Biomod/2013/Harvard/results
<html>
<head>
<link href='http://fonts.googleapis.com/css?family=Open+Sans' rel='stylesheet' type='text/css'>
</head>
<style>
body {
font-family: 'Open Sans', sans-serif; overflow-y: scroll;
}
.container {
background-color: #ffffff; margin-top:0px
} .OWWNBcpCurrentDateFilled { display: none; }
h1 {
font-size: 36px; line-height: 36px; padding-top: 5px; border-bottom-width: 0;
}
h3 {
font-size: 18px;
}
h5
{
font-family: 'Open Sans', sans-serif; font-size: 11px; font-style: normal; text-align: center; margin:0px; padding:0px;
}
- column-content
{
/* Uncomment to Dewikify width: 0px; float: left; */ margin: 0 0 0 0; padding: 0;
} .firstHeading {
display:none; width:0px;
}
- column-one
{
display:none; width:0px; padding-top: 35px; background-color: #ffffff;
}
- globalWrapper
{
width: 900px; background-color: #ffffff; margin-left: auto; margin-right: auto
}
- content
{
margin-left: 0px; margin-top: 0px; padding-top: 0px; align: center; /*padding: 12px 12px 12px 12px; width: 30%; background-color: #ffffff; border: 0; */
}
- bodyContent
{
width: 850px; align: center; background-color: #fffffff;
}
- column-content
{
width: 900px; background-color: #ffffff;
}
- footer
{
position: center; width: 900px;
} @media screen {
body { background: #000000 0 0 no-repeat; /* changed default background */ }
}
- menu
{
align: left; width: 10em; padding: 0px 10px 10px 10px; background-color: #FFFFFF; float: left;
}
- pagecontent
{
width: 620px; min-height: 400px; float: left; margin-left: 0px;
}
.group:after {
content: ""; display: table; clear: both;
}
.editsection {
/*display: none*/
}
a:link {color:#FF6060;}
a:active {color:#B24343; }
a:hover {color:#B24343; text-decoration: none}
a:visited {color:#FF6060;} /* visited link */
/*Expanding list*/
- exp { list-style: none; }
- exp li {
height: 1.8em; border-top: 1px solid #dedede; margin: 0 0 0 0; padding-top: .2em
}
- exp li:hover { background-color: #F8F8F8}
- exp li a:hover { display: block }
</style> </html>
Results
Input
We were able to achieve all three of our goals.
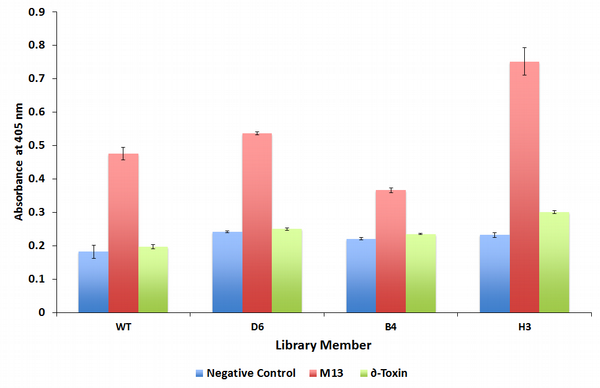
One round of directed evolution produced one library member with improved affinity to δ-toxin. Variant H3 of BlaCaM saw a marked increase in δ-toxin activity over the negative control (no peptide) (Figure 1). While the improved activity is not nearly as high as the m13 activity, this variant appears to have begun mutating in the correct direction to improve binding. For variant H3, activity with both m13 and δ-toxin has increased, suggesting that any improvements to δ-toxin binding are not specific to δ-toxin. This evolution of "promiscuity," where a protein expands the number of targets it can bind to, is seen as a crucial intermediate step in directed evolution of novel protein targets (Bloom JD, Arnold FH. 2009. PNAS. 106 (Supplement 1) 9995-10000).
Sequencing of variant H3 revealed 4 mutations to the calmodulin region of the encoding gene. One of these mutations, F19L, altered the large hydrophobic residue phenylalanine to a smaller, still hydrophobic leucine. Residue 19 sits directly in the peptide binding area of calmodulin, indicating that this mutation, and this specific residue, could be very important for determining calmodulin binding specificity.
