final test conclusion, given a positive PCR reaction and the probability that a patient will get a negative final test conclusion, given a negative diagnostic signal will demonstrate the reliability of this diagnosis. Furthermore, the probability that a patient will develop the disease, given a positive final test conclusion and the probability that a patient will not develop the disease, given a negative final test conclusion will demonstrate more information on the reliability of these diagnosis.
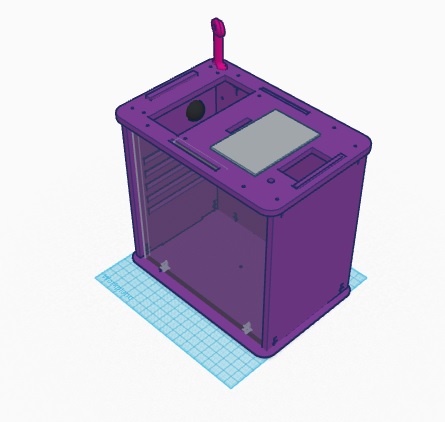
What Bayes Statistics Imply about This Diagnostic Approach Based off calculations 1 and 2, it can be concluded that the the Bayes values were very close to 100%. Which means that the diagnosis approach is reliable to decide whether or not a patient has the disease SNP or not. Based off calculations 3 and 4, the reliability of the diagnostic approach can be both reliable and unreliable. The values waver in the middle values, therefore, they are not close to 100%, but they are not small either. Therefore, these results can be either true or not true. Computer-Aided DesignTinkerCAD The online software, TinkerCAD, is an free online designing software, very similar to SolidWorks. The main difference between the two programs is the versatility. SolidWorks has a more advanced functions compared to TinkerCAD. The use of this software for the Computer-Aided Design lab allowed us to visualize the product, identify weaknesses and problems, as well as design a new and improved product. The 3D design feature allowed us to make a digital template of our product, making it easier to understand the features. Our Design
The new and improved PCR design has a variety of different features, compared to the original design, that will help with the effectiveness of the device. The three new features of the design include: a double chamber, integrated camera, and wireless capabilities. The double chamber feature will increase the efficiency and the maximum work capacity of the design. This will allow for more reactions to be analyzed. The integrated camera feature will make the results more accurate. The camera will be constantly taking pictures and vidoes of the reacting DNA. This will allow for individuals to be able to control whether or not the PCR reaction is taking place efficiently and correctly. Finally, the wireless capacity of the new design will allow for wireless connection to a phone or app that can notify and individual when the reaction is done. This will make the whole process more efficient and easier to manage.
Feature 1: Consumables KitBased of the original design, there really is no need to change this packaging feature of the design. Our team decided to keep the original packaging of the consumables. Therefore, there will be small plastic test tubes, a box with clean micropipettor tips, as well as the micropipettor itself. The same process for using these consumables will be the same as the original design.
Feature 2: Hardware - PCR Machine & FluorimeterThe PCR machine will be used to duplicate the DNA to be able to use this in the fluorimeter. The same process will be used, however there will be an additional step between transferring the DNA from the PCR machine and the fluorimeter. It will be encouraged to make sure the DNA was duplicated accurately, by using the camera feature of the newly designed machine. This is a major weakness in the previous original design, because the user has no way of knowing if that DNA duplicated accurately. Therefore, this extra step will allow the results to be more accurate. The fluorimeter was not changed in the new design. Thus, this process will remain the same.
| ||||||