|
OUR TEAM - GROUP 9
LAB 2 WRITE-UP
Descriptive Statistics
Human Study
Varying Statistics for LPS
| LPS Dose |
Average Inflammotin (pg/ml) |
Standard Deviation |
End Point # |
Standard Error |
Average Age
|
| 0mg |
3.834 |
1.523010177 |
10 |
0.481618106 |
66.7
|
| 5mg |
8.932 |
1.593931547 |
10 |
0.504045412 |
68.1
|
| 10mg |
61.622 |
30.11069386 |
10 |
9.521837451 |
68
|
| 15mg |
657.941 |
212.9429762 |
10 |
67.33848166 |
67.9
|
Overall LPS Doses
|
Inflammotin (pg/ml)
|
| Average |
183.08225
|
| Standard Deviation |
297.1368812
|
| End Point # |
40
|
| Standard Error |
46.98146608
|
Rat Study
Varying Statistics for LPS Doses
| LPS Dose |
Average Inflammotin (pg/ml) |
Standard Deviation |
End Point # |
Standard Error
|
| 0mg |
10.516 |
2.225551617 |
5 |
0.995296941
|
| 10mg |
11.112 |
7.402885924 |
5 |
3.310671231
|
Overall LPS Doses
|
Inflammotin (pg/ml)
|
| Average |
10.814
|
| Standard Deviation |
5.163022801
|
| End Point # |
10
|
| Standard Error |
1.632691166
|
Results
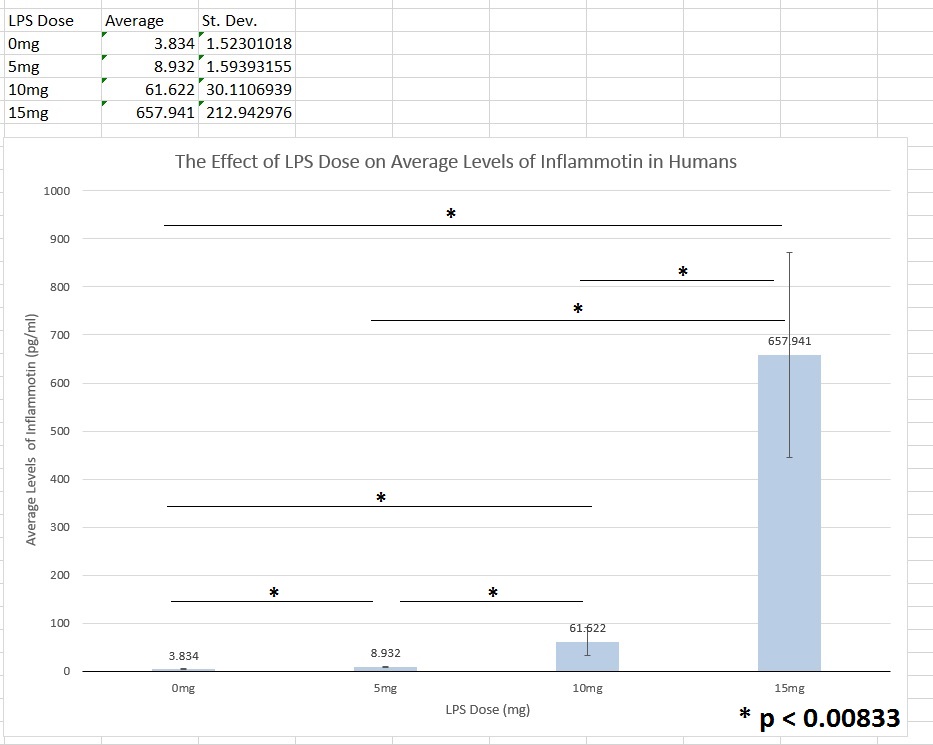
Human Study
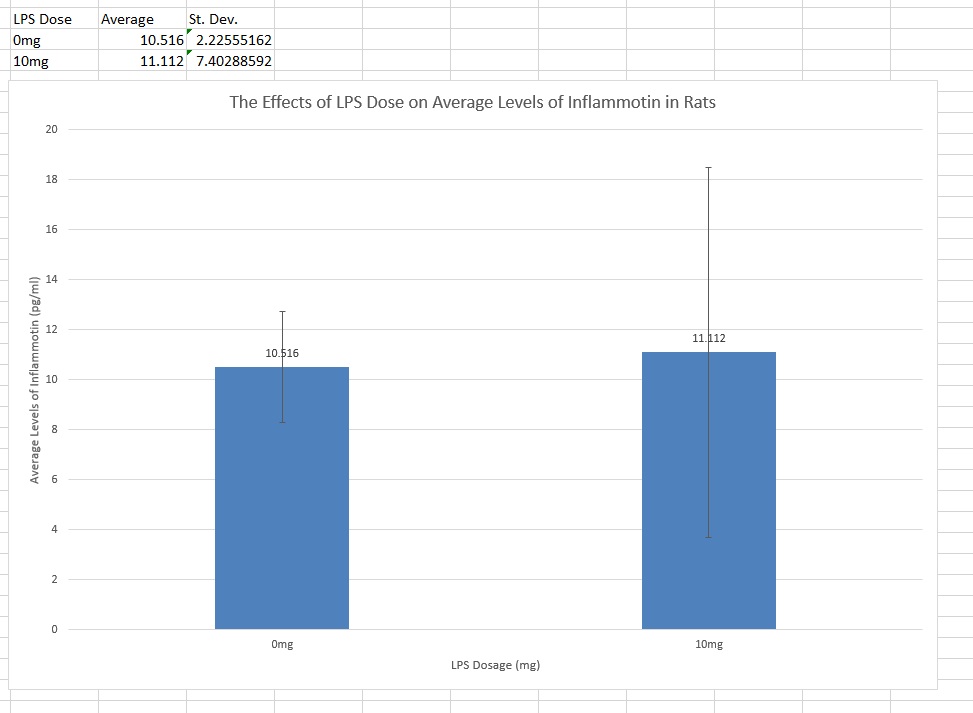
Rat Study
Analysis
Human Study
The data consisted of results from 4 different groups of subjects (0mg, 5mg, 10mg, and 15mg doses), so a one-way ANOVA test was run to find a p-value. The test yielded a p-value of 1.4 x 10-16, which means that the experiment had a percent uncertainty of less than 0.01%. This is much lower than the accepted 5% (or a p-value of 0.05), which means that the results were statistically different between groups. This leads to the conclusion that different dosages of the LPS will produce significantly different levels of Inflammotin in humans.
