BISC110/S12: Series 3 Experiment 9 Hill Reaction
Series 3- Lab 9 Photosynthesis -Photosynthetic Pigments and The Hill Reaction
In today's lab, you will measure the rate of electron transport in thylakoid membranes isolated from spinach chloroplasts using a procedure called the Hill Reaction. This procedure will allow you to measure the rate of oxygen evolution, and thus the rate of photosynthesis, in the thylakoids of isolated spinach chloroplasts.
Photosynthesis and Photosynthetic Pigments
Central to life on Earth is the photochemical process carried out by plants and cyanobacteria, wherein quanta of light are converted into chemical energy. In plants, light is absorbed by chlorophyll a and b and other antenna pigments of the light-harvesting complexes of Photosystems I and II.
Chlorophyll a and b impart the green color that one associates with plant leaves. Carotenoids, which are yellow pigments, are also present in leaves but are usually masked by the chlorophylls. It is only in the fall when the chlorophylls are degraded faster than the carotenoids that the yellow color becomes visible to us. The chlorophyll and carotenoid contents of plants can vary markedly with its age, or depend on environmental factors such as light intensity or quality during growth. The pale green appearance of a willow tree in early spring is markedly different from its olive-green of late summer. The intense dark green of "shade adapted" plants differs from the lacy green colors one sees at the top of a forest canopy.
Chlorophylls are found in the chloroplasts and are associated with the thylakoids, the internal membrane network of these organelles. It is now established that all chlorophylls are organized as discrete chlorophyll-protein complexes within the lipid matrix of the photosynthetic membrane.
The majority of chlorophyll a molecules (and all chlorophyll b and carotenoid molecules) function as antenna pigments. In combination with proteins, they form the light-harvesting complexes, which absorb and funnel light energy to the reaction center chlorophylls, thereby allowing the plant to utilize a broad spectrum of wavelengths for photosynthesis. Some of the chlorophyll a molecules serve specialized functions in the reaction centers of photosystems I and II, where the light energy is used to drive the reduction of components of the electron transport chain.
The energy from photons is passed via resonance energy transfer to a special pair of chlorophyll a molecules located in the reaction center, leading to the excitation and loss of electrons from these molecules. In each photosystem the excited electron from one reaction center chlorophyll is passed to the quinone primary electron acceptor thereby reducing it. The primary acceptor immediately donates its electrons to a neighboring molecule and so on through an electron transport chain to ultimately reduce NADP+ to NADPH. The resulting oxidized reaction center of photosystem II is able to split water molecules into protons, electrons and O2. The electrons extracted from water replace the electrons lost by the reaction center II chlorophyll. The reaction center chlorophyll of photosystem I is reduced by electrons coming from photosystem II.
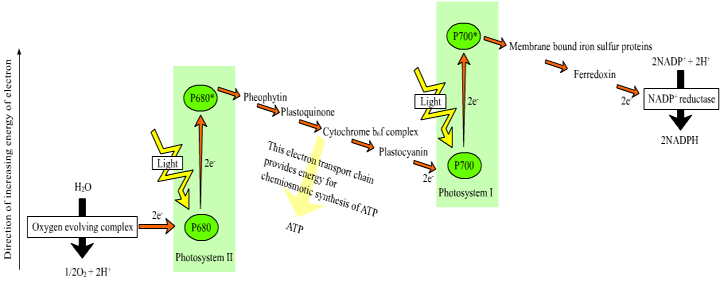
Figure 1. Process of electron transport in the light reactions of photosynthesis. Image from http://commons.wikimedia.org/wiki/File:Z-scheme.png
This electron transport is coupled in two steps to the formation of ATP through the mechanism of chemiosmosis. First, during the light reactions, the transport of electrons is coupled to the movement of protons from the stroma to the thylakoid lumen, forming a pH gradient across the thylakoid membrane. The sources of these protons are the splitting of water, which occurs on the lumenal side of the thylakoid membrane, and the transport of protons from the stromal side across the membrane into the lumen by the electron transport chain components plastoquinone and cytochrome b/f complex. In the second step, this gradient of protons is released when the protons diffuse through the membrane-spanning ATP-synthase molecule, which couples proton movement to the synthesis of ATP from ADP and Pi.
In today's lab, you will study the photosynthetic electron transport of thylakoid membranes isolated from spinach chloroplasts. During the isolation procedure, some of the more water-soluble components that function near the terminus of the main electron transport chain are lost. This makes it impossible to follow the production of ATP or the reduction of NADP+ in your preparations.
