20.109(F13): Mod 2 Day 3 Prepare for LTS
Introduction
The human cell line that we are working with in Module 2 originated in 1973 from a 64-year old woman with ovarian cancer. Specifically, the cells were isolated from malignant ascites which collect in the peritoneal cavity of patients with advanced disease. The 20.109 instructional staff obtained your SKOV3 cells from ATCC, a repository started in 1925 to provide a source of common reagents for scientists. If you investigate the SKOV3 product page, you'll notice that these cells have been thoroughly characterized by ATCC for chromosomal abnormalities and for contamination by the common blood borne pathogens that were discussed by our EHS officer.
Protocol
Part 1: Prepare SKOV3 cells for Western blot analysis
Today you will seed SKOV3 cells for your Western blot analysis. Many parts of the protocol below will seem familiar to you, but each type of cell requires slightly different sub-culturing conditions, so please read the protocol all the way through before starting. You and your partner will utilize a seeding density of 21,000 cells / cm2 to seed the wells of one 6-well tissue culture plate. Seeding (or plating) density is the most flexible way to report your initial cell density when reporting your experimental methods. For your reference, the total cell growth surface area of a single well in a 6-well plate is 9.5 cm2.
Let's get started:
- Calculate the total number of cells that you will need for your 6-well plate.
- Remove one 10 cm2 plate of SKOV3 cells from the TC incubator.
- Take a look at the plate under the microscope -- notice how these epithelial cells look different from your mouse embryonic stem cells from Module 1.
- Remove the growth media and rinse the cells 1x with 7 mL of PBS.
- Add 1.5 mL of Trypsin/EDTA to the plate and put back in the incubator.
- After 7 min, check if the cells have lifted from the surface of the plate.
- If the cells are mostly floating in the trypsin, use the ball of your hand and gently rap the plate to dislodge the remaining cells.
- If more than 50% of the cells remain on the plate, place it back in the incubator for an additional 3 min.
- Next, add 8.5 mL of McCoy's 5A growth medium to the plate and triturate well to mix the cells and collect in a 15 mL conical.
- Remove 18 μL of cells into an eppendorf tube for counting.
- Outside of the TC hood, add 2 μL of Trypan blue to your eppendorf tube and mix well.
- Count the cells using the hemocytometer. Calculate the number of viable cells that you have.
- Now, add the number of cells that you need for seeding your 6-well plate to a new 15 mL conical. Use the table below to help you.
- Spin the cells at 500xg in the TC centrifuge for 5 min.
| [Total Cells] | [Dead Cells] | [Viable Cells] | Total Numb. of Cells Needed | Total Numb. of Viable Cells | Volume of Cell Suspension Needed |
|---|---|---|---|---|---|
| \ | \ | \ | \ | \ | \ |
After the cells have been pelleted to remove the residual trypsin, seed the cells:
- Remove the supernatant from the cells -- be careful not to aspirate your cell pellet!
- Resuspend the cells in 12 mL of growth media -- mix the cells very well!.
- Add 2 mL of cell suspension to each well of the 6-well plate.
- Use the squeaky technique taught in pre-lab to distribute the cells evenly in the plate.
- Place the cells in the TC incubator.
- The teaching staff will replace the media with fresh growth media two days after seeding.
Part 2: Mutant sequence analysis
Our analysis will proceed in three steps. First, you will align your sequencing output to the known exon 19 and 21 sequences and determine any mismatch locations. Second, you will closely examine the trace files to determine if there is a mixture of mutant alleles present. You can compare your exon 21 results to our MDA-MB231 negative control and your exon 19 results to the positive control HCC-827 sequencing reactions that you set-up. Finally, you will translate your gene sequence to an amino acid sequence.
Retrieve your sequencing data
- Log in to GENEWIZ using the user name: skalford AT mit DOT edu and the pswd: be20109
- View or download the sequence files that are associated with your mutants. These should open up as either a text file, which you can copy into the ApE program that's on your laptop or as an ApE file directly. If there were ambiguous areas of your sequencing results, these will be listed as "N" rather than "A" "T" "G" or "C." It's fine to include Ns in the steps listed below.
- Be sure to name your sequence files something more meaningful than the default "New DNA" so that you don't get mixed up with all of the sequencing data that you'll have!
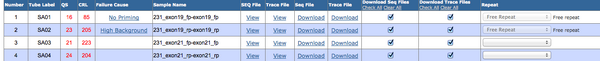
Below is a screen capture of the Genewiz website to illustrate the user interface.
