BME100 f2013:W1200 Group1 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
OUR TEAM
LAB 2 WRITE-UPDescriptive StatisticsExperiment 1: Human Study
Experiment 2: Rat Study
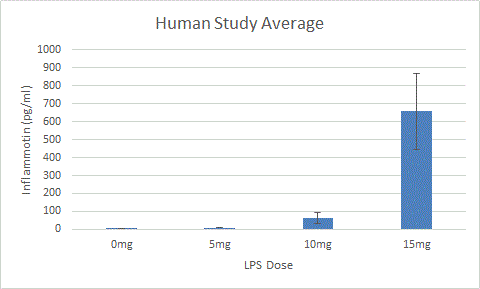
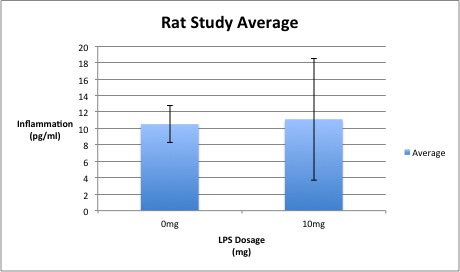
ResultsExperiment 1: Human Study Experiment 2: Rat Study
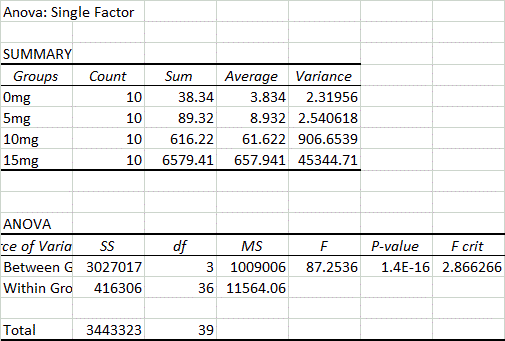
AnalysisExperiment 1: Human Study In order to evaluate the human study, an ANOVA test must be used because there are more than two groups of data. Because the ANOVA test resulted in a p-value of 1.4*10^-16, which is less than 0.05, it is considered statistically important. However, a post-hoc test using the Bonferroni Correction must be used to account for any errors that may have occurred while comparing the multiple groups of data. Six calculations of comparison must be done in order to compare each of the four groups of dosage to each other. Because there are six comparisons, the alpha number of comparison, which is usually 0.05, is adjusted to 0.0083333. A t-test for each of the six comparisons was calculated. Each test resulted in a value that is less than the corrected p-value, which means that it is indeed significant.
Experiment 2: Rat Study In order to evaluate the rat study, a t-test must be used because there are only two groups of data. Because the t-test resulted in a p-value of 0.8674, which is greater than 0.05, it is not statistically important; in this case, it does not correlate to a confidence level of 95%. Therefore, the changes in LPS dosage does not have a statistically significant effect on the rats.
Summary/DiscussionTwo experiments were completed - the first one on humans and the second one on rats. These two experiments were designed to test whether or not the amount of LPS increases the amount of Inflammotin protein (as anticipated). The first experiment was done on four groups of ten people. The dosage of LPS was increased by increments of 5mg per group starting with 0mg. Test subjects varied in age, ranging from 60-82 years old. The second experiment, which was tested on rats, used two sample groups of five rats each. The amount of LPS was increased by increments of 10mg per group. By observing the Inflammotin protein level in all four groups and evaluating the P-value number, the data showed that an increase in LPS dosage resulted in an increased level of Inflammotin protein in humans. However, the study on rats shows that this relation does not correlate since the P-value is greater than 0.05.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||