User:Yale J.T. Friedman/Notebook/Biology 210 at AU
3/18/16 Zebra Fish Experiment - Final Week - Final Observations and Cleanup
Purpose The purpose of this experiment was to make any final observations about the zebra fish and to dispose of the tap water that they were housed in. Earlier during this week on Monday, all the zebra fish in the tap water control had passed away, so they had been preserved in paraformaldehyde. Part of this experiment was to make any final observations about the morphology of the zebra fish now that they had been preserved. It was predicted that the zebra fish had changed in texture and color due to the preservation with paraformaldehyde.
Materials and Methods First the tap water from both the petri dish and the well plate were disposed of. Next the zebra fish that had been preserved in paraformaldehyde were observed by looking through the vials containing them.
Data and Observations At this point, the color, texture, or any morphological feature did not appear to have been affected. The solution was clear with the deceased fish inside.
Conclusions and Further Directions No conclusions were made other than that at this point the paraformaldehyde did not affect the morphology of the zebra fish.
Y.J.T.F
3/3/16 Zebra Fish Experiment - Week 2 & Bacterial Sequencing Results
Purpose The purpose of this week's lab experiment was to check the current status of the zebrafish development and continue to collect data to determine results that the treatment condition of Cadmium Bromide, and the control conditions of tap water and Deerpark water had on the zebrafish. Again the hypothesis for this experiment is that there will be little or no affect on the growth and development of the zebrafish from the tap water control condition, and that there will be a significant impact on the growth and development of the morphological and behavioral traits in the Cadmium Bromide treatment condition. The Deerpark water control condition is predicted to have normal growth. The purpose of this lab is also to analyze the bacterial sequencing results from Transect 1.
Materials and Methods In order to replace the zebrafish from the tap water control group that did not hatch by Friday, Feb. 26, approximately twenty new zebrafish that were nine days post fertilization were placed in a petri dish. The petri dish was filled up to a line on the bottom of the plate with half deer park water and half tap water from the same container that was used in the first batch that did not survive. The zebrafish were then given 30μL of brine shrimp as food to last them till Monday, Feb. 29.
Handling the Deceased Zebrafish: Monday, Feb. 29 The deceased zebrafish in the petri dish were observed under the dissecting scope using 1x magnification. A few were separately put on a depression slide to view under the microscope to measure them and to confirm that they had no heartbeat. The zebrafish were observed under 4x and 10x magnification, and were measured using the ocular micrometer. Once it was confirmed that the zebrafish did not survive, the twenty zebrafish were preserved by placing them in a vial, and add 1mL of the substance that makes them numb, and then added 4mL of paraformaldehyde.
Nucleotide Blast: Bacterial Sequencing The nucleotide bacterial sequence from transect one was copied into the nucleotide blast from the NIH website. This was done in order to determine if the bacteria characterized after the Gram stain procedure several labs ago.
Data and Observations On Friday, Feb 26, the zebrafish in the tap water control group still had not hatched, so it was concluded that none would not survive. A total of 24 zebrafish did not survive from that batch. A new batch of healthy zebrafish that were nine days post fertilization replaced the previous group, and were a healthy greenish yellow color. They were approximately 3.6mm in length, were very fast swimmers, and reacted very quickly when a transfer pipet came near them to put them in the petri dish. On Monday, Feb. 29, all of the fish from the new batch were deceased and became rather black in color and dried up. The tissue almost looked necrotic.There were signs of movement, and many of them were almost stuck to the petri dish. There were no signs of a heartbeat, and many of the fish had their heads down towards the bottom of the plate with their tails floating upward. Approximately 20 zebrafish did not survive the control condition of half Deerpark water and half tap water. These results for the the tap water condition are recorded below in Table 1.
