Biomod/2012/Potsdam/DnanoPROT/Project
<html> <head> <style type="text/css">
html,body {height:100%;margin:0;padding:0;}
body {
/*background-image: url(http://openwetware.org/images/e/ee/UP12_background.jpg); background-repeat:repeat-y repeat-x; */
} .gradient_golden {
-ms-filter: "progid:DXImageTransform.Microsoft.gradient(startColorstr=#0e8cd0, endColorstr=#104871)"; background-image: -ms-linear-gradient(top, #0e8cd0 0%, #104871 100%); background-image: -moz-linear-gradient(top, #0e8cd0 0%, #104871 100%); background-image: -o-linear-gradient(top, #0e8cd0 0%, #104871 100%); background-image: -webkit-gradient(linear, left top, left bottom, color-stop(0, #0e8cd0), color-stop(1, #104871)); background-image: -webkit-linear-gradient(top, #0e8cd0 0%, #104871 100%); background-image: linear-gradient(to bottom, #0e8cd0 0%, #104871 100%);
}
- sidebar-main{
visibility:hidden;
}
- content {
left: 50%; margin-left: -480px; width: 975px;
}
- igem_home {
font-size: 95%; left: 425px; position: absolute; top: -4px;
}
.gradient_grey{
-ms-filter: "progid:DXImageTransform.Microsoft.gradient(startColorstr=#FFFFFF, endColorstr=#E6E6E6)"; background-image: -ms-linear-gradient(top, #FFFFFF 0%, #E6E6E6 100%); background-image: -moz-linear-gradient(top, #FFFFFF 0%, #E6E6E6 100%); background-image: -o-linear-gradient(top, #FFFFFF 0%, #E6E6E6 100%); background-image: -webkit-gradient(linear, left top, left bottom, color-stop(0, #FFFFFF), color-stop(1, #E6E6E6)); background-image: -webkit-linear-gradient(top, #FFFFFF 0%, #E6E6E6 100%); background-image: linear-gradient(to bottom, #FFFFFF 0%, #E6E6E6 100%);
}
.white_bg {
background-color: white;
}
.box_round {
width: 955px; padding: 5px; border-radius: 5px; box-shadow:5px 5px 5px #666;
}
.box_round p {
text-align:justify;
}
.no_padding {
padding: 0px;
}
.nav_menu {
width: 965px; height: 35px; box-shadow:5px 5px 5px #666;
}
- menu .selected {
background-color:#B41111; height: 27px; border-top-right-radius: 5px; border-top-left-radius: 5px;
}
- menu {
display: block; margin: 0px; padding: 8px; position:relative; left: 50%; text-align: center;
}
- menu ul {
margin: 0px;
}
- menu li {
list-style: none outside none; float:left; padding-left: 15px; padding-right: 15px; margin-right:25px; margin-left:25px; font-weight: 900;
}
- menu li ul{
list-style: none outside none; display:none; position: absolute; margin : 0px; padding: 0px;
}
- menu ul ul {
padding: 0px;
}
- menu li ul li{
margin : 0px; width:inherit; font-weight: normal;
}
- menu li ul li:hover {
height:auto; float:none; text-align:left;
}
- menu ul li {
clear: both;
}
- menu a {
display: block; color: white;
}
- menu a:hover {
text-decoration:none;
}
- menu li:hover {
background-color:#B41111; height: 27px; border-top-right-radius: 5px; border-top-left-radius: 5px;
}
- menu li:hover ul {
display:block;
}
.box_round table {
background-color: transparent;
}
.block {
display: inline-block; width : 350px; height : 200px;
}
table {
background-color: transparent;
}
table th {
padding-left: 5px; padding-right: 5px;
}
- content {
border: none; background-color: transparent;
}
- catlinks {
border:none; background-color: transparent;
}
- footer-box {
border:none; background-color: transparent;
}
- footer {
background-color: transparent; border: none;
} .center_box {
text-align: center;
}
- p-logo {
display:none;
}
- top-section {
border: none; height:0px;
}
.firstHeading{
display: none; padding:0px;
}
- banner-wrap {
padding: 0px;
}
- portfolio {
z-index: -200; height: 280px;
}
- portfolio img{
box-shadow:5px 5px 5px #666; width:965px; height: 280px;
}
.rollover {
width: 975px; text-align: center;
}
.rollover a{
display: inline-block; width : 200px; margin: 20px; height : 200px; border-radius : 12px; margin: 20px 20px 20px 20px; box-shadow:5px 5px 5px #666;
}
- rollover-image {
background-image:url(http://openwetware.org/images/7/77/UP12_Video1.jpg); background-position: -0px -0px;
}
- rollover-image:hover {
background-image:url(http://openwetware.org/images/4/46/UP12_Video2.jpg); background-position: 0px -0px;
}
- rollover-image-2 {
background-image:url(http://openwetware.org/images/6/6d/Project1.jpg); background-position: 200px -0px;
