BISC 219/F10: Lab 4
Lab 2: Gene Mapping
Lab 3: Linkage Test Part 1
Lab 5: Mapping Part 2
Lab 6: Score
Lab 4: Complete Linkage Analysis; Start Mapping and Complementation Testing
Complete Linkage Testing
To determine in what chromosome or linkage group your dpy mutation is located record the number of wild type, dpy, unc and dpy unc mutants by examining, scoring the phenotype and removing that animal from the plate for each of your four crosses with reference unc mutations. If you see no double mutants (du/du), it could be taken as an indication that your dpy mutation is on the same chromosome or linkage group as that unc mutation. Remember that your mutation will only reside on a single chromosome; therefore, the ideal result is that your mutation segregates independently with respect to 3 of the 4 reference mutations. If you didn't get 4 successful reference crosses, score those you have and see your instructor for help. Can you determine the linkage group of your dpy mutation?
Mapping a Mutation to a Specific Location on a Chromosome and Gene
Assuming that you have determined the linkage group on which your Dpy mutation resides, we will continue working with that strain only. You now suspect that the two mutations (the Dpy mutation of interest and the reference marker Unc) are on the same chromosome. Can you use Worm Base to confirm which chromosome that is from looking up the reference mutation and noting its location? To begin to calculate distance between the marker and your mutation of interest, you will separate 5 Unc mutants to 5 individual plates. As described above, most of these individuals are wild type for the Dpy mutation (+ u/+ u); however, we hope some are heterozygous for the Unc mutation (d u/+ u). How does this happen?
To Do Today
- Separate 5 Unc mutants to 5 individual plates (Hint choose these Unc worms from plates that also have Dpy mutants!)
- Label each of these plates with "Mapping 1" and your initials and the date with your PURPLE Sharpie.
- Incubate the plates at 23°C for 3 days
3 days later
- Screen your 5 plates for double mutants (dpy mutants that either do not move or coil).
- Pick 3 such putative double mutants to separate plates to allow them to self fertilize. If they are truly double mutants then all of their progeny should be double mutant as well and the progeny will be used for the next cross.
- Label each of these plates with "Mapping 2", the genotype d u/d u and your initials and the date with your PURPLE Sharpie.
- Incubate the plates at 23°C until next lab period.
Complementation
It is not unusual to have series of mutations that confer similar phenotypes and also map to a identical or similar location on a chromosome. In such cases, the practicing geneticist performs a complementation test to determine if the mutations are allelic (that is, in the same gene) or non-allelic (different genes with the same phenotype).
If the mutations are allelic (in the same gene but on different sister chromatids) there should be no complementation, thus you observe the Dpy phenotype.
If the mutations are non-allelic (in different genes) there should be complementation, thus you observe the wild type phenotype.
These conditions are true when you are working with recessive mutations.
For more information about complementation see your Genetics textbook or Wikipedia Definition.
The specifics of strain construction vary depending on the experimental organism; however, the basic strategy in all cases is to construct a double heterozygote and then to examine the phenotype of this organism. Remember that a wild-type phenotype indicates that the two mutations complement one another (cancel each other out) and are therefore in different genes. Conversely, a mutant phenotype suggests the mutations are allelic to one another (that is, they fail to complement).
Over the next two weeks we will construct different double heterozygotes containing your dumpy mutation of unknown location (dpy-u) and dumpy mutations of known location (dpy-k) on the same chromosome as your unknown dpy mutation as follows:
First obtain heterozygotes for your dpy mutation of unknown location through
Cross #1: unknown Dpy hermaphodites (dpy-u/dpy-u) x N2 males (+/+) yields dpy-u/+ progeny
Then use those heterozygous males resulting from Cross #1 in the next cross:
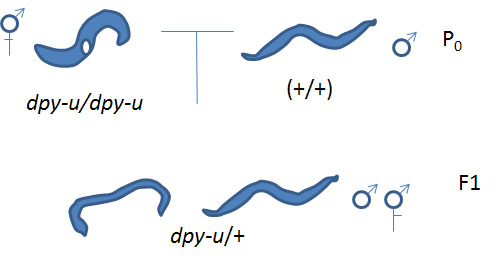
Cross #2: dpy-u/+ males x known dpy hermaphrodites (dpy-k/dpy-k) yields some dpy-u +/+ dpy-k progeny. [Note: we need to use heterozygote males for our unknown dpy mutation (dpy-u/+) because homozygote dumpy males do not mate properly]
Available Dpy strains:
| Chromosome 1 | Chromosome 2 | Chromosome 3 | Chromsome 4 |
|---|---|---|---|
The phenotype of the double heterozygote is then scored.
Keep in mind that, in this experiment, you will determine the allelic counterparts of your “unknown” Dpy mutation by placing it (your unknown mutation) with mutations in different Dpy genes that have been previously mapped (i.e., known mutations) to your chromosome of interest. Our goal is to determine whether or not our “unknown” Dpy mutations are allelic to any of the known mutations.
To Do Today
- Determine which Dpy mutations are located on the same chromosome as your unknown.
- See which of these mutant strains we have available to determine how many crosses you will perform.
3 days after lab:
You will set up Cross #1: Cross your unknown Dpy hermaphrodites with N2 males [(dpy-u/dpy-u) x N2 males (+/+)] by placing three to five L4 Dpy's on a new plate with 3-4 N2 males. You will be picking your own wild type males from a mixed population of worms so be sure to transfer your males to a transfer plate before adding to the crosses. It is essential that the ONLY wild-type animals present on this plate are males.(There should be no wild type hermaphrodites because you desire +/dpy-u males from this plate for the next cross, and not the +/+ males that would result of wild-type hermaphrodites were included on the plate). Do this in duplicate.
Label your plates with your initials and the date and the genotype dpy/dpy (H) X +/+ (M) with your ORANGE Sharpie.
Incubate the worms at 23°C until next lab period.