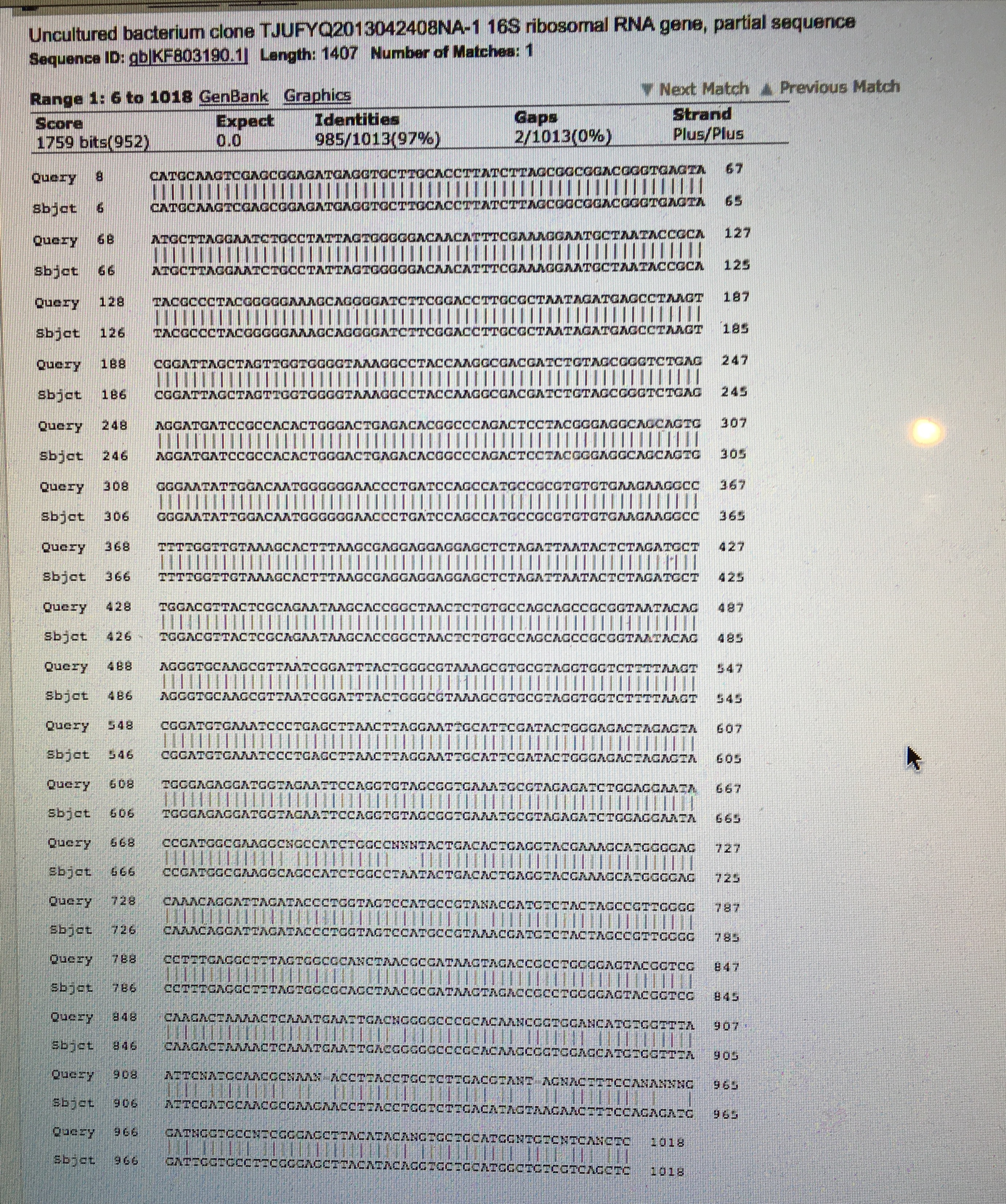
The nucleotide bacterial sequence from Transect One is shown below in Figure 1. After running the nucleotide blast, the results showed that the bacteria in the transect are uncultured bacterium clone. While it is possible that uncultured bacteria are in Transect One, the bacteria that was observed with the gram stain does not match up. It appears that uncultured bacterium is refering to bacteria that cannot be grown in a lab. However, the bacterial samples from Transect One grew on the agar and blood Petri dishes after one week. There was one agar plate however that did not get any growth at all, which could indicate that either the antibiotics restricted the growth or that the bacteria was uncultured. The results of the mei peptide blast are shown below in Figure 2.
