BISC110/S10: Lab 7 Taster SNP2
Lab 7 -Taster SNP Week 2
Today you will finish determining your genotype at the PTC gene locus by testing for one of the SNPs in the PTC gene. Remember that last week you began the process by isolating your own DNA and setting up a PCR reaction to amplify a region of the PTC gene which contains the SNP of interest. Today you will digest your PCR product with the restriction enzyme Fnu4H1 and analyze the products by agarose gel electrophoresis. We should also have time for a data analysis discussion and writing workshop in preparation for the paper you will write on your findings (paper due at the beginning of Lab 8).
PART I: Digestion of your PCR products with the restriction enzyme Fnu4H1
Now that you have plenty of copies of the region of DNA we’re interested in, you will set up a restriction enzyme digest. How will this tell you what SNP you have? Restriction enzymes are quite specific about what DNA sequences they will cut. If the sequence is different, even by one base, then they will not cut. We have made use of this fact by choosing a restriction enzyme, Fnu4H1, which will cuts one variant of the PTC gene SNP, but not the other. Because the restriction digest will take an hour, you will set it up at the beginning of the lab period.
1. Locate your PCR tubes from last week, and set them out at your bench to thaw.
2. Each student will set up a restriction enzyme digest of her own PCR product and one student of each pair will also digest the pcr product of a heterozygous(PAV/AVI) control.
3. Get a clean microfuge tube and label it with your id number on the course spreadsheet, and D to indicate that this will be the digested DNA. If you are doing the control, label another microfuge tube with a D and a C.
4. Use your P20 set to 10μl and carefully pipette 10 μl of the appropriate PCR product into the labeled microfuge tube(s). Be sure to save remaining PCR product to run the DNA uncut on your gel. Add the label U, for undigested, to the pcr tubes that contain the undigested, amplified Taster gene from the DNA extracts prepared last week.
5. Add 10 μl of restriction enzyme “cocktail” to the D tube(s). The "cocktail" contains Fnu4H1 and an appropriate buffer. (In each 20 microliter reaction volume there is 2 µl New England Biological Buffer #4 (http://www.neb.com), 2.5 units (in 0.5µl) of FNU4H1 (New England Biological product RO171), 7.5 µl purified water and the remainer is template DNA). Mix well by pipetting up and down.
6. Incubate your digests in the 37C heat block for 60 minutes.
DO NOT THROW AWAY THE PCR TUBES because you will run both the cut and the uncut DNA on your gel after your restriction enzyme digestion is completed.
PART II: Agarose gel electrophoresis.
An animation of how to pour and run an agarose gel is available at: [| http://www.wellesley.edu/Biology/Concepts/Html/gel.html]
You can do several of these steps while you are waiting on your restriction digests.
We will be using a Biorad gel system. Each pair of students will use one of these systems.
Setting up the gel apparatus
a. Place the gel-casting tray in the gel box on the raised platform, positioning the comb slots nearest to the negative electrode (Black).
b. Place the metal wedges into the gel box in the slots on either side of the gel-casting tray.
c. Place the comb into the slots closest to the edge of the gel-casting tray.
Ask your instructor to check your set up.
1. Dilute the buffer and make an agarose solution:
Each pair of students should make a 30 ml solution of 1.5% (w/v) agarose in 1X TBE (89mM Tris Base (TRIZMA), 089mMboric acid, 2mM EDTA (pH 8.0).
a. You will be provided with a 10X TBE stock solution. You will need 300ml of buffer total for preparing your agarose solution and for running your gel. Calculate the amount of 10X TBE you will need to prepare 300ml of 1X TBE.
measure out _____ ml of 10X TBE
add_____ml of water.
b. Use a stir bar to mix your 1X TBE solution (briefly) in a 500ml flask.
c. Calculate the amount of agarose you should weigh out to make your gel.
for 30 ml 1.5% agarose:
weigh out _____ g of agarose and put that in ______ ml of the 1x TBE buffer that you diluted in step a.
d.Check your calculations with your lab instructor before proceeding.
e. Using one of the balances in lab, carefully weigh out the appropriate amount of agarose using a piece of weighing paper and one of the top loading balances. Transfer the agarose to the small, empty 125 ml Erlenmeyer Flask at your bench and add the proper amount of 1X TBE buffer that you previously diluted. Swirlto mix. The agar will NOT go into solution without high heat (the next step).
f. Cover the open top of your 125ml flask with the little beaker you found on it originally and microwave the agarose solution for ~15-20 seconds on high power. Use the “hot hands” to handle the flask when removing it from the microwave, since the glass will get very hot.