Align Sequences
Next, align your DNA sequences to the EGFR exons with "bl2seq" from NCBI
BLAST is an acronym for Basic Local Alignment Search Tool, and can be accessed for free through the National Center for Biotechnology Information (NCBI) home page
- Paste one of your sequences into the "Sequence 1" box at the BLAST2 sequences site. The alignment program can be accessed through the NCBI BLAST page or from this link.
- Paste either the EGFR exon 19 or exon 21 sequence from here into the "Sequence 2" box. Make sure to match the primer set you used to the correct exon.
- Align the sequences. Matches will be shown by lines between the aligned sequences.
- Save a screenshot of the relevant alignment (using shift/command/4 or the Grab program under utilities), and draw conclusions about the alignment in your notebook. You might want to paste the alignment screen shot in your Evernote notebook.
- In addition to aligning the sequences of the exon 19 and exon 21 reactions that you performed, you should also align the MDA-MB231 cell exon 21 sequences. They are included on the third page of the file you downloaded above with the exon 21 and exon 19 sequences. These alignments will serve as your negative control, as MDA-MB231 cells contain wild type EGFR.
- Make a note of any mismatches -- it is helpful to write down the base pair number so that you can easily find it on the sequencing trace file.
Some of the mismatches that you find will be noise and due to a low quality sequencing reaction. However, if you find a mismatch (or missing sequence) in the commonly mutated areas of EGFR (exon 19 aa746-750 deletion or exon 21 L858R) you may have identified a mutant.
Examine Trace Files
This part of the analysis is more subjective than the sequence alignment. Here you will examine the sequencing trace files within regions of exon 19 and exon 21 that are most likely to contain mutations. You'll find that sequencing reactions are rarely perfect, and background noise will likely be present. Therefore, you will have to determine, based on the level of background noise within your negative control, the banding pattern on your agarose electrophoresis gel, and using what you know about ovarian cancer, if your SKOV3 cells harbor mutations within EGFR.
Start with the EGFR exon 21 trace files for your negative control -- MDA-MB231 cells (forward here and reverse here). Examine the trace files for a L858R mutation. Figure 3 contains the translated sequence from the MDA-MB231 cells. A L858R mutation would replace a T (highlighted in the figure) with a G.
Next, download the trace files for the SKOV3 exon 21 sequencing reactions. Using your trace files (and the information from your sequence alignment above) determine if there are any possible mutations within exon 21. Concentrate on the most common mutation -- L858R. But you may also look back at the M2D2 lecture on slide 19 for other potential mutation sites if you have time or interest.
Examination of the exon 19 traces is much trickier. The clean traces that were shown in class are ideal data and not necessarily what you find in the lab. Download this exon 19 trace from our positive control cell line HCC-827. Remember that these cells are heterozygous for the aa746-750 deletion in exon 19. Figure 4 shows the DNA and amino acid sequence of exon 19 with and without the deletion -- you can use this to search for the deletion region and examine the traces. You should be able to identify a deletion within this trace file, please ask the teaching faculty if you would like some help finding it.
Remember to take a look at the agarose gel electrophoresis on the Talk page that illustrates the number of bands that were amplified during our COLD-PCR reaction. Are your sequencing results consistent with the gel image? In other words, if there are several bands, would you expect a clean DNA sequencing reactions (similar to the reactions completed using the MDA-MB231 cells) or might you expect these results to be difficult to interpret? Think about how you might optimize the experiment if that is the case -- and keep those notes/thoughts fresh in your mind for your module 2 report.
Translate sequence
If you find through your analysis above that the SKOV3 cells harbor a mutation, the next step is to determine what affect that nucleotide change will have on the amino acid sequence. Shannon's favorite translation tool is from Switzerland, but if you have a favorite of your own feel free to use it. ExPASy's translate tool can be found at this link. If you'd like to translate your sequence in the format shown in Figures 3 & 4, choose the includes nucleotide sequence option. Make sure to choose the correct reading frame! (Hint: it is unlikely that you sequence will include any stop codons, so that is one handy way to narrow down the choice of reading frame.)