Table 1. Development Observations Week 2 - Control Condition
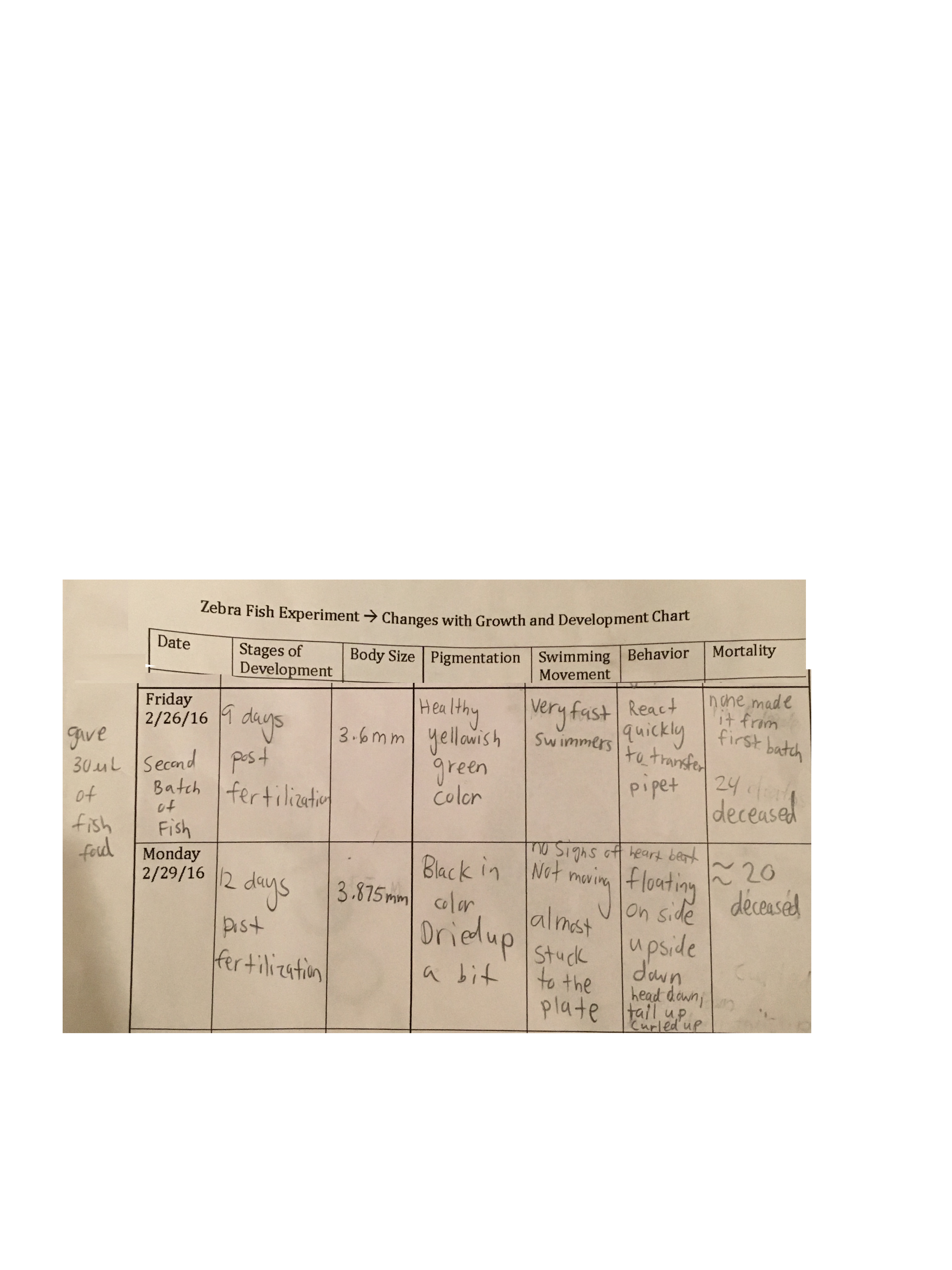
Figure 1. Bacterial Nucleotide Sequence from Transect One NNNNNNNNNNNNNGCNTACNCATGCAAGTCGAGCGGAGATGAGGTGCTTGCACCTTATCTTAGCGGCGGACGGGTGAGTA ATGCTTAGGAATCTGCCTATTAGTGGGGGACAACATTTCGAAAGGAATGCTAATACCGCATACGCCCTACGGGGGAAAGC AGGGGATCTTCGGACCTTGCGCTAATAGATGAGCCTAAGTCGGATTAGCTAGTTGGTGGGGTAAAGGCCTACCAAGGCGA CGATCTGTAGCGGGTCTGAGAGGATGATCCGCCACACTGGGACTGAGACACGGCCCAGACTCCTACGGGAGGCAGCAGTG GGGAATATTGGACAATGGGGGGAACCCTGATCCAGCCATGCCGCGTGTGTGAAGAAGGCCTTTTGGTTGTAAAGCACTTT AAGCGAGGAGGAGGAGCTCTAGATTAATACTCTAGATGCTTGGACGTTACTCGCAGAATAAGCACCGGCTAACTCTGTGC CAGCAGCCGCGGTAATACAGAGGGTGCAAGCGTTAATCGGATTTACTGGGCGTAAAGCGTGCGTAGGTGGTCTTTTAAGT