After running the ANOVA test, a Bonferroni correction was needed to test the statistical difference in each individual comparison. There were 6 comparisons based on the 4 groups (0 vs. 5mg, 0 vs. 10mg, 0 vs. 15mg, 5 vs. 10mg, 5 vs. 15mg, and 10 vs. 15mg). This means that the original alpha value of 0.05 needed to be adjusted; dividing 0.05 by 6 resulted in a new p-value of 0.008333. If the individual comparison yielded a p-value less than 0.008333, then the comparison was statistically different. Each comparison was less than 0.008333 (8.59631 x 10-07, 9.94377 x 10-06, 1.39436 x 10-08, 3.01859 x -05, 1.57101 x 10-08, and 6.4824 x 10-08, respectively), so all the results were statistically different from each other.
Rat Study
The data consisted of results from only 2 different groups (0mg and 10mg doses), so a T-test was run to find the p-value. The test yielded a p-value of 0.867403497, which means that the experiment had a percent uncertainty of about 86.7%. This is much higher than the accepted 5% (or a p-value of 0.05), which means that the results were not statistically different between groups. This leads to the conclusion that giving rats the LPS did not increase Inflammotin levels.
Summary/Discussion
Based on the data and Anova results, the level of Inflammotin in the blood in the Human Study generally increases as the dosage of LPS increases. This is because a p-value of 1.4 x 10-16 was achieved (p-value < .05), meaning that the differences in dosages are significantly different. They have a clear direct, positive relationship. However, in the rat study, the dosage of LPS had no clear relationship with the amount of Inflammotin in the blood as the p-value achieved was .867403487, meaning that the two groups are not significantly different (p-value > .05). All in all, as the LPS dosage rises in humans, the Inflammotin levels rise; however, due to the p-value outcome, the same cannot be said about rats with confidence.
|