}
- rollover-image-2:hover {
background-image:url(http://openwetware.org/images/d/d1/Project2.jpg); background-position: 200px -0px;
}
- rollover-image-3 {
background-image:url(http://openwetware.org/images/9/94/UP12_Lab1.jpg); background-position: 400px -0px;
}
- rollover-image-3:hover {
background-image:url(http://openwetware.org/images/5/58/Up12_Lab2.jpg); background-position: 400px -000px;
}
- rollover-image-8 {
background-image:url(http://openwetware.org/images/c/cf/UP12_Team1.jpg); background-position: 600px -0px;
}
- rollover-image-8:hover {
background-image:url(http://openwetware.org/images/5/5c/UP12_Team2.jpg); */ background-position: 600px -0px;
}
- totop {
/* background-image:url(http://2012.igem.org/wiki/images/0/0d/Pfeil.png); */ filter:alpha(opacity=50); height: 76px; width: 75px; position: fixed; right: 10px; z-index: -20;
}
- search-controls {
width: auto; top: -4px; right: 200px; z-index: 15;
}
input.searchButton {
color: #ffffff;
}
- menu_center {
float:right; position:relative; left:-50%; text-align:left;
}
input.searchButton:hover {
color: #B41111;
}
</style> </head> </html> <html> <head> <script src="//ajax.googleapis.com/ajax/libs/jquery/1.8.2/jquery.min.js"></script> <script type="text/javascript"> (function($) {
$.fn.innerfade = function(options) {
return this.each(function() {
$.innerfade(this, options);
});
};
$.innerfade = function(container, options) {
var settings = {
'animationtype': 'fade',
'speed': 'normal',
'type': 'sequence',
'timeout': 1000,
'containerheight': 'auto',
'runningclass': 'innerfade',
'children': null
};
if (options)
$.extend(settings, options);
if (settings.children === null)
var elements = $(container).children();
else
var elements = $(container).children(settings.children);
if (elements.length > 1) {
$(container).css('position', 'relative').css('height', settings.containerheight).addClass(settings.runningclass);
for (var i = 0; i < elements.length; i++) {
$(elements[i]).css('z-index', String(elements.length-i)).css('position', 'absolute').hide();
};
if (settings.type == "sequence") {
setTimeout(function() {
$.innerfade.next(elements, settings, 1, 0);
}, settings.timeout);
$(elements[0]).show();
} else if (settings.type == "random") {
var last = Math.floor ( Math.random () * ( elements.length ) );
setTimeout(function() {
do {
current = Math.floor ( Math.random ( ) * ( elements.length ) ); } while (last == current ); $.innerfade.next(elements, settings, current, last);
}, settings.timeout);
$(elements[last]).show();
} else if ( settings.type == 'random_start' ) { settings.type = 'sequence'; var current = Math.floor ( Math.random () * ( elements.length ) ); setTimeout(function(){ $.innerfade.next(elements, settings, (current + 1) % elements.length, current); }, settings.timeout); $(elements[current]).show(); } else { alert('Innerfade-Type must either be \'sequence\', \'random\' or \'random_start\); } }
};
$.innerfade.next = function(elements, settings, current, last) {
if (settings.animationtype == 'slide') {
$(elements[last]).slideUp(settings.speed);
$(elements[current]).slideDown(settings.speed);
} else if (settings.animationtype == 'fade') {
$(elements[last]).fadeOut(settings.speed);
$(elements[current]).fadeIn(settings.speed, function() {
removeFilter($(this)[0]); });
} else
alert('Innerfade-animationtype must either be \'slide\' or \'fade\);
if (settings.type == "sequence") {
if ((current + 1) < elements.length) {
current = current + 1;
last = current - 1;
} else {
current = 0;
last = elements.length - 1;
}
} else if (settings.type == "random") {
last = current;
while (current == last)
current = Math.floor(Math.random() * elements.length);
} else
alert('Innerfade-Type must either be \'sequence\', \'random\' or \'random_start\);
setTimeout((function() {
$.innerfade.next(elements, settings, current, last);
}), settings.timeout);
};
})(jQuery);
// **** remove Opacity-Filter in ie **** function removeFilter(element) { if(element.style.removeAttribute){ element.style.removeAttribute('filter'); } }
$(document).ready( function(){
$('#portfolio').innerfade({ speed: 'slow', timeout: 8000, type: 'sequence', containerheight: '280px' });
