20.109(S13):Bacterial amplification of DNA (Day3)
Introduction
Assuming all went well, your reaction tubes from last time contain mutagenized DNA that encodes mutant inverse pericam. However, the desired DNA plasmid is likely present at a low concentration, and moreover it is nicked rather than in intact circular form. What we would like to do now is repair and further amplify only the mutagenized product. Thankfully, we have E. coli bacteria to do this for us quite efficiently!

Recall from Module 1 that bacteria can take up foreign DNA in a process called transformation. Today you will transform a cell strain engineered to be good at plasmid DNA amplification, using cells that are already competent. Next time you will transform a cell strain engineered to produce protein on demand, and prepare your own competent cells. Whether prepared by a company or by you, remember that competent cells are extremely fragile and should be handled gently, i.e., kept cold and not vortexed.
Bacterial transformation is efficient enough for most lab purposes, resulting in as many as 109 transformed cells per microgram of DNA, but even with highly competent cells only 1 DNA molecule in about 10,000 is successfully transformed. Moreover, the process is somewhat less efficient with a mutagenesis product rather than an intact plasmid. The vector carrying inverse pericam is called pRSET and also encodes a gene that leads to ampicillin-resistance; thus, our selection can be performed on ampicillin-containing agar plates. Given the low concentration and nicked structure of your DNA to begin with, you should perform your transformations today with great care.
Before setting up transformations, you will test your mutagenized DNA for the presence and approximate concentration of product via gel electrophoresis. Because the product is several Kbp long, a standard 1% agarose gel will serve us just fine. The long mutant plasmid DNA should be separated from the short digested fragments of parental DNA and thus can be identified. Note that the parental plasmid is originally present at a concentration too low to detect on a gel, and even the amplified mutant may show up as only a faint band. If you do not see any band at the expected size of the mutant plasmid, you might increase the amount of DNA used during the transformation procedure at the end of lab.
Protocols
Part 1: Agarose gel electrophoresis
Using a 1% agarose gel prepared by the teaching faculty, you will each run your digested reaction mixture. Half of you will also load either a reference lane containing standards of known molecular weight or a somewhat concentrated sample of wild-type plasmid. Divide up those responsibilities however you see fit. Recall that you should always handle all gels and gel equipment with nitrile gloves.
- In an eppendorf tube, combine 10 μL of of your DpnI-digested product with 2.5 μL of loading dye. Save the rest of the digested DNA, keeping it on ice.
- Remember to flick and quick-spin your sample, or pipet up and down to mix.
- Recall that loading dye contains xylene cyanol as a tracking dye and glycerol to help the samples sink into the well.
- Load the gel in the order shown in the table below, 11 μL per SDM sample and 10 μL per ladder lane.
- We will use a 14-lane rather than a 10-lane gel, to concentrate the DNA into a smaller area.
- To load, lower your sample-containing tip below the surface of the buffer and directly over the well. Expel your sample into the well. Do not release the pipet plunger until after you have removed the tip from the gel box.
- Once all the samples have been loaded, the teaching faculty will attach the gel box to the power supply and run the gel at 100 V for 45 minutes.
| Lane | Sample | Lane | Sample |
|---|---|---|---|
| 1 | Red group | 8 | Pink group |
| 2 | Orange group | 9 | DNA Ladder |
| 3 | DNA Ladder | 10 | Parent IPC |
| 4 | Parent IPC | 11 | Purple group |
| 5 | Yellow group | 12 | Platinum group |
| 6 | Green group | 13 | BLANK |
| 7 | Blue group | 14 | BLANK |
While the gel runs, we will have today's pre-lab lecture. During the remaining time, you can work on Parts 3 and (optional) 5, label the tubes you will need in Part 4, work on your notebooks, start the FNT assignment (it's a little long for a weekday assignment!), etc. Be sure to pre-chill your 14 mL tubes on ice for at least a few min before adding competent cells to them.
Part 2: Gel analysis
- The teaching faculty will photograph and post a digital image of the gel.
- In the following analysis, you will need the information for the 1 Kbp ladder you used, which is available at this link.
- First, see if you got a band at the expected size of the pRSET plasmid with an inverse pericam insert, or ~ 4 Kbp.
- If you did not get a band, you should use 2-3x the usual recommended DNA amount in your mutant transformation.
- If you did get a band, estimate the approximate amount of DNA in that lane in ng (by comparing to the ladder standards), then the concentration in ng/μL (based on the sample volume that you loaded). Write this information in your notebook.
Part 3: Prepare tubes for liquid O/N cultures
Please label 2 large glass test tubes with your team color and sample name in duplicate (X#Z-1, X#Z-2). Prepare a stock of LB culture broth with ampicillin (provided at 1000x concentration) and aliquot 2.5 mL of this mixture per tube. As in Module 1, these will be used to set up liquid overnight cultures from your two colonies for next time.
Part 4: Bacterial transformation
You will transform competent cells called XL1-Blue with your X#Z mutagenesis reactions and plate them on ampicillin-containing Petri dishes. Before next time, two candidate colonies will be chosen from each group's plate. The efficiency of this mutagenesis protocol is reported to be ~80%. We will test two candidates per mutation to cover our bases, so to speak.
- Get an aliquot of competent cells from one of the teaching faculty. Keep these cells on ice at all times, allowing them to thaw slowly (over a few minutes).
- Label three 14 mL polypropylene round-bottom tubes as follows: (-) control, (+) control, X#Z.
- The negative control will receive no DNA, but otherwise go through all of the following steps.