CGGATGTGAAATCCCTGAGCTTAACTTAGGAATTGCATTCGATACTGGGAGACTAGAGTATGGGAGAGGATGGTAGAATT CCAGGTGTAGCGGTGAAATGCGTAGAGATCTGGAGGAATACCGATGGCGAAGGCNGCCATCTGGCCNNNTACTGACACTG AGGTACGAAAGCATGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCATGCCGTANACGATGTCTACTAGCCGTTGGGG CCTTTGAGGCTTTAGTGGCGCANCTAACGCGATAAGTAGACCGCCTGGGGAGTACGGTCGCAAGACTAAAACTCAAATGA ATTGACNGGGGCCCGCACAANCGGTGGANCATGTGGTTTAATTCNATGCAACGCNAANACCTTACCTGCTCTTGACGTAN TAGNACTTTCCANANNNGGATNGGTGCCNTCGGGAGCTTACATACANGTGCTGCATGGNTGTCNTCANCTCNTNNCNNTN NNNNNNTTNNN
Figure 2. Uncultured Bacterium Clone
New batch of healthy zebrafish 9 days post fertilization

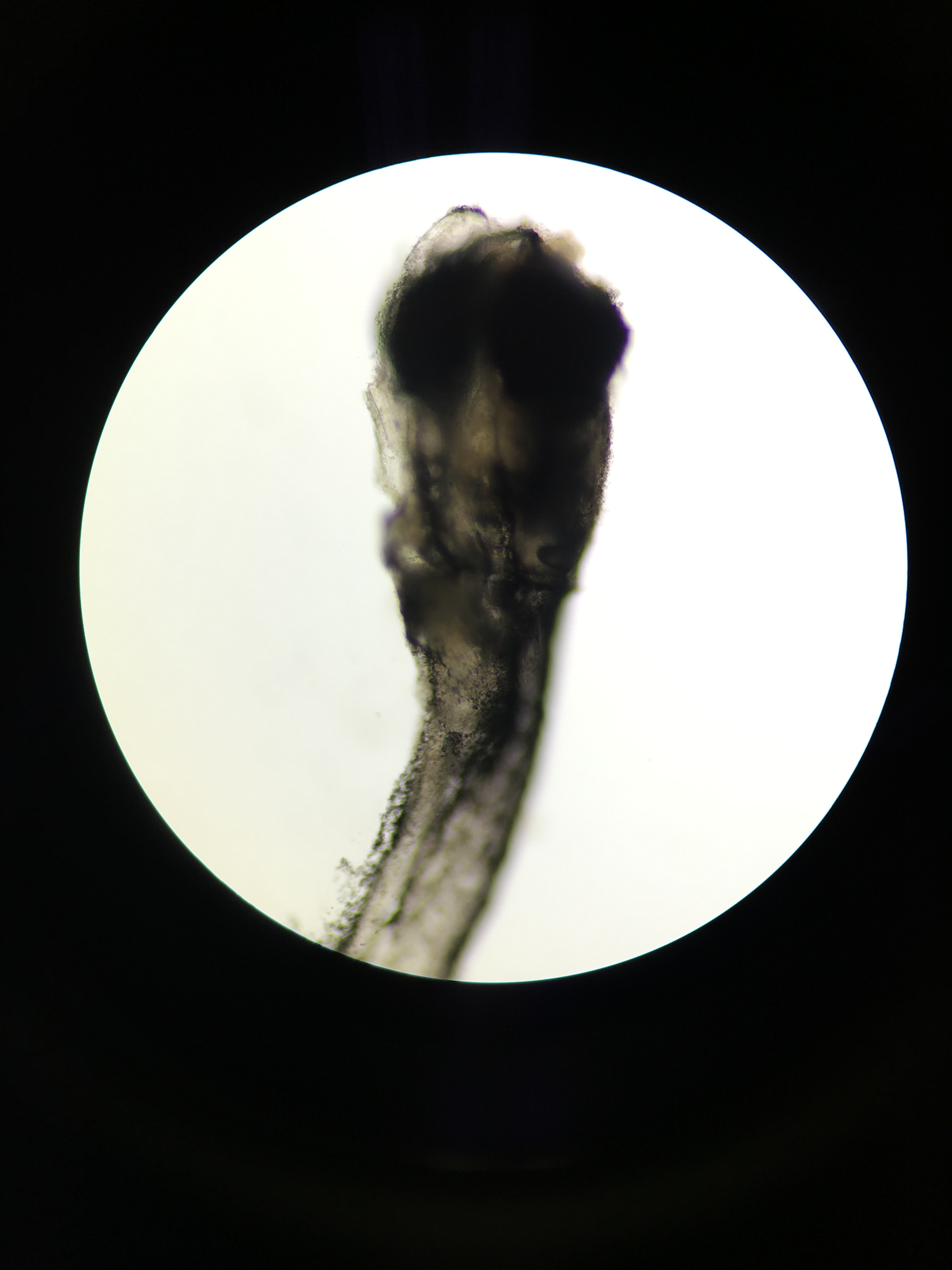
Deceased zebrafish 11 days post fertilization; 4x magnification