$('.fade').innerfade({ speed: 'slow', timeout: 1000, type: 'sequence', containerheight: '1.5em' }); } );
</script>
</head>
</html>
<html>
</html> <html>
</html>
Project Results
Biotinylated DNA-Origami with anti-biotin-antibody generates two dimensional patterns
The basis for the triangular DNA-origami is the 'sharp triangle composed of trapezoidal domains' as published in the supplement of the 2006 nature article by Paul Rothemund. To generate DNA-origami displaying the chemical compound biotin, oligos were either modified within the sequence ( e.g. bridging oligo t-11s8e_t-12s29 TCAAGATTAGT_T_GTAGCAATACT was modified at the middle T) or a A20 sequence was added with a two nucleotide spacer to the desired staple oligonucelotides and a T20oligonucleotide coupled with biotin was added. The latter approach is more flexible with respect to different modifications and was preferred. The origami were folded and purified with the biotin attached, and then the monoclonal anti-biotin antibody was added such that the number of binding sites of the antibody approximately match the number of binding sites on the origami. Fig. 1 and Fig. 2 show the results of such antibody-mediated DNA origami cross-linking.
Recombinant virus particle with penta Gly sortase motif
To insert the N-terminal sortase motif into the viral capsid we designed a plasmid required for viral assembly. The BioBrick pCMV_DARPin-E01_Middle-Linker_[AAV2]-VP23 was used to get the VP2 cap-gene. PCR was made with self-designed primers. The forward primer contains the Kozak sequence followed by sortase motif Pentaglycin and myc-tag. The Kozak sequence was inserted to improve the expression and myc-tag to recognize that the sortase motif is on the surface of the virus. The reversed primer was designed to enclose the whole VP2 region. For the following digestion the restriction sites for XbaI and PstI were also presented in the primers. The desirable part was ligated into the plasmid containing the CMV-promoter.
To get the completely assembled recombinant adeno-associated virus with the N-terminal sortase motif on the surface and CFP as gene of interest, we transfected the AAV-293 cells with four plasmids:
- the sequenced plasmid with sortase-motif fused to VP2
- the rep-cap-plasmid containing the VP2 knock-out
- the helperplasmid
- plasmid containing the gene of interest. As gene of interest we used CFP-gene which was packaged into the capsid.
into the HEK293 cells. To check whether the viral assembly worked we infected the HT1080 cells with recombinant adeno-associated virus, like it was described in the AAV Helper-Free System Instruction Manual. Few days after infection of HT1080 cells were gleaming cyan by the excitation.

EGFR ecotodomain-3 with LPXTG Sortase motif
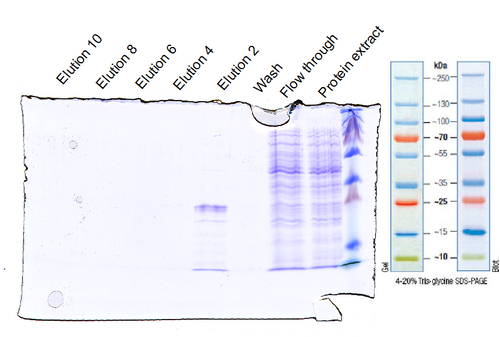
Ecto-domain III of the Epidermal Growth Factor Receptor (EGFR-d3) was modified at the genetioc level with a PelB-leader sequence adjacent to the amino-terminus and the the LPETG sortase motif at the carboxy terminus. The gene was cloned downstream of an arabinose promoter and the protein was expressed in XL1-Blue E.coli with arabinose induction. The PelB leader directs the expressed EGFR-d3 to the E. coli periplasm where it is cleaved by the membrane-anchored peptidase. We purified EGFR-d3 first by a periplasmic extraction followed by dialysis to remove sucrose (from the extraction) and to exchange the buffer. The protein was further purified via immobilized ion metal affinity chromatography using a Ni-NTA sepharose. Finally, the receptor was concentrated by ultrafiltration.
Coupling of aza modified peptides to alkyne modified oligonucleotides
We ordered an aza modified penta-Gly peptide and an alkyne modified oligonucleotide from specialized companies. Using both we tested coupling following published protocols (see lab-info) and tried to analyze the coupling efficiency by HPLC using an oligonucleotide analysis procedure recommended by the HPLC column manufacturer (Phenomenex, Jupiter proteo column). We have no clean results yet, but we are confident that the coupling works.