- In your case, the positive control is a reference mutant SDM reaction, specifically E67K. We could instead use a control SDM that comes with the QuickChange mutagenesis kit, but comparing to an IPC derivative is more meaningful. Be sure to use a fresh pipet tip when taking from the positive control stock DNA!
- Add 50 μL of competent cells to each tube, followed by 2 μL of the appropriate DNA. Gently swirl (do not vortex) to mix, then incubate on ice for 10 min.
- Bring the tubes over to the 42 °C water bath, and immerse them for exactly 45 seconds according to your digital timer.
- Immediately return the cells to ice for 2 minutes, and take an aliquot of pre-warmed LB medium.
- Add 0.5 mL of warm LB to each sample, then move them to the 37 °C incubator. Ask the teaching faculty to show you how to operate the roller and balance your tubes.
- Allow the cells to recover and begin expressing ampicillin resistance for 30 minutes. At the same time, pre-warm and dry three LB+AMP plates by placing them in the 37°C incubator, media side up with the lids ajar.
- Plate 250 μL of each transformation mix on LB+AMP plates. Safety reminder: After dipping the glass spreader in the ethanol jar, you should pass it through the flame of the alcohol burner just long enough to ignite the ethanol. After letting the ethanol burn off, the spreader may still be very hot, and it is advisable to tap it gently on a portion of the agar plate without cells in order to equilibrate it with the agar.
- Once the plates are ready, wrap them together with one piece of colored tape and incubate them in the 37°C incubator overnight. One of the teaching faculty will remove them from the incubator and set up liquid cultures for you to use next time.
Part 5: Statistics practice (optional)
You may find averages, standard deviations, and t-tests useful when you report on class results. (For your own unique mutant, however, you will only have one trial unless someone else has repeated that mutant.) You will also revisit these topics during Module 3.
You can practice the steps below using the male and female heights that we collected during pre-lab lecture.
- Begin by downloading the following Excel file as a framework to carry out the basic statistical manipulations we discussed in pre-lab lecture. The file is modified from one originally written by Professor Bevin Engelward.
- Find and plot 95% confidence intervals for the male and female heights.
- Compare the means of these two populations. At what confidence level (if any) are they different?
- Would a one-tailed or two-tailed test be more appropriate for this comparison?
For next time
The following assignment will prepare you to set up your digestion reactions next time. On the whole, Day 4 has a tendency to run long, so reading ahead and completing as many calculations as possible in advance will be of help in making the day run smoothly.
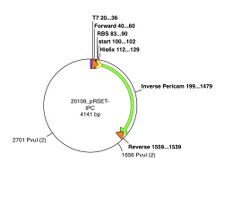
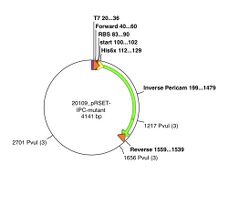
The pRSET plasmid with inverse pericam insert, or pRSET-IPC, is 4141 basepairs long and its GenBank file is linked here. According to the cutters list that you used on Day 1, restriction site PvuI occurs at ~1655 bp, and again at ~2700 bp into pRSET-IPC. Thus, digesting this parental plasmid with the PvuI enzyme should result in two linear fragments of DNA, with about 1045 and 3095 bp sizes.
A silent mutation can be introduced that results in a new PvuI site at the 341st-342nd residues of inverse pericam (ATT → ATC and TAC → GAC) , or approximately the 1020th basepair of IPC. When IPC is inserted into pRSET, its starting point is ~200bp into the pRSET plasmid. Thus, if the mutated pRSET-IPC plasmid is digested with PvuI, three linear fragments of DNA are the result: 440, 1045, and 2655 bp. To understand these calculations, see also the plasmid maps above. Make sure you can reproduce the numbers in this example before proceeding with your own samples. Plasmid maps can be made using the Enzyme Selector and Graphic Map functions in ApE.
For this assignment, you should plan restriction enzyme digests that allow you to distinguish parental and mutant pRSET-IPC for your X#Z mutation and for your reference mutant. You are probably best off doing a single enzyme digest for each mutant in this particular experiment. For example, above you would digest with PvuI. However, in other kinds of experiments (notably cloning) using two enzymes per digest can give more information. We will discuss these types of digests in a future pre-lab if there is time.
Use the tabulated information on the Day 1 Talk page along with the cutters lists from the Day 1 protocol to determine the cut sites relevant for M124S, T79P, or D24H as needed. For E67K, the students could not create an enzyme site on the 0- or 1-and-2-cutters lists. Instead, they ended up making a site for a 6-cutter (linked here). The banding pattern should nevertheless be distinguishable for the parent and mutant case, even though not all the bands will necessarily be visible.
Please clearly show all your work and reasoning throughout this assignment. Parts A through C should be completed for two different digests: X#Z and the reference mutant.
(A) First, use the NEB site to determine the appropriate buffer and temperature for your reaction. Searching for your enzyme by name is probably easiest.
(B) Next, you should explicitly show how the digest distinguishes between parent IPC and mutant IPC. In other words, what are the expected band sizes for each plasmid upon digestion?
(C) Finally, to prepare for Day 4 in lab, calculate the amount of enzyme and buffer needed for your digestion master mixes. Refer to Part 5 (first sub-part) of the Day 4 protocol.
Reagent list
- QuikChange II Site-Directed Mutagenesis Kit from Stratagene
- XL1-Blue supercompetent cells
- LB (Luria-Bertani broth)
- 1% Tryptone
- 0.5% Yeast Extract
- 1% NaCl
- autoclaved for sterility
- Ampicillin: 100 mg/mL, aqueous, sterile-filtered
- LB+AMP plates
- LB with 2% agar and 100 μg/ml Ampicillin