As is routine in studies using mitochondria and chloroplasts, we will supply an artificial electron acceptor to monitor "partial reactions" of the electron transport. These are chemicals that accept electrons at positions of the electron transport chain where exogenous compounds normally do not act in vivo. Frequently these electron acceptors are dyes specifically selected so their reduction can be monitored by spectrophotometry.
The dye reagent we are using in this experiment is 2,6-dichlorophenol indophenol (DCPIP). It is blue when oxidized and colorless when reduced. DCPIP accepts electrons between the electron chain components plastoquinone and cytochrome. The electrons are ultimately derived from water.
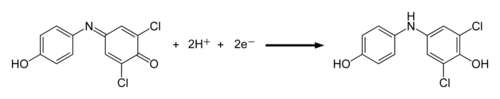
Figure 2. DCPIP reduction reaction
Figure 3. DCPIP acting as an electron acceptor. Animation from smabiology.blogspot.com/2008_12_01_archive.html
In 1937 Robert Hill showed that this partial reaction of the electron transport chain using DCPIP could be used to investigate the rate of oxygen evolution (from the splitting of water molecules in PSII) and thus the rate of photosynthesis in thylakoids of isolated chloroplasts. The reaction is now known as the Hill reaction and is still used today to determine photosynthetic rates in chloroplast preparations.
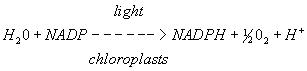
A. In vivo reaction
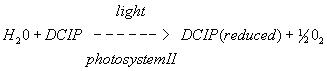
B. In vitro reaction
Figure 4. A. In vivo reduction reaction of NADP to NADPH in chloroplasts B. In vitro reduction reaction of DCPIP in photosystem II
Procedure
For the isolation of chloroplasts from spinach, a buffer is used since the leaves, when homogenized, can yield a low pH suspension. In the initial isolation step, the sorbitol serves to maintain an osmotic potential similar to that of an intact leaf. The divalent cation Mg2+, known to be important to membrane structure and function, is also included in the medium. All isolation and fractionation steps are performed at 4 degrees C to minimize proteolytic degradation of proteins.
In this lab you will examine the light reactions of photosynthesis by measuring the so-called Hill reaction in lysed chloroplasts. You will practice these measurements until you get reproducible results. Next week you will investigate the effect of various environmental factors on the light reactions of photosynthesis.
Be sure to keep all beakers and buffers on ice. Note that there are four different media used: grinding medium (100 mM Tricine NaOH pH 7.8, 400 mM sorbitol, 5 mM MgCl2), breaking medium (20 mM Tricine NaOH pH 7.8, 5 mM MgCl2), resuspension medium (50 mM , 100 mM sorbitol, 5 mM MgCl2)and reaction solution (100 mM sorbitol, 5 mM MgCl2, 50 mM NaPO4, 0.05 mM DCPIP). Note the differences among them. Be sure to use the correct solution. Read labels carefully throughout the experiment.
Preparation of thylakoid stock solution
- Work in groups of eight students in steps 1 and 2. Throughout the procedure, be sure to wash glassware with water and the round bottom centrifuge tubes that have come in contact with chlorophyll using 70% ethanol. Please do this as soon as you have finished using this equipment.
- Prior to lab, 60 g of spinach leaves were ground by our lab specialists with 160 mL of grinding medium in two blenders. The ground up spinach in each blender will be given to you in a 500ml iced cold beaker. A group of 8 students will filter the spinach through first 2 layers of cheese cloth into another iced cold 500 ml beaker. You must gently squeeze the spinach in the cheese cloth to express the liquid and leave the pulp and debris in the cheesecloth. Discard the cheesecloth with the pulp in the trash can. Filter the expressed liquid (containing your intact cholorophasts) through 8 layers of cheesecloth into another 500ml cold beaker. Gently squeeze the cheesecloth to express the refiltered liquid, then discard the cheesecloth and pulp in the trash can. Clean Up: Rinse beakers with water immediately BUT DO NOT DISCARD THE FILTRATE to be used in the next step!