Discussion and outlook
We achieved patterning of DNA origami with proteins and made great progress towards our goal to couple proteins with DNA and protein nano-structures. There are still challenges ahead of us, such as the high Km value of the sortase enzyme. Nonetheless, we hope that our ideas and initial data contribute to the further development of the Biomod field.
Project Background
DNA origami
The process of folding DNA sequences to form DNA origami was developed by Paul Rothemund 2006. To construct DNA origami, we deisgned different DNA sequences with overlapping regions. After increasing the temperature and subsequenctly cooling of the sample mix. The overlapping regions hybridize and therefore form the DNA origami constructs.
Anti-biotin-antibody binds to biotinylated DNA origami
To ligate proteins and DNA molecules, we used biotinylated DNA origamis. These short biotin molecules are bound by anti-biotin molecules. With this approach, it is possible to built up threedimensional DNA origami strutures using protein to keep the origamis together.
Click Chemistry
The Click Chemistry was firstly described by K. Barry Sharpless 2001. The approach of the Click Chemistry is to use smaller subunits to built up a larger target molecule by using a chemical reaction. To achieve these target molecule, cells use different enzymes to produce larger molecules. The Click Chemistry approach is only based on chemical reactions. There are a lot of chemical reactions which can be used for the Click Chemistry. For our approach, we used the Azide alkyne Huisgen cycloaddition. In this reaction, a terminal or internal alkyne (1) reacts with an azide (2) catalysed by copper ions to form a 1,2,3-triazole (3). We used this reaction to ligate our DNA origami with a short aminoacid motif, the so called sortase motif.
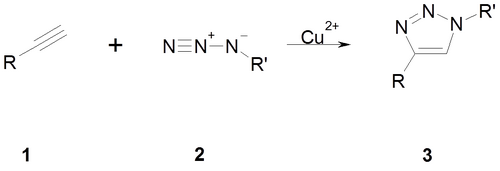
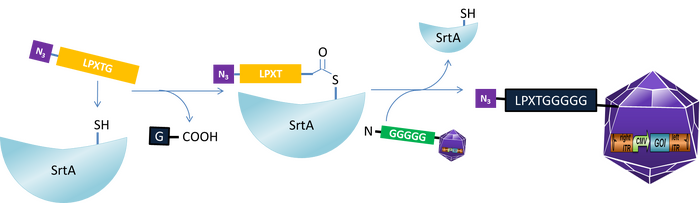
Sortase catalysed Protein-DNA ligation
The enzyme sortase is commonly used in gram-positive bacteria to covalently ligate the proteins to the peptidoglycan layers. Sortase is an enzyme which catalyzes specific ligation of two proteins to each other. In the first step the enzyme recognizes the C-terminal conserved LPxTG sortase motif and cleaves this motif between Gly and Thr. The resulting thioester intermediat reacts with an N-terminal glycine, regenerating a native amide bond. We used this enzyme to ligate the DNA origami with the EGFR (epidermal growth factor receptor domain three) protein and the adeno-associated virus (AAV). For this reaction, the sortase has to recognize the sortase motif GGGGG on the DNA origami and the LPXTG motif on the EGFR protein.
Adeno-associated virus 2
Genome
AAV is a small virus with single stranded linear genome of 4.7 kb. The genome consists of two open reading frames rep and cap which are flanked by two inverted terminal repeats (ITR).
The rep gene located on the 5'-end of the genome encodes four overlapping multifunctional regulatory proteins. The transcription of the Rep proteins begins at the promoters p5 (Rep 78/Rep 68) and p19 (Rep 52/Rep 40). The two larger Rep proteins Rep78 and Rep68 are required for genome replication, the regulation of gene expression, and for site specific integration. The smaller Rep proteins Rep52 and Rep40 are involved in the encapsidation of the AAV-Genome.
The open reading frame cap on the 3'end of the AAV-genome encodes the three viral capsid proteins VP1, VP2, and VP3. The transcription of the mRNA is regulated by the p40 promoter. Alternative splicing results in two mRNA molecules: one encoding VP1 and the other VP2/ VP3. Application of an alternative splice acceptor removes the first AUG start codon for VP1. Thereby mRNA that is mainly formed encodes for the VP2 and VP3 capsid proteins. The first AUG codon in the transcript is the initiation codon for VP3. The translation of VP2 begins at an upstream ACG non-methionine start codon resulting in an approximately 10-fold lower translation of VP2. The stop codon of the three capsid proteins is identical. The stoichiometric ratio of the three capsid proteins VP1, VP2 and VP3 forming the icosahedral form is 1:1:10.