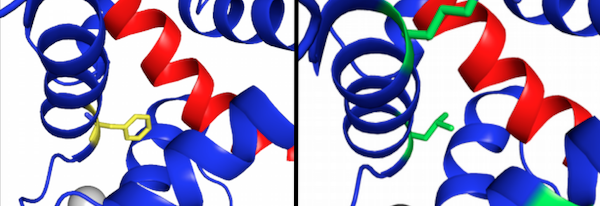
While random directed evolution will eventually produce positive results, knowledge of important regions or residues for peptide binding in CaM can greatly narrow the mutation space that must be explored to develop improvements. Identifying key residues like residue 19 will help direct further evolution and accelerate the process developing binding to δ-toxin. Through directed evolution, we improved the binding of BlaCaM to δ-toxin and identified a possible key residue for peptide specificity and affinity.
Output
We achieved the first two out of the three goals. We successfully expressed GLucCaM in E.coli and purified the protein, and GLucCaM retained its GLuc activity; furthermore, we explored various structure-function relationship of GLucCaM, and the two major results were the effects of cysteine mutations and linker variations.
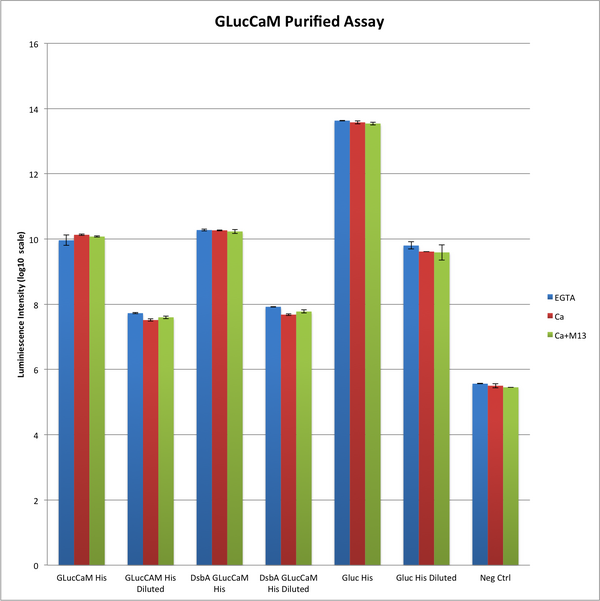
GLuc activity retension
The following graph illustrates the activity of GLucCaM with the substrate coelentarazine measured by the luminescence level. The three conditions are EGTA (which strips away Ca(2+) that might be bound to CaM), Ca(2+), and Ca(2+) with M13. The negative control was expression sample from E.coli that did not contain our expression vector. We can see that the activity of GLucCaM His (His for Hisx6 tag) was substantial higher than negative control and matched the activity of diluted GLuc expression.
Cysteine Mutations
GLucCaM contains ten cysteine residues. Since our protein could not be deactivated with calcium, we considered the possibility of disulfide bonds forming across the protein, preventing conformation changes. Unfortunately, we do not know the specific amino acids responsible for the disulfide bonds, so we selected a reasonable set of four cysteine residues highlighted in yellow below. The light blue indicates the two halves of GLuc, the red indicates calmodulin, and the green regions indicates the two linkers.
We mutated out various combinations of the four cysteine residues to alanine, but these mutated GLucCaM showed no activity. This suggests that these cysteine residues are essential to the structure of GLucCaM.
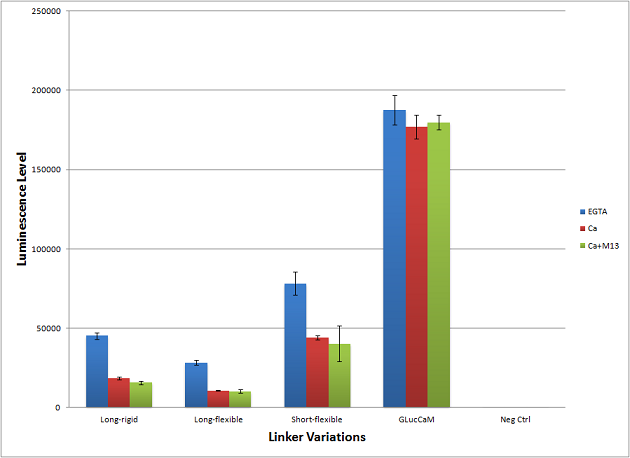
Linker Variations
Another possibility we explored was varying the linker length and rigidity relative to the GLucCaM linkers. The original GLucCam structure has amino acid strands GGGGT and GGGS as the first and second linker sequences. We explored the following linker variations:
Short-rigid: GPG GPG
Long-rigid: APAPAP APAPAP
Long-flexible: GGGGSGGGGS GGGGSGGGGS
All linker variations showed a decrease in luminescence level, but they all exhibited significant decrease in activity when exposed to high concentration of calcium. Unfortunately, the protein switch does not respond to increased levels of M13, but this is a promising start for continued modification to the output domain.
Raw Data
Raw data can be found at this link.