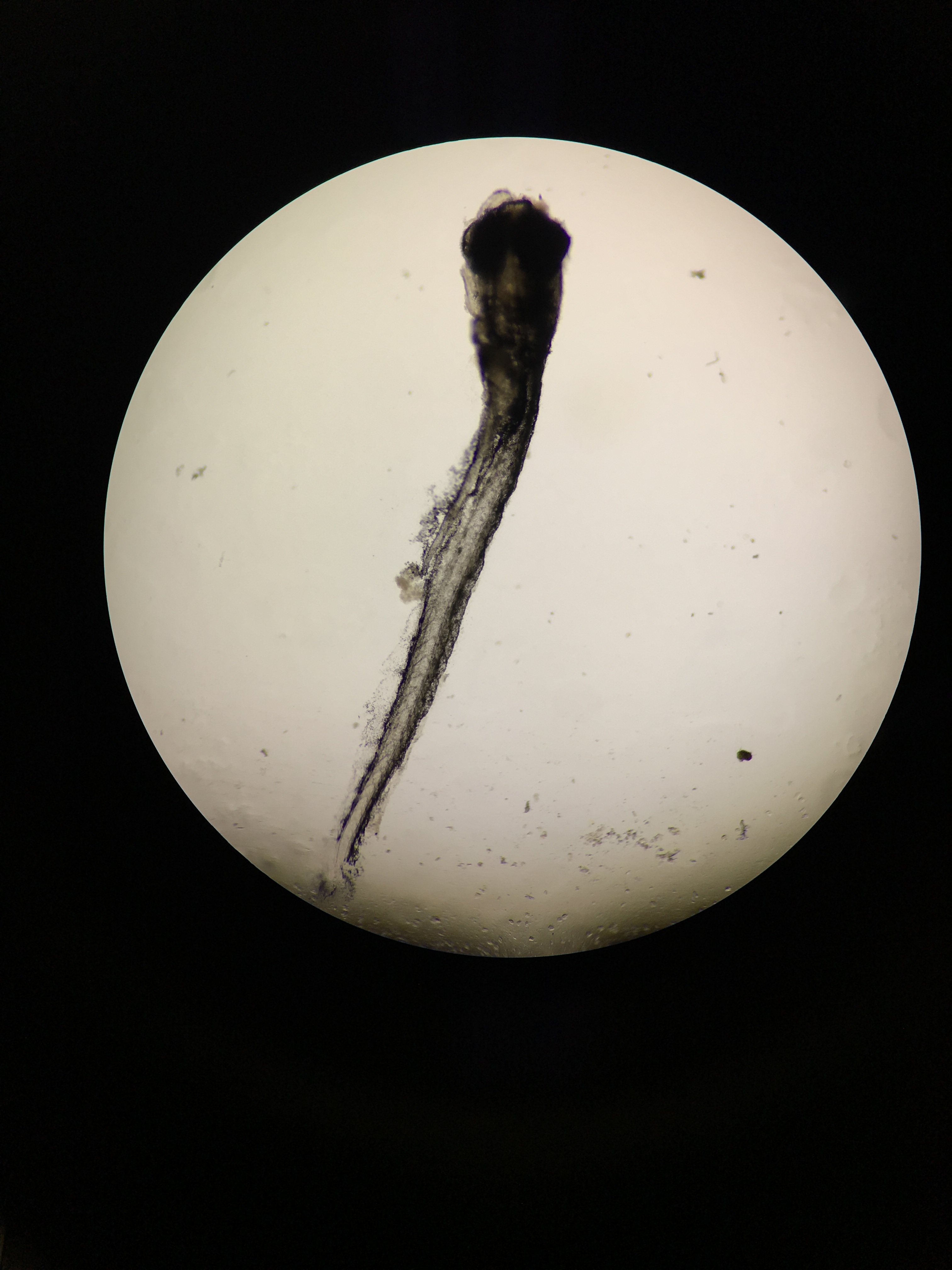
Deceased zebrafish under 10x magnification; 11 days post fertilization
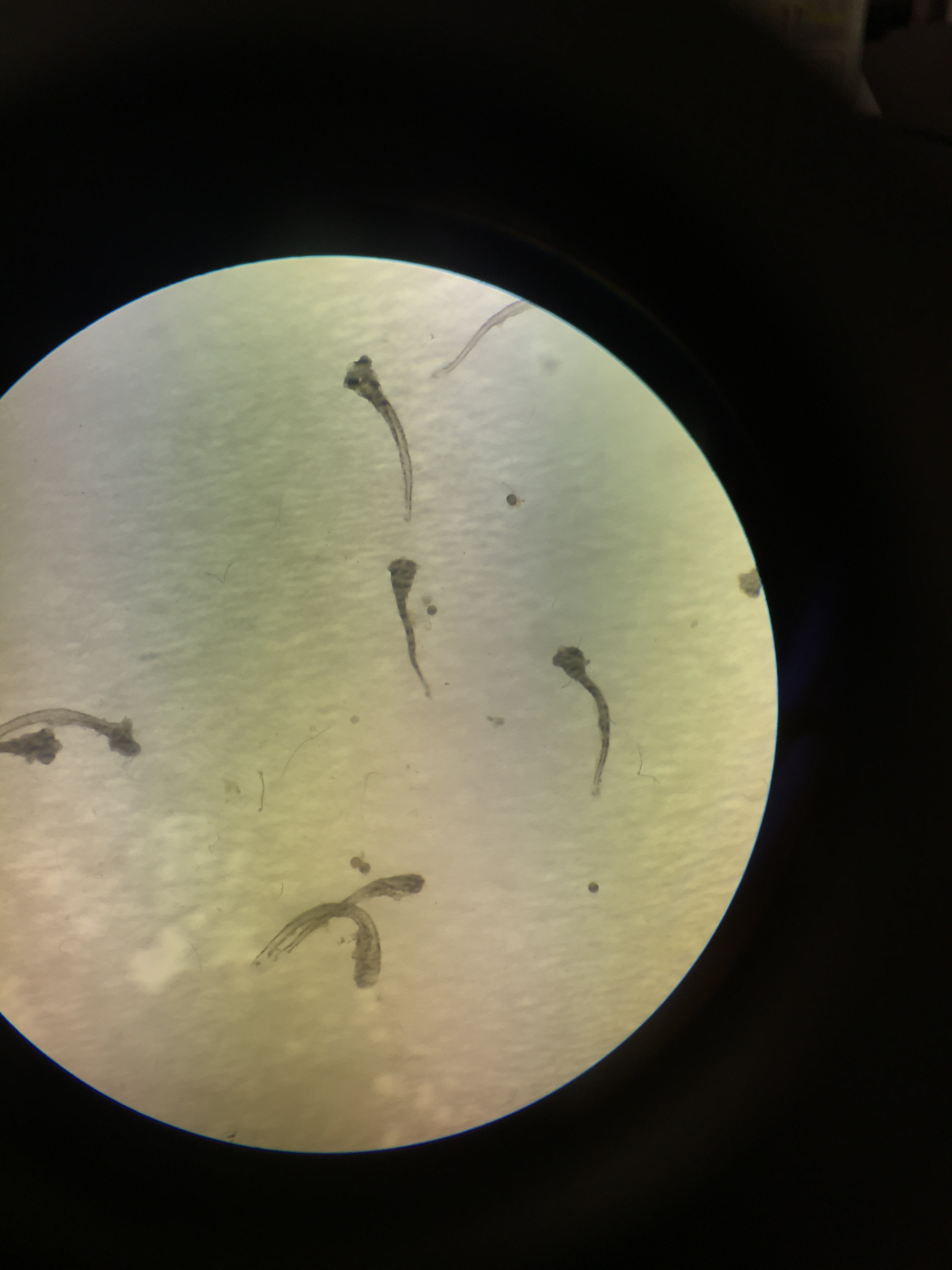
Group of deceased zebrafish under dissecting scope - 1x magnification; 11 days post fertilization
Conclusions and Further Directions Based on the fact that all of the fish in the tap water condition passed away and quite quickly, it indicates that there is some sort of a chemical in the tap water that is affecting them. Given the fact that zebrafish in the Cadmium Bromide survived for a much longer period of time than the fish in the tap water condition (according to lab partner) there is clearly another chemical in the water other than Cadmium Bromide that is harmful to the zebra fish. These other chemicals could be chlorine, nitrate, or chloramine. These results go against the hypothesis in the experiment in that there was a major impact on the zebrafish from the tap water condition. It was most likely unrelated to the Cadmium Bromide in the tap water however, as they became deceased extremely quickly. The next step is to continue researching other chemicals that could are typically in the tap water supply and are harmful to zebrafish. In terms of the bacterial sequence results, we know that the primers did not bond after PCR, but it is possible that there was uncultured bacteria in Transect One as one of the agar Petri dishes did not have any growth in the lab.