- To isolate chloroplasts, divide the filtered extract into 4 equal portions and pour them into four 50 mL plastic round-bottom, capless centrifuge tubes. Balance each pair of tubes by transferring extract from the heavier tube to the lighter one, and centrifuge at 1000 x G in the SS34 rotor (see the conversion chart next to the centrifuge to convert RPMs to RCF which is also called g) for 5min in a Sorval refrigerated (4ºC) centrifuge. After the spin, each pair of students continues with one tube containing a pelleted chloroplast-rich fraction. From now on you are working in pairs. Carefully decant (pour off) the pale green supernatant and discard it. Save the green pellet (chloroplast-enriched fraction).
- Using a glass rod, gently resuspend the pellet by mixing 1–2 mL of breaking medium into the pellet. Make sure the pellet is completely detached from the wall and resuspended COMPLETELY. Avoid air bubbles (O2) that may oxidize enzymes and thereby reduce activity. The breaking medium is intended to shock the chloroplasts osmotically, thereby breaking open the organelles' outer membranes and releasing the stroma while leaving the thylakoid membranes intact. (Breaking medium: 20 mM Tricine NaOH pH 7.8, 5 mM MgCl2. Note the absence of sorbitol.)
- Bring the resuspended pellet to a volume of about 25 mL with cold breaking medium (50mL centrifuge tube will be about half-full), balance against a tube of water or another group, and centrifuge at 1900 x G for 5min. Discard the supernatant.
- Resuspend the resulting pellet containing your thylakoid-rich fraction in 1.5 mL of resuspension medium. This suspension is your stock preparation of thylakoids to be used in the Hill reaction. Keep it on ice. (Resuspension medium: 50 mM Tricine NaOH pH 7.8, 100 mM sorbitol, 5 mM MgCl2).
Table 1. Composition of Media
| Type of Medium | Tricine NaOH pH 7.8 |
Sorbitol | MgCl2 | Sodium Phosphate pH 6.8 |
DCPIP | |
|---|---|---|---|---|---|---|
| Grinding | 100mM | 400mM | 5mM | — | — | |
| Breaking | 20mM | — | 5mM | — | — | |
| Resuspension | 50mM | 100mM | 5mM | — | — | |
| Reaction | — | 100mM | 5mM | 50mM | 0.05mM |
Running the Hill reaction (work in pairs)
Materials: Each pair will have a ringstand with a test tube holder, and a 150W quartz-halogen projection lamp. You will have to find the best arrangement to measure the reduction of DCPIP accurately and reproducibly. A light meter and timer will be provided. Set up 5 ‘reaction tubes’ to begin with, adding 5 mL of reaction mixture, but do not add the thylakoid membranes, which must be kept on ice. Add these thylakoid membranes just before each reaction mixture is tested. [Reaction mix: 50 mM sodium phosphate, pH 6.8 buffer, 100 mM sorbitol, 5 mM MgCl2, 0.05 mM DCPIP]
Blanking the Spectrophotometer ( Appendix B): Use a tube containing 5 mL of resuspension solution and 50µL of your thylakoid suspension to blank the Spec20 at 580 nm (filter level to the left). With Parafilm® over the top of the tube, invert once to mix, then wipe the tube with a Kimwipe® before blanking the instrument. Do not vortex. You may need to prepare a fresh blank periodically.
Varying the Reaction Rate: The Hill reaction occurs at different rates depending on the light intensity supplied to the thylakoids. You will measure the reaction rates by taking absorbance readings over a 90 s period in 15 s intervals. We will use the drop in absorbance over time as a measure of the rate of the Hill reaction.
Depending on the number, distribution (clumped or not) and condition of thylakoids in your reaction tube, you may obtain varying reaction rates with a given light intensity. We measure the incident light using a light meter in units of µmol photons m-2 s-1.
Typically, the light intensity is set to 80-100 µmol photons m-2 s-1 in order to drive the reaction at the desired rate—a drop in absorbance per 15 s that is consistent throughout the 90 s trial. The 15 s interval consists of 10 s of illumination and 5 s to read the absorbance. In a 90 s experiment, this data collection interval of 15 s yields 7 data points (i.e., absorbance readings at time = 0, 15, 30, 45, 60, 75, and 90 s).
Measurement of Hill Reaction Rates: For each reaction tube, do the following in turn.
- Mix thylakoid suspension gently by flicking bottom of tube.
- Immediately remove 50 µL of thylakoid suspension and add it to a reaction tube containing 5 mL of reaction mixture, mix gently by inverting once and place tube in the Spec20 to obtain a "dark reading" with the spectrophotometer set at 580 nm. This is the time = zero seconds absorbance reading. Expose the thylakoids to the light and take care not to shield the tube from the light with your hand.
