BME100 f2013:W1200 Group17 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
|
OUR TEAM
LAB 2 WRITE-UPDescriptive StatisticsExperiment 1 Experiment 2
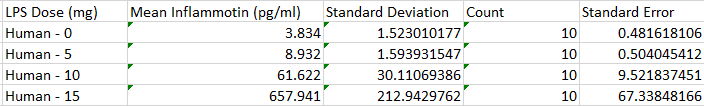
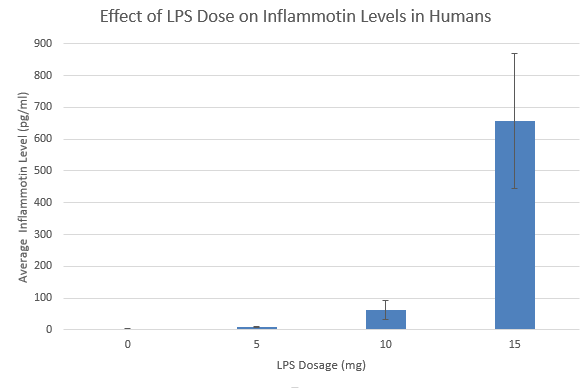
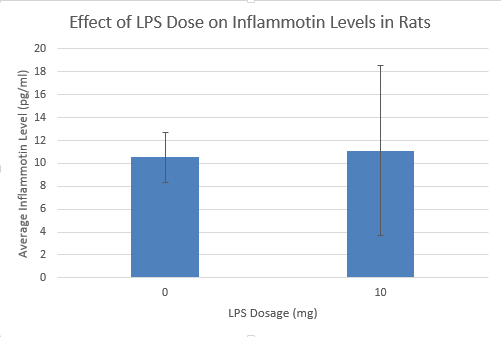
Results
Contibuted by Kevin Luong, Sonia Malek, Andrew Prindle, Jason Yang
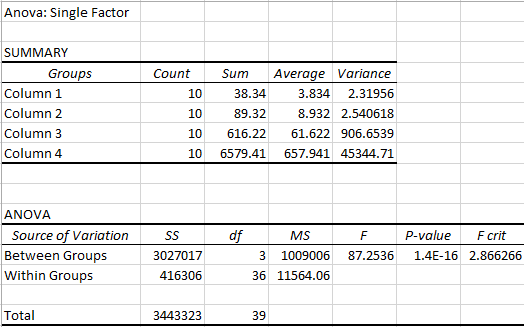
AnalysisContibuted by Kevin Luong, Sonia Malek, Andrew Prindle, Jason Yang
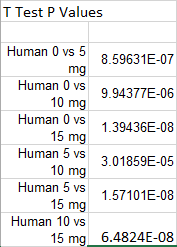
Summary/DiscussionTo determine whether the LPS dosage had an effect on the inflammotin levels. We examined the data of both the human and the rat. We averaged each trial in both the humans and the rats. We also calculated both the standard deviation and the standard error to assess the validity and the variability of the data. These calculations revealed that one of the trials ( human 15 mg ) had high standard deviation and error due to an outlier within the data of that trial that was markedly different than any of the other data in that trial. From the results of the data and the descriptive statistics performed, it appeared that there was a significant difference between the Human trials. However, there did not seem to show any significant difference between the rat trials. In order to verify that there was not a significant difference between the two rat trials a T-test was performed. This T-test has a P value well above the acceptable 0.05 limit and confirmed the suspicion that there is no significant difference between the two rat trials. In order to determine whether the apparent differences between the human trials were in fact significant, an analysis of variance was conducted on the human data. This analysis of variance yielded a P value that was well below the accepted threshold of 0.05, proving that the differences between human trial groups were in fact significant. From this discovery, it became necessary to look for significant differences between individual human trials. To do this, 6 post-hoc T-tests were conducted to test for significant difference between each of the trials. Each of these tests yielded a P-value result much lower than the required 0.05 and thus it can be concluded that all of the human sample results are significantly different.
Contibuted by Kevin Luong, Sonia Malek, Andrew Prindle, Jason Yang
| |||||||