The agarose should be completely melted and the solution should be uniformly clear when you swirl it around. If it isn't, microwave it again for a few more seconds (~10).
g. Immediately take your flask to the instructor's bench and ask your lab instructor to add 3 μl of SYBR Safe stain to your flask.
PUT ON GLOVES.
Any time you work with your agarose or gel for the remainder of the lab, you will need to be wearing gloves.
Wearing gloves, swirl the flask gently to mix, taking care to avoid the formation of bubbles. If you sit the flask on the lab bench and make a slow figure eight, this form of mixing works pretty well. Then immediately proceed to the next step since everything is highly time sensitive at this point.
3. Cast a gel:
If you pour the agarose while it is too hot, you can crack or warp the casting tray, which may cause leaking, but if it too cool it will be a lumpy mess when you pour it and you will have to start all over again.
d. When the lip of the glass flask is cool enough to touch with your hands (approximately one minute after you add the Sybr green) you can begin to pour the agarose into the get-casting tray that your instructor has checked for correct assembly.
e. Start out very SLOWLY pouring agarose into the CENTER of gel-casting tray, trying to avoid the formation of air bubbles. If you start in the center, by the time the agarose gets to the edges, it will be just cool enough to seal them without leaking. If you start out pouring VERY slowly, the casting tray is less likely to crack from the heat because the temperature can equilibrate incrementally.
If bubbles form, remove them quickly before the gel starts to solidify by having your partner, pop them with a Pasteur pipette.
f. When you have poured the whole flask of molten agarose, allow it to set completely. When it has solidified, it will appear relatively opaque. Ask your instructor to check before you go on the next step.
g. Carefully remove the metal wedges from the gel box.
h. Fill both sides of the gel box with 1xTBE running buffer, until the buffer is about 3mm above the gel surface.
i. To remove the comb from the gel, pull it slowly and straight up so that the wells are not damaged.
4. While you are waiting for your restriction digests to finish:
a. Prepare your undigested samples: Each student will run their own undigested PCR product and student who digested the heterozygous control will set up an additional tube for the uncut remainer of the heterozygous control pcr product.
i. Label a microfuge tube with your code number and indicate that it contains undigested DNA (U). One student per pair will label an additional tube for the pcr product called "Undigested Control (UC)"
ii. Pipette 10 μl of the appropriate PCR product from the PCR tube into the labeled microfuge tube.
If you do not have 10 μl left, add as much of your PCR product as you have.
iii. Add 10 μl of water to each labeled microfuge tube. Mix thoroughly by pipetting up and down.
iv. Add 2.2 μl of loading dye to the microfuge tube. Mix thoroughly by pipetting up and down.
Don’t forget to use a clean, new tip for each tube.
Loading dye contains the following components:0.4 % Ficoll 400, 1.8 mM EDTA, 0.55 mM Tris-HCl, 0.00117 % SDS, 0.025 % Bromophenol Blue pH 8.0 @ 25°C)
Bromophenol blue
This is a negatively charged dye that move towards the positive pole..
Ficoll
Ficoll is added to increase the density of the sample.
This causes the sample to sink into the well, since it is heavier than the liquid in the well.
EDTA
EDTA helps keep the nucleic acid in the sample stable by inhibiting nucleases that might be present.
It does this by binding divalent cations, such as Mg2+, that are necessary for nuclease activity.
Do not throw the stock tube of loading dye away when you clean up after this lab!
b.Your lab group should acquire a sample of marker lane DNA (ready to load) from your lab instructor .
This marker lane DNA contains DNA fragments of the following sizes: 766 bp, 500 bp, 300 bp, 150 bp and 50 bp. Using what you know about the SNP region we amplified with PCR and the location of the Fnu4H1 II cutting site, you should be able to predict where you might see DNA fragments (looking like glowing bands) in relation to the position of the marker lane bands.
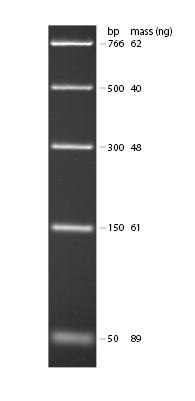
DNA Ladders: 0.3µg of “PCR Ladder” and visualized by ethidium bromide staining on 1.8% TBE agarose gel. Ladder from New England Biological http://www.neb.com (NEB3234)
d. Practice gel loading.
Loading an agarose gel can be tricky. Your lab instructor will demonstrate how to use the practice dishes to learn to load a gel properly. Practice until you are comfortable and proficient.
5. Prepare your restriction digest for loading:
a. After your restriction enzyme digest is complete, microfuge your tubes briefly (5-10 seconds) to get any condensation from the lid into the bottom of the tube.
b. Add 2.2 μl of loading dye to each tube of digested DNA (one tube for each member of your lab group, plus the heterozygous digested control).