- Take absorbance readings every 15 s (10 s illumination + 5 s reading) for 90 s (7 readings). Record absorbance readings and times in your lab notebook. Repeat the experiment, i.e., do several 90 s runs, until you obtain a consistent rate of DCPIP reduction. Then repeat the experiment, to see if you can reproduce your data consistently.
A typical absorbance reading at time = 0 s is 0.8. If the drop in absorbance per 15 s interval is 0.05, then at time = 90 s, the absorbance reading should be 0.5.
If the drop in absorbance per 15 s is not consistent in the 90 s period, you can try varying the light intensity by varying the setting of the intensity knob on the fiber-optic illuminator or by changing the distance between the light source and your thylakoids. Be sure to hold your test tube in the same position for each 10 s of light exposure during the 90 s trial. If increasing or decreasing the light intensity and maintaining consistent positioning of the test tube does not work, please see your instructor.
Plotting the Data: Please plot your data with Time (s) on the x-axis and Absorbance at 580 nm on the y-axis. The reduction of DCPIP is linear until DCPIP becomes limiting which is indicated by a leveling off of the absorption near the end of the reaction. Since you are interested in the maximum rate of DCPIP reduction, you are only concerned with the straight-line portion of the curve. Remove data points that are not in the linear portion of the curve. To facilitate your comparison of the different rates of reaction, plot each data set as a scatter graph, and add a trendline. With time in seconds plotted on the x-axis the slope of the line given will reflect the change in absorbance per second. A description of simple linear regression can be found in Appendix E Microsoft Excel 2004 Instructions for Regression Plots .
Self-Designed Experiment On A Variable Affecting Electron Transport Rate In Photosynthesis
Here is a downloadable list of reference articles on variables affecting the Hill Reaction: Media: Hill_Reaction_resources.doc
In Lab 10, you and your partner(s) will isolate thylakoids from spinach using the same protocol as you used today. You will test the effects of one environmental parameter on the Hill reaction rates. Please sign up today with your group to test the effects of one of the following parameters: temperature, light intensity, wavelength, inhibitors or uncouplers. See Lab 10 for additional information. You will work on a protocol as a group of 4, and in Lab 10 you will work as partners to do replicate experiments using the same protocol.
All partners should take part in composing the proposal, which should include:
- A hypothesis with rationale and a plan for controlling confounding variables and/or acquiring an appropriate baseline for comparison.
- A list of reagents needed with protocols of how you will make all solutions and dilutions used in your experiment
- Your protocol (with sufficient detail, i.e., how to set up your tubes, etc.)
Your instructor will either approve your proposal, or suggest improvements.
Laboratory Cleanup
- Wash glassware with water and centrifuge tubes using 70% ethanol.
- Discard reaction mixtures in sink and test tubes in glassware disposal box.
- Glass pipettes should be left to soak in the pipette canisters, tips down.
- Place micropipette tips in the small orange bags at the bench.
- Pasteur pipettes and cover slips go in the glass container (blue cardboard box).
- Remove the last test tube from the sample chamber, and turn off your Spec20.
- Quit out of all applications on your computer.
Assignment
- If you have not had your protocol approved by your instructor for your experiment exploring a variable that affects the Hill reaction, e-mail your proposed experiment to your lab instructor (cc your partners) early enough to leave time for feedback, modification of the proposal by you, resubmission of the proposal to your instructor, etc. Also, the prep staff needs sufficient time to provide the materials you require. Your compliance with getting an approved final experimental protocol before the deadline set up your instructor will be factored into the grade that you will receive for the paper based on this experiment.
- Since the paper that you will write on your experiment in Lab 10 will be a full report that includes a Materials and Methods section, you will write a practice Materials and Methods section on the Hill Reaction protocol that you performed today and turn it in at the beginning of lab next time. Remember that a Materials and Methods section does not read like the protocol descriptions in this lab wiki. Refer to the Materials and Methods section in the Guide to Scientific Writing in the Resources part of this wiki for help and to this downloadable handout: Media: How_TO_WRITE_HILL_M&M.doc. You should also refer to the Methods section in some of the journal articles that you will use as references to model what should be included or left out. Remember that concentrations of key reagents should be expressed as final (effective concentration) at crucial points in the experiment.