The ITRs are the indispensible cis-acting elements, which are critical for genome replication and packaging. The ITRs contain the Rep-binding site (RBS), which binds the Rep78/68 by unwinding the ITR at the terminal resolution site (trs).
Infection
The virus particles present in the cell media come closer to the cells by diffusion. After several attachments to the cell membrane the virus binds to the cell receptors and enter the cell by receptor mediated endocytosis. There are three cell receptors playing a major role in non-effectivity. Ubiquitously expressed cell surface Heparan sulfate proteoglycan (HSPG) functions as a primary receptor account for the broad host range of AAV. aVß5 integrin and fibroblast growth factor 1 (FGFR-1) have a co-receptor activity for successful viral entry into the host cell. Bound AAV particles enter the cell very rapidly through clathrin mediated endocytosis. Each endosome carries a single AAV particle. After internalization the virus requires acidic environment for sufficient release into the cytosol. AAV accumulates in the perinuclear space and enters through the nuclear pore complex (NPC) into the nucleus, where uncoating and gene expression take place.
AAV2 viruses kill cancer cells without harming healthy ones. Because of its efficient and specific delivery of genes to the cells the Adeno-associated-Virus 2 has gained a big interest in gene therapy.
Life cycles
After infection AAV escapes the lysosome and enters the nucleus where the viral genome is released. In the presence of a helper virus, Adenovirus or Herpesvirus, the AAV genome is replicated, new capsids are produced, filled with the AAV DNA and later release into the environment.
If there is no helper virus, AAV can establish latency by integration into the host genome. The Rep expression is limited. When AAV infects a human cell, it integrates in a site-specific manner into the chromosome 19, called AAVS1. Essential components are the ITRs and the AAVS1 in cis and Rep proteins 78/68 in trans. Rep78/68 binds to the Rep-binding site (RBS) on ITR and unwinds the ITR at the terminal resolution site (trs).
The productive cycle is initiated by infecting with the helper virus, what affects stronger Rep expression and episomal AAV DNA replication. Rep proteins excise the AAV genes out of the host cell’s chromosome. The excised genes are replicated in the nucleus and the capsid proteins package the replicated genes in the cell. When a large number of these particles have assembled, the helper virus induces cell lysis and release the newly assembled particles into the environment.
Recombinant AAV2
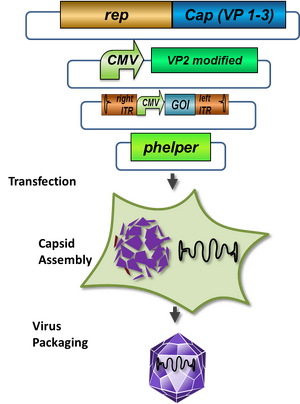
AAV are replication-deficient parvoviruses. Although AAV can replicate in the absence of a helper virus, they require a co-infection of adenovirus for an efficient replication. To bypass the co-infection with adenoviruses, which is associated with disadvantages like the contamination of AAV preparation, all of the required genes of adenovirus were encoded on a helperplasmid. The helperplasmid consisting of adenovirus E2A, VA und E4 genes, is transfected in the AAV-293-cells, which stably express the adenovirus E1 gene. The production of infectious adeno-associated virus particles is able by co-transfecting the AAV-293 cells with four plasmids:
- the plasmid that carries the helper genes taken from adenovirus
- a plasmid coding the protein fused on VP2 cap-gene,
- a vector plasmid that contains the wildtype AAV2 Rep-Cap genes with VP2 knock out
- AAV2 ITR-providing plasmid carrying the gene of interest. The transgene cassette flanked by the ITRs is wrapped as a single strand in the virus capsid. The replication of the plasmid with transgene cassette is implemented with rep-proteins and enveloped in the viral capsid formed by cap-proteins.
Applications
The combination of the encoding properties of DNA and binding functions of proteins provide us with many applications in medicine. One of the most recently discussed therapies is the gene therapy. For this purpose, DNA sequences are transported into target cells via different cargo systems for example virus. However, using virus molecules can cause many problems like immune reactions or unspecifically integration into the genome. By ligating therapeutic DNA sequences with binding proteins like antibodies, it is possible to treat only target cells with DNA avoiding the usage of virus.