Y.J.T.F
2/26/16 Zebra Fish Experiment - Week 1
Purpose The purpose of this lab is experiment is to be able to determine the affect that a chemical, in this case the carcinogen Cadmium Bromide as the treatment group, has on the growth and development of zebra fish, and compare it to the growth and development of zebra fish in tap water. The independent variable of this experiment is the Cadmium Bromide, and the dependent variable is the impact that it has on the growth and development of the zebra fish. The control condition is the zebra fish exposed to tap water during their growth and development. The tap water already has a bit of Cadmium Bromide in it, which may have an affect on the growth and development of the zebra fish. The hypothesis of this experiment is that the tap water will have little to no affect on the growth and development of the zebra fish, while the Cadmium Bromide will have a significant influence on the growth as well as mental and morphological development.
Materials and Methods The process of setting up the zebra fish experiment began by taking two 24 well plates, one for the control condition and one for the treatment condition, and filling each with 2-3mL of water. The control condition plate was given tap water, and the treatment condition plate was given Deerpark water. Two petri dishes were used to transfer 48 zebra fish (24 on each plate) from the bowl that they arrived in. To determine the approximate age of the zebra fish and verify that they were each around the same age, the petri dishes were observed under the dissecting scope. Next, the zebra fish embryos from one of the petri dishes were individually placed into each of the 24 wells of the well plate containing the tap water control condition using a transfer pipette. The same process was repeated in the other well plate for the treatment condition, and then Cadmium Bromide was added to that well plate to determine the developmental affects this chemical has on the zebra fish. Throughout the week observations about the growth and development were made every two days.
Data and Observations The lab manual reports that the zebra fish should be approximately 18-36 hours post fertilization, and zebra fish observed on the dissecting scope appeared to be at a rather early development within that time range post fertilization. The embryos were small, spherical, and overall transparent with a small spot in the center of the embryos.
Results of the Control Condition in Tap Water: The results four days after the zebra fish were placed in their wells (Monday, Feb. 22) show that only one of the zebra fish hatched, but had very little further development at that point. The other zebra fish embryos did not hatch, and remained rather clear in color and transparent. There was no behavioral activity as the embryos remained immotile. One the embryos did not survive as it was surrounded by fungus. The results six days after the zebra fish were placed in their wells (Wednesday, Feb. 24) showed that no additional embryos hatched during the two day period between Monday and Wednesday. The embryos remained immotile, as did the one fish that seemed to hatch the previous Monday. Some of the embryos appeared to have a rather beige tint to them but remained overall transparent as they had been before. There was no behavioral activity observed during this period, and there were no more fatalities. These observations about the development of the zebra fish in the control condition are recorded in Table 1 below.
Table 1: Development Observations Week 1 - Control Condition

Unhatched Embryo from Monday Check in

Hatched Embryo from Monday Check in

Unhatched Embryos from Wednesday Check in
Hatched Embryo from Wednesday Check in
Conclusions and Further Directions
Based on the fact that only one out of twenty-four zebra fish embryos hatched out of their eggs in a 6 day period, there is not enough evidence yet to conclude whether or not the tap water control condition has an affect on the growth and development of the zebra fish, nor are the fish developed enough to compare to the treatment condition. It is possible that an agent in the tap water prevented the zebra fish from developing, but that seems very unlikely. The results so far suggest that the batch of eggs are not developing as expected in normal conditions, and that this particular group needs more time to develop. The zebra fish in the control condition well plate will be observed again on Friday, February 26th to reevaluate their development.
Y.J.T.F
2/19/16
Invertebrates and Vertebrates - Berlese Funnel Collections and Food Web
Purpose The purpose of this lab experiment is to be able to collect invertebrates from the transect by using a Berlese Funnel Collection, and identifying the different invertebrates that reside within the transect. It is also important in this lab to be able to differentiate between vertebrates and invertebrates and how each play a role in ecology. Prediction: Given the wide range of diversity within the plant life in the transect, we predict that there will also be a large variety of invertebrates living there.
Materials and Methods
The process of the Berlese Funnel setup began by taking a funnel and taping down a piece of screening material in the bottom center of it. Next, a conical tube was filled with 25mL of ethanol, and was attached to the base of the funnel using Parafilm (to prevent evaporation of the ethanol) and tape. A handful of leaf litter from the transect was then added to the funnel. The funnel was placed on a ring stand under a 40 watt incandescent lamp, and it was surrounded by aluminum foil. The funnel was left there for one week to allow the invertebrates present in the leaf litter to migrate down to the ethanol in the conical tube.