Each pair of students should now have 7 tubes which contain DNA and loading dye: one digested tube for each group member, one undigested tube for each group member, one digested control DNA sample, one undigested control DNA sample and one tube of marker lane DNA.
6. Loading and Running the Gel
a. Fill out the gel template provided so you know which lane each sample will be loaded. Copy this template into your lab notebook. You will load 20 microliters from each tube (including the pcr marker) into separate gel lanes. .
Check your tubes before loading: microfuge briefly (around 15 seconds) if necessary. Be careful not to let any bubbles into the pipette tip when you are drawing the sample into the tip. Give the template to your lab instructor.
b. When all of the samples have been loaded, check the orientation of the gel with respect to the electrical leads.
Remember “RED AHEAD”; DNA, having a net negative charge, will migrate toward the positive (red) electrode.
c. Placing the lid on the box connects the leads to the gel box. Connect the other ends of the leads to the power supply. Check that the power supply is set at 100 volts, and then turn on the "juice". Observe bubbles rising from the electrodes at each end of the tank (more from the negative electrode than the positive electrode). After 1–2min, observe the dye leaving the wells. Run the gels for approximately 25 minutes or until the tracking dye has moved about 3-5 cm from the starting point. Ask your instructor to check before you stop the electrophoresis by turning off the current.
7. Gel photography
a. When the gel has run sufficiently to separate, but not to lose the DNA fragments of interest, disconnect the leads from the power supply and remove the lid from the gel rig.
b. Wearing gloves, carefully remove your casting tray (still containing the gel) from the gel rig and transfer it to a labeled plastic sandwich box at your bench.
c. After the electrophoresis is complete, your instructor will take you to the instrument room next door, so that you may take pictures of your gels. Follow the directions in Appendix G to use one of the imagers to visualize your DNA fragments and to take a photo.
SYBR SafeTM stain is a fluorescent dye that binds to nucleic acids. It has excitation maxima at 280nm and 502nm, and an emission maximum at 530nm. You will place your gel on a UV transilluminator, a device that shines ultraviolet light through the gel. Where nucleic acids have been stained with the SYBR SafeTM stain, a fluorescent green glow will be detected by the digital camera connected to the transilluminator.
d. Once you have captured a satisfactory image of your gel, send it to your FirstClass® account, as well as posting it on your lab conference.
8. Analyzing the Gel
a. Ascertain whether your enzymes cut your DNA.
How can you tell? How many fragments do you see for each person in your lab group?
b. Use the DNA ladder to estimate the size of the fragments you see.
9. Recording Your Data
Determine what genotype you are: PAV/PAV, PAV/AVI, or AVI/AVI. Be sure to check your interpretation of the gel with your lab instructor. Once you are sure of your genotype, record your data in the class excel spread sheet where you have already recorded your PTC sensitivity phenotype. NO NOT LEAVE LAB UNTIL YOU HAVE RECORDED YOUR GENOTYPE ON THE COURSE SPREADSHEET AT THE INSTRUCTOR COMPUTER!
Class results will be compiled and presented to protect anonymity. You are not required to share your personal data with anyone else in lab (though you are free to do so if you wish).
Part III. Data Analysis & SCIENCE WRITING DISCUSSION
While your restriction enzyme digestion and electrophoresis are running, we will have time to talk about your expected results and their significance. We will also address how you might write about these data in the form of a primary research report due at the beginning of the next lab. This will be a full paper (except that you may omit the Materials and Methods section) so we will concentrate on the sections you have yet to write (Abstract and Introduction) using the Wooding article as a possible model. You should have brought a hard copy of this article with you to lab: Wooding et al., Natural Selection and Molecular Evolution in PTC, a Bitter-Taste Receptor Gene. Am. J. Hum. Genet. 74:637–646, 2004.
Assignment
Write a full scientific research report due at the beginning of Lab 8 using your data to answer the question, What is the distribution of the Taster PTC phenotype and genotype at Wellesley College?
You will use your data to address the last and one other of the following possible aspects of this question:
Tasters vs Non-tasters
Genotype: PAV/PAV vs. PAV/AVI vs. AVI/AVI
Allele frequencies: PAV vs. AVI
Agreement and disagreement between prediction of phenotype from genotype and what that might mean in terms of the presence of rarer alleles (other than PAV and AVI--use the Wooding 2004 paper as a source)
Link to Labs in this Series and the Photosythnthesis Series
Lab 6 Taster SNP 1
Lab 7: Taster SNP 2
Lab 8: Photosynthetic Pigments
Lab 9: The Hill Reaction
Lab 10: Variables Affecting the Hill Reaction