Part 3: Journal article discussion
We suggest reading the paper in this order:
- Introduction
- Results: Computational and experimental modeling of ErbB2/3-driven tumor growth (Figure 1)
- Materials & Methods: Cell lines and reagents
- Materials & Methods: Multiplex Luminex protein assays (Figure S1)
- Materials & Methods: Computational methods -- Approach and quantitative logic
- Materials & Methods: Simplifying assumptions (Figures S9, S10)
- [Materials & Methods: the rest of the model description]**
- Results: Calibration of ErbB-mediated signaling…. (Figure 2A), (Figures S2, 2B), (Figure S3)***
- Results: Regulation of tumor growth by PI3K and… (Figure 3A, S6, 3B) (Figure 3C)
- Results: In vivo translation: Assessing drug combination…(Figure 4A, 4B) (Figure 4C, D)
- Discussion (We will not discuss Figures 5 - 8 in class)
While it is not necessary for you to completely understand the model that has been built, it is good practice to read these sections and it will help you to follow the rest of the text.** These figures are tricky. Please see Shannon with questions and do your best to make an explanatory slide.***
Figure 1
Team:
Read the first subsection under Results, titled "Computational and experimental modeling of ErbB2/3-driven tumor growth", and the "Computational methods -- Approach and quantitative logic" subsection in the Materials and Methods (which is located at the end of the paper). Answer the following questions to guide you:
- (1) What is the system being modeled and what specific pathways are represented?
- (2) What do the authors state is the key insight to required to accurately related clinical response to cell stimulation?
- (3) How do the authors take advantage of this insight to simplify their model -- what is the name of the modeling technique that they used?
- (4) Under simplifying assumptions -- what is the most important assumption included in the model construction?
Figure S1
Team:
Read the first paragraph under the Results subsection "Calibration of ErB-mediated signal transduction and feedback regulatory circuits". Answer the following questions to guide you:
- (1) How are luminex-based total and phosphoprotein measurements completed? A good read of the Method section covering this assay and a Google search will help you make a good slide.
- (2) How is a z-score calculated?
- (3) Why is this an important control experiment?
Figure S9 & S10A
Team:
These figures are referenced in the Material & Methods: Simplifying assumptions section. Answer the following questions to guide you:
- (1) How do MM-111 and Lapatinib work? (You may need to read ahead to the Ligand-receptor interactions section to find this).
- (2) How were the experiments performed? Where did you find that information? (If you can't, how do you feel about that? Can you find enough information to provide an explanation to the class)
- (3) How would the model be changed if the assumptions experimentally verified in S9 & S10 did not work out?
Figure 2A***
Team:
Read the Results subsection titled Calibration of ErbB-mediated signaling….and the Materials & Methods sections: Approach and quantitative logic, Simplifying assumptions, Ligand-receptor interactions, Intracellular signal transduction and Transcriptional feedback regulation. Think about the following points/questions to guide you:
- (1) How do MM-111 and Lapatinib work?
- (2) Draw a box, label the corners with (low, low), (low, high), (high, low), and (high, high) according to the inhibitor concentrations. Use this to orient yourself while interpreting the figure.
- (3) Remember the following: High MM-111 = low Erb3 (E3) activity, High Lapatinib = low ErbB2 activity (E2).
- (4) What experiments were used to populate the left hand column of response surfaces?
- (5) How did the authors make the right hand column?
Figure S2, 2B***
Team:
Read the Results subsection titled Calibration of ErbB-mediated signaling….and the Materials & Methods sections: Approach and quantitative logic, Simplifying assumptions, Ligand-receptor interactions, Intracellular signal transduction and Transcriptional feedback regulation. Think about the following points/questions to guide you:
- (1) What connections in Figure 1 best describe the data in Figure S2?
- (2) If a phosphorylated Akt leads to decreased ErbB3 levels (Figure S2) and activated ErbB3 leads to significantly more phosphorylated Akt than activated ErbB2 (see Figure 2A, middle response surface on left side) – what should happen to ErbB3 levels if Akt phosphorylation is decreased due to MM-111 treatment?
- (3) Think about revisiting Fig1 or drawing your own feedback loop.
Figure S3***
Team:
Read the Results subsection titled Calibration of ErbB-mediated signaling….and the Materials & Methods sections: Approach and quantitative logic, Simplifying assumptions, Ligand-receptor interactions, Intracellular signal transduction and Transcriptional feedback regulation. Think about the following points/questions to guide you:
- (1) If a phosphorylated Akt leads to decreased ErbB3 levels (Figure S2) and activated ErbB3 leads to significantly more phosphorylated Akt than activated ErbB2 (see Figure 2A, middle response surface on left side) – what should happen to ErbB3 levels if Akt phosphorylation is decreased due to MM-111 treatment?
- (2) Think about revisiting Fig1 or drawing your own feedback loop.
- (3) Do agree with the authors that their ErbB3 feedback loop is real evidenced by 50% of the cancer cell lines showing some signs of it?
- (4) Is this figure easy to interpret? How did they obtain the data? How would you improve the visual ease?
