BME100 f2013:W900 Group12 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
|
OUR TEAM
LAB 2 WRITE-UPDescriptive StatisticsExperiment 1 Human Study Descriptive Statistics
Experiment 2 Rat Study Descriptive Statistics
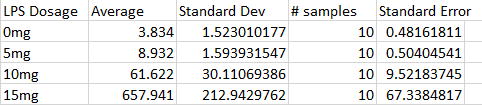
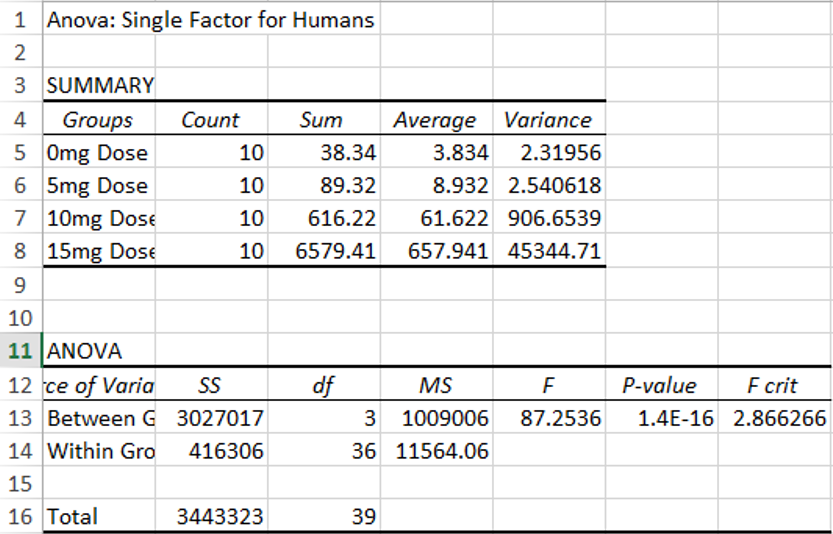
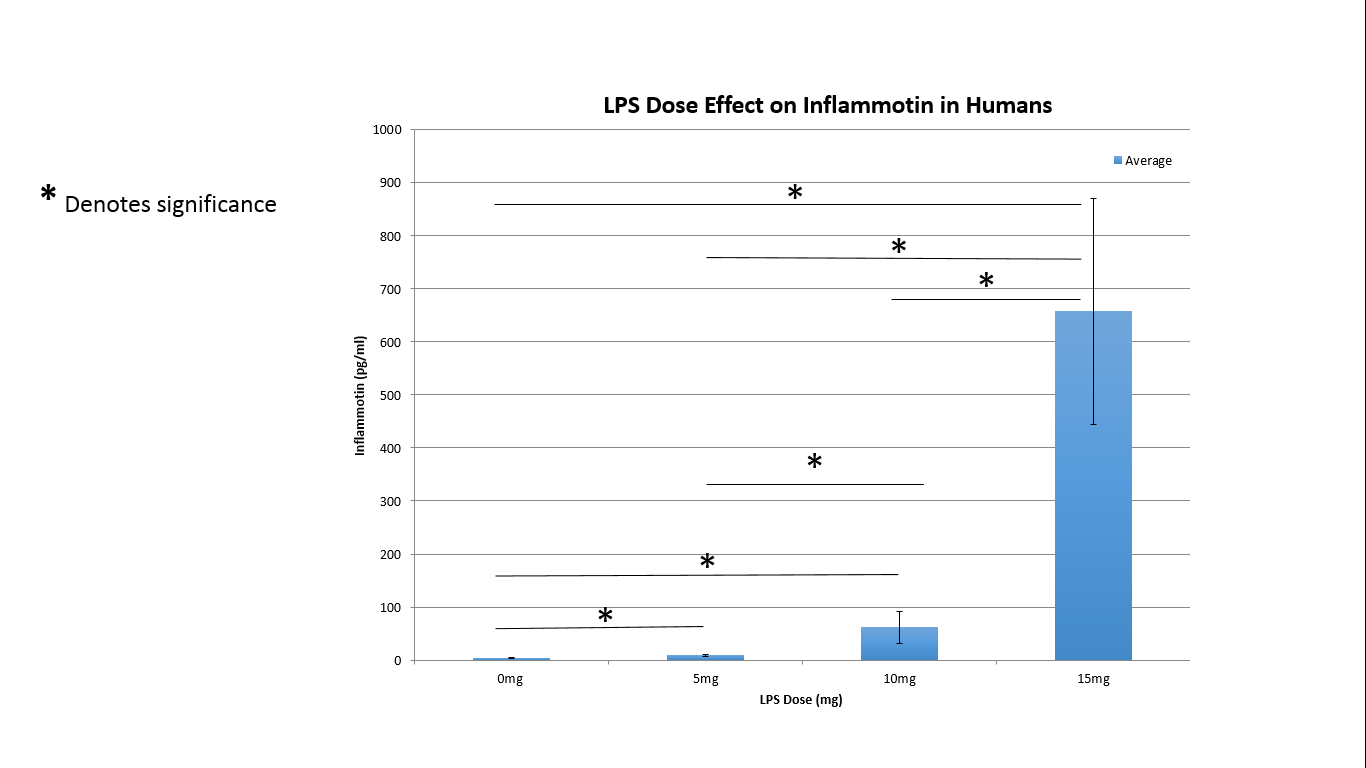
ResultsExperiment 1
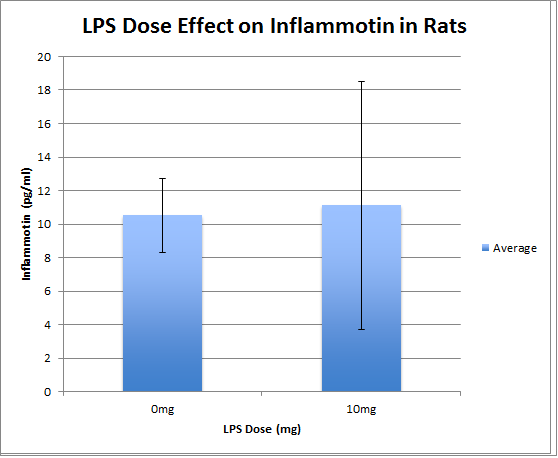
Experiment 2
AnalysisExperiment 1 Using the ANOVA test for the effect of LPS doses on Inflammotin levels in humans, we found that the p-value between groups was 1.4 x 10^-16, proving to be far below our .05 significance level. We can thus safely conclude that there is a significant statisical difference in Inflammotin levels between the groups of humans given different doses of the LPS drug.
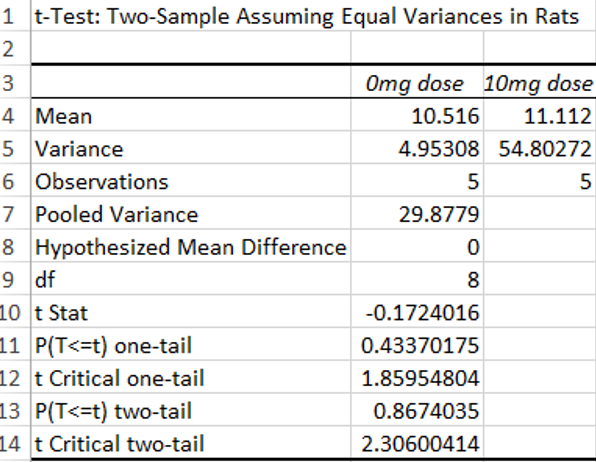
Using the T-Test to determine the effect of LPS doses on Inflammotin levels in rats, we discovered that our P-value is 0.867403, which is greater than 0.05, which is not significant. Because of this there is no statistical difference in Inflammotin levels between the two groups of rats that were given different doses of the LPS drug.
Summary/DiscussionIn Experiment One we observed the effect different doses of the inflammation inducing agent lipopolysaccharide (LPS) had on the amount of Inflammotin in the blood of humans. Through ANOVA, we determined there is indeed a statistically significant difference among all the dosages. This therefore provides support for our anticipated conclusion that increasing LPS doses increases the amount of inflammotin in the blood. In Experiment Two, in which inflammotin was measured in rats, the ELISA test revealed results that were not statistically significant. After performing a two sample t-test assuming equal variance, we found our p-value to be much higher than the accepted significance cut-off of 0.05. This evidence contradicts Kristen's assumption that LPS treatment in the rats significantly increased inflammotin levels. In conclusion, according to our study, LPS treatment in human subjects is a valid way to significantly increase inflammotin levels in the blood. In rats, however, LPS does not prove to be an effective treatment.
| |||||||