Vascularization by Julia Tomaszewski
Introduction
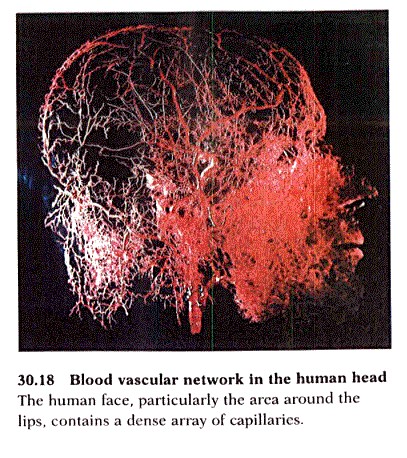
Vascularization is the development of vascular networks within an engineered tissue once being implanted and interacting with a host’s tissue [1]. Capillary formation may take up to several weeks, depending on the thickness of this tissue. This is because the vascular interactions from the host are only able to penetrate several tenths of a micrometer through the tissue per day [1]. The main challenge of vascularization is providing an immediate blood supply to engineered vascular tissues implanted into a host. Methods to improve the mass transfer characteristics of this tissue are currently being developed, which would allow rapid oxygen and nutrient diffusion throughout the engineered tissue. Capillaries are typically 100-200 µm away from the cells, allowing for easy transfer of oxygen and nutrients to the cells [1][2]. Tissues exceeding 200 µm in thickness, such as heart, brain, and liver tissues, require additional methods for diffusion and transfer of oxygen and nutrients [3].
Motivation
The need for advances in tissue engineering is crucial in order to meet the demands of patients requiring organ transplants and tissue repair. Organ donations are extremely limited but they are the most ideal form of treatment because they are typically fully functional once implanted into a host [3]. These transplanted organs already consist of integrated vascular networks which can easily diffuse into the patient once they interact with one another. Tissue engineering techniques are limited when it comes to the formation of vascular networks in engineered tissue. Advanced approaches to vascularize tissue are essential in order to maintain the vitality and functionality of the implanted cells [4].
Strategies of Vascularization
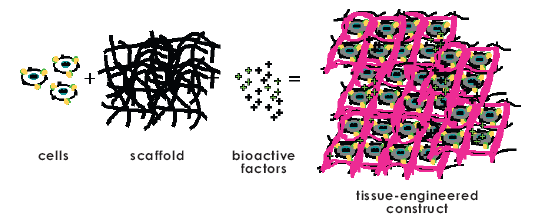
Scaffolds
One of the easiest designed strategies of vascularization is the use of engineered scaffolds to implant tissues [1]. They are beneficial not only because of the simple transplantation of the desired tissues, but they may also comprise additional elements or factors to promote diffusion and transport within the tissue.
Tissues engineered in vitro are cultured in controlled environments allowing oxygen and nutrients to be transferred within the tissue, but problems begin once the tissue is implanted into the host because of diffusion limitations [1]. To overcome these limitations, angiogenic factors can be incorporated into scaffolds with engineered tissues to assist in the diffusion process to promote vessel production. Growth factors such as vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), and platelet-derived growth factor (PDGF) may be used to produce this angiogenic effect [2]. With the addition of these growth factors in the scaffold, the implanted tissue is able to integrate into the host with increased stability while also increasing the rate of blood flow throughout the tissue, improving the viability of the cells [2]. When multiple growth factors are combined in a scaffold, the formation of blood vessels is increased. For example, when VEGF and PDGF are coupled in a scaffold, VEGF is released rapidly while PDGF is released slowly and more uniformly [2] because of their varying stimulation levels in the blood vessel [1]. Growth factors show great promise for vascularization, but if added in excess, they may be toxic to the tissue and blood vessels. Tissues containing a high concentration of growth factors may lead to vessel leakage and may even cause hypotension [1].
The design of the scaffold may also be engineered to increase vascularization. Pore construction of the scaffold is essential for vessel diffusion. A pore size greater 250 µm has shown to increase the rate of vessel diffusion of the tissue [1]. In addition to pore size, pores must also be connected in a way to permit cell mobility to promote vessel transmission [1]. Difficulty persists in pore construction because of the randomness of formation [1].
In vivo Systems
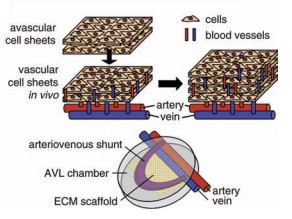
The incorporation of angiogenic factors and manipulation of the design of the scaffold to increase vessel diffusion in the tissue are suitable methods to increase vascular network formation rates, but these strategies are limiting for thick tissues [2]. Methods designed to distribute capillaries throughout a thick tissue require in vivo prevascularization [5]. Cell sheet technology can be utilized for this strategy. It is a multi-step process requiring multiple surgeries, but once these tissue are implanted into a host, blood vessels are able to immediately integrate throughout it [1][2].
A study conducted in rats showed a successful in vivo vascularization process [2][5]. First, an 80 µm thick cell sheet tissue is implanted directly onto a healthy site in the host [2]. The advantage of using in vivo systems is that blood can be supplied to the implanted tissue by the host [1]. Vascular networks formed within the tissue in 1-3 days [2]. With the successful network organization, additional cell sheet layers can then be added to this tissue on the rat over time [2]. Depending on the thickness of the desired tissue, development of vascular tissue can take several weeks [1]. Once the thick tissue has completely vascularized, it is then removed from this healthy site of the host and transplanted onto the damaged site. The implanted tissue is immediately able to integrate its capillary network with the host capillary network throughout the entire engineered tissue [1], which is the main advantage of this strategy.
Conclusions and Future Directions
There are still many obstacles to overcome in the field of vascularization in thick tissues. Advanced methods to promote vascular tissue are still being researched. The combination of already developed methods may be a promising approach to promote vascular formation [1]. Also, in addition to engineering the speed of vessel development, the quality of the vessels should also be taken into consideration [1]. Improved vessel formation may enhance the development of vascular networks in the tissues. Clinical trials have shown some positive results, but a great deal of research still remains [5].
References
[1] Rouwkema, Jeroen, Nicolas C. Rivron, and Clemens A. van Blitterswijk. Vascularization in tissue engineering. Cell Press. June 2008;26(8):434-441.
[2] Lovett, Michael, Kyongbum Lee, Aurelie Edwards, et al. Vascularization Strategies for Tissue Engineering. Departments of Biomedical Engineering and Chemical and Biological Engineering, Tufts University, Medford, Massachusetts. 2009;15(3):353-370.
[3] Jain, Rakesh, Patrick Au, Josh Tam, et al. Engineered Vascularized Tissue. Nature Biology. 2005;23(7):821-823.
[4] Chen, Xiaofang, Anna S. Aledia, Cyrus M. Ghajar, et al. Prevascularization of a Fibrin-Based Tissue Construct Accelerates the Formation of Functional Anastomosis with Host Vasculature. Tissue Engineering Part A. June 2009;15(6):1363-71.
[5] Stegemann, Jan, Stephanie N. Kaszuba, and Shaneen L. Rowe. Review: Advances in Vascular Tissue Engineering Using Protein-Based Biomaterials. NIHPA Manuscripts. Nov. 2007;13(11):2601-13.
[6] Bordalier Institue. <http://www.bordalierinstitute.com/target1.html>
[7] Department of Bioengineering. National University of Singapore. Dec. 2012. <http://www.bioeng.nus.edu.sg/research/Keyresearch/keysrh1.html>
