BIO254:Gprotein: Difference between revisions
| Line 8: | Line 8: | ||
===A molecular switch=== | ===A molecular switch=== | ||
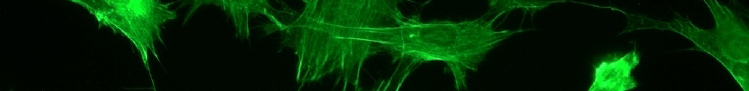
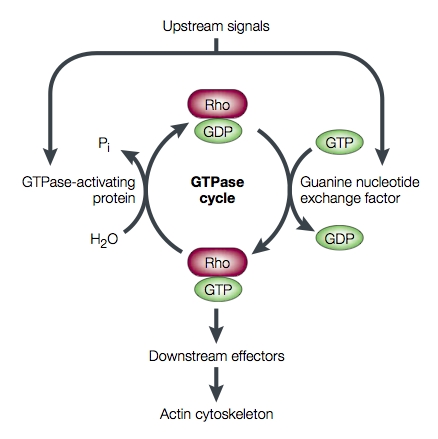
G protein activity is dependent on whether it is binding GTP or GDP. This useful property has led to the appropriation of G proteins by many cellular processes to be used as "molecular switches". G proteins are generally thought to be "active" when binding GTP and "inactive" when binding GDP. The transition from the GTP-bound state to the GDP-bound state depends on the hydrolysis of GTP. This GTPase activity is either completely intrinsic to the G protein or is enhanced by another class of proteins, "'''GTPase activating proteins'''" ('''GAP'''s). The GDP to GTP transition requires the dissociation of GDP, so that GTP may again bind at the active site. Proteins that mediate this GDP dissociation are known as '''guanine nucleotide exchange factors''' ('''GEF'''s). Figure 1 schematizes the switch mechanism for the Rho protein (Luo, 2000). [[Image:Luo_rho_schem.jpg]] | G protein activity is dependent on whether it is binding GTP or GDP. This useful property has led to the appropriation of G proteins by many cellular processes to be used as "molecular switches". G proteins are generally thought to be "active" when binding GTP and "inactive" when binding GDP. The transition from the GTP-bound state to the GDP-bound state depends on the hydrolysis of GTP. This GTPase activity is either completely intrinsic to the G protein or is enhanced by another class of proteins, "'''GTPase activating proteins'''" ('''GAP'''s). The GDP to GTP transition requires the dissociation of GDP, so that GTP may again bind at the active site. Proteins that mediate this GDP dissociation are known as '''guanine nucleotide exchange factors''' ('''GEF'''s). Figure 1 schematizes the switch mechanism for the Rho protein (Luo, 2000). <center>[[Image:Luo_rho_schem.jpg]]</center> | ||
Heterotrimeric G proteins are unique in that they exist as a complex (G<sub>αβγ</sub>) in the GDP-bound state but dissociate (into G<sub>α</sub> and G<sub>βγ</sub>) upon the release of GDP/binding of GTP. | Heterotrimeric G proteins are unique in that they exist as a complex (G<sub>αβγ</sub>) in the GDP-bound state but dissociate (into G<sub>α</sub> and G<sub>βγ</sub>) upon the release of GDP/binding of GTP. | ||
Revision as of 16:54, 23 October 2006
Introduction
The term G protein refers to proteins that bind the nucleotide guanine as guanosine triphosphate (GTP) and guanosine diphosphate (GDP). There are two types of G proteins: heterotrimeric, or large, G proteins and small G proteins. Heterotrimeric G proteins are membrane-associated and, along with G protein-coupled receptors, function primarily in cell signalling and signal transduction. Small GTP-binding proteins function in diverse cellular processes including signal transduction, cytoskeletal reorganization, and vescicle trafficking. The large small G protein superfamily includes the Ras family (signal transduction), the Rho/Rac family (cytoskeleton), the Rab and Sar1/Arf families (vescicle trafficking), and the Ran family (nuclear import/export) (Takai et al., 2001).
A molecular switch
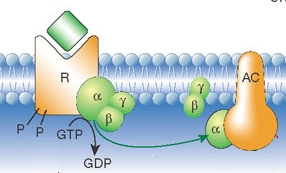
Heterotrimeric G proteins are unique in that they exist as a complex (Gαβγ) in the GDP-bound state but dissociate (into Gα and Gβγ) upon the release of GDP/binding of GTP.
G Protein-Coupled Receptors
References
1. Takai Y, Sasaki T, Matozaki T. Small GTP-Binding Proteins. Physiol Rev. 81, 153-208 (2001).
2. Luo L. Rho GTPases in neuronal morphogenesis Nat Rev Neurosci. 1, 173-180 (2000).
3. Firestein, S. How the olfactory system makes sense of scents. Nature 413, 211-218 (2001)