Figure 3A, S6, & 3B
Team:
Read the Results subsection titled "Regulation of tumor growth by PI3K and MAPK pathway integration". Consider the following to guide you:
- (1) How is the timescale of the experiment in Fig 3A different from Fig 2A? How do the authors use that to motivate their logic-based modeling technique? What is a logic gate?
- (2) What do the authors do to verify that their final model (Fig 3B) is the best one?
- (3) According to figure 3B, what two things contribute to tumor growth and (according to the Materials & Methods: Ligand-receptor interactions section) what is the driver in this cell line?
Figure 3C
Team:
Read the Results subsection titled "Regulation of tumor growth by PI3K and MAPK pathway integration". Answer the following to guide you:
- (1) What do MM-111 and Lapatinib target? (You may need to read the Materials & Methods:Ligand-receptor interactions section to find this).
- (2) What do UO126 and MK2206 target?
- (3) What data was used to 'train' the logic model? How would you use the logic model to predict the effect of MMM-111 and Lapatinib?
Figure 4A, 4B
Team:
Read the Results subsection titled "In vivo translation:…" Answer the following to guide you:
- (A) What is the major modeling assumption the authors are making when going from in vitro to in vivo?
- (B) What is a syngeneic cell line? What is a xenograft?
- (C) Why was it required to estimate the rates of proliferation, cell death, and [HRG] before utilizing the logic model parameterized using in vitro data to predict tumor growth in vivo? A visit to the Materials & Methods: Simplifying assumptions section might help here
- (D) Would you consider the logical model approach successful here?
For next time
- Next time you will perform an assay to measure total protein concentration in your cell lysate. The reagent that we will use absorbs light at 600 nm, and the amount of protein present in the lysate will be calculated as:
- protein concentration (μg/mL) = 125 * A600 * 30
- where 30 = the dilution factor used to prepare the assay
- and 125 = the conversion factor from absorbance A to concentration
- To save time during the next lab, you should prepare an Excel spreadsheet that will automatically calculate the volume of your lysate needed to provide 30 μg of total protein.
- The variable input to the sheet will be your measured A600 value.
- Your sheet will be most versatile and easy to follow if each element (such as the dilution factor) is listed and labeled separately, including units.
- The first output of your sheet will be lysate volume. Consider that you will probably want to convert the volume to μL right away.
- When preparing your SDS-PAGE analysis, you may only load a total volume of 15 μL on the gel. Therefore, your spreadsheet should also calculate the amount of water required to adjust the lysate volume to 15 μL.
- Finally, just to be safe (in case the protein concentration is low) -- repeat your calculations finding the volumes of lysate and water required to use 20 μg of total protein in a diluted volume of 15 μL.
- Try doing your calculations with an A600 of 0.71. If you'd like to check your answer in advance, just email Shannon (skalford at mit dot edu) and she'll let you know if your spreadsheet works.
- protein concentration (μg/mL) = 125 * A600 * 30
- Please download the midsemester evaluation form found here (DOC download). Complete the questionnaire and then print it out without including your name to turn in. If there is something you'd like to see done differently for the rest of the course, this is your chance to lobby for that change. Similarly, if there is something you think the class has to keep doing, let us know that too. These will be collected in a separate folder from your FNTs, to preserve anonymity.
- The major assessment for this module will be a research article describing your Systems Biology study. For this assignment, you will start outlining and drafting the introduction to your report. Recall that the introduction provides a framework for the story you are about to tell, with respect to both background and motivation. You are encouraged to revisit the scientific writing guidelines before you begin.
- What might the big picture section consist of? Will you focus on systems biology in general, the use of high throughput screening technologies, mechanisms of drug resistance, or ... what?
- What background would the reader like to have as you zoom in?
- When you define your specific study, be sure to share your hypothesis about the affect of the inhibitor you chose. You may also choose to speculate about how your inhibitor will perform in relation to the others used by the class.
At a minimum, you should hand in some high-level ideas and supporting notes for the first two sections, and write a draft of the third section in complete sentences.
Reagents list
- 0.4% Trypan blue
- 0.25% (0.05%?)Trypsin/ 1 mM (0.5 mM?) EDTA
- SKOV-3 growth medium
- McCoys 5A[is this the base?]
- 10% FBS (Atlanta Biologicals)
- 1% Glutamine from 100X stock (0.3 mg/mL final concentration)
- 1% Pen-Strep from 100X stock (100 U/mL penicillin, 100 μg/mL streptomycin)
- 1% Non-essential Amino Acids from 100X stock (varying amounts of 7 AA)
Navigation Links
Next Day: Mod 2 Day 4: Phosphotyrosine Western Blot Analysis
Previous Day: Mod 2 Day 2: Mutation Analysis