One week later, the funnel was removed from the ring stand and separated from the conical tube. The ethanol solution that contained the leaf litter matter and invertebrates that transferred from the top of the funnel into the ethanol, were poured into two dishes. One contained the top half of the sample, and the other contained the bottom half. The samples were observed under the dissecting scope using 3x magnification, and the invertebrates were measured with a basic ruler in millimeters. Lastly, the invertebrates were identified using a dichotomous key. The results consisting of the invertebrates found and their descriptions are located in Table 1 below.
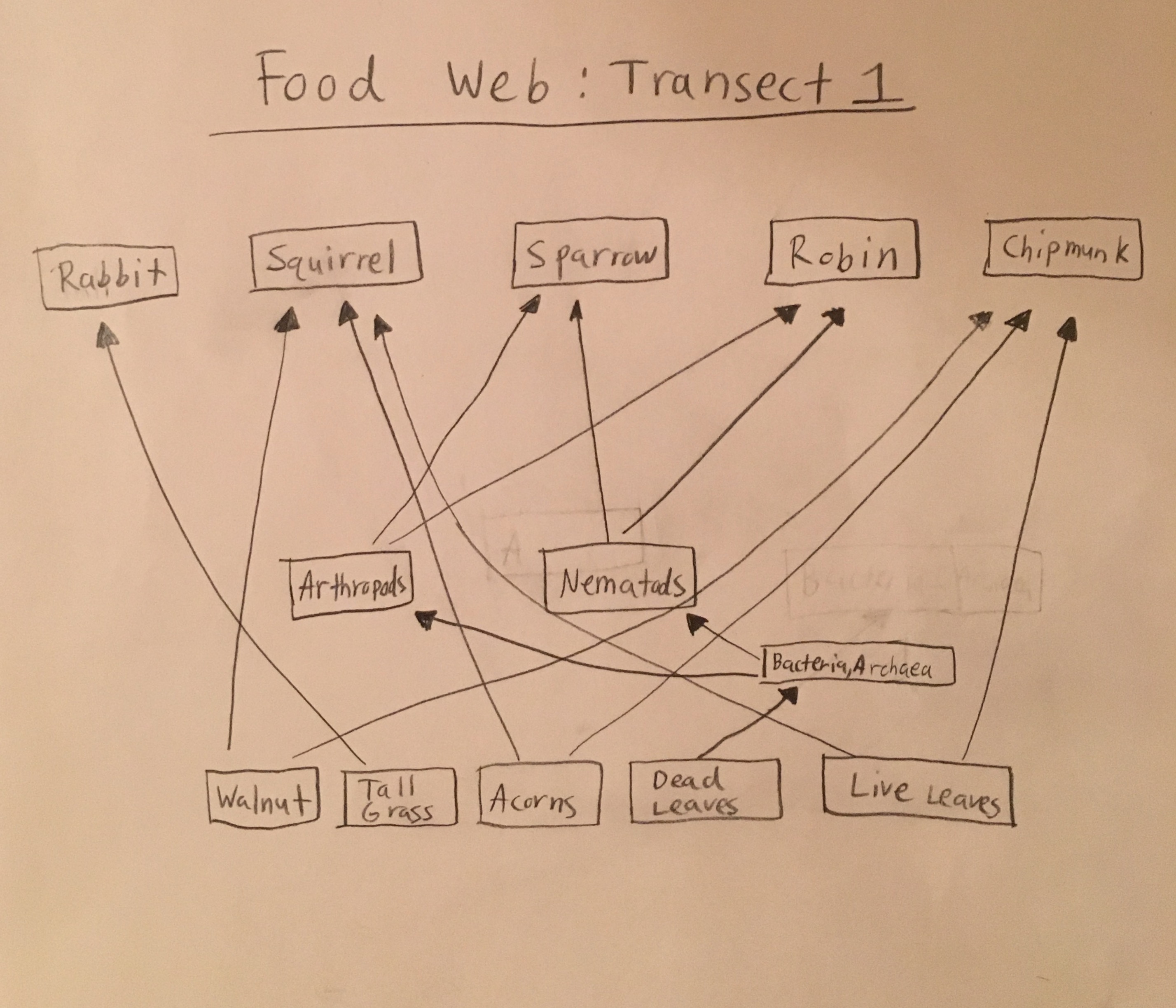
A food web was created to demonstrate the food sources that the different biotic features of the transect provide for each other, and to identify the primary, secondary, tertiary, etc. consumers and the primary producers in the transect.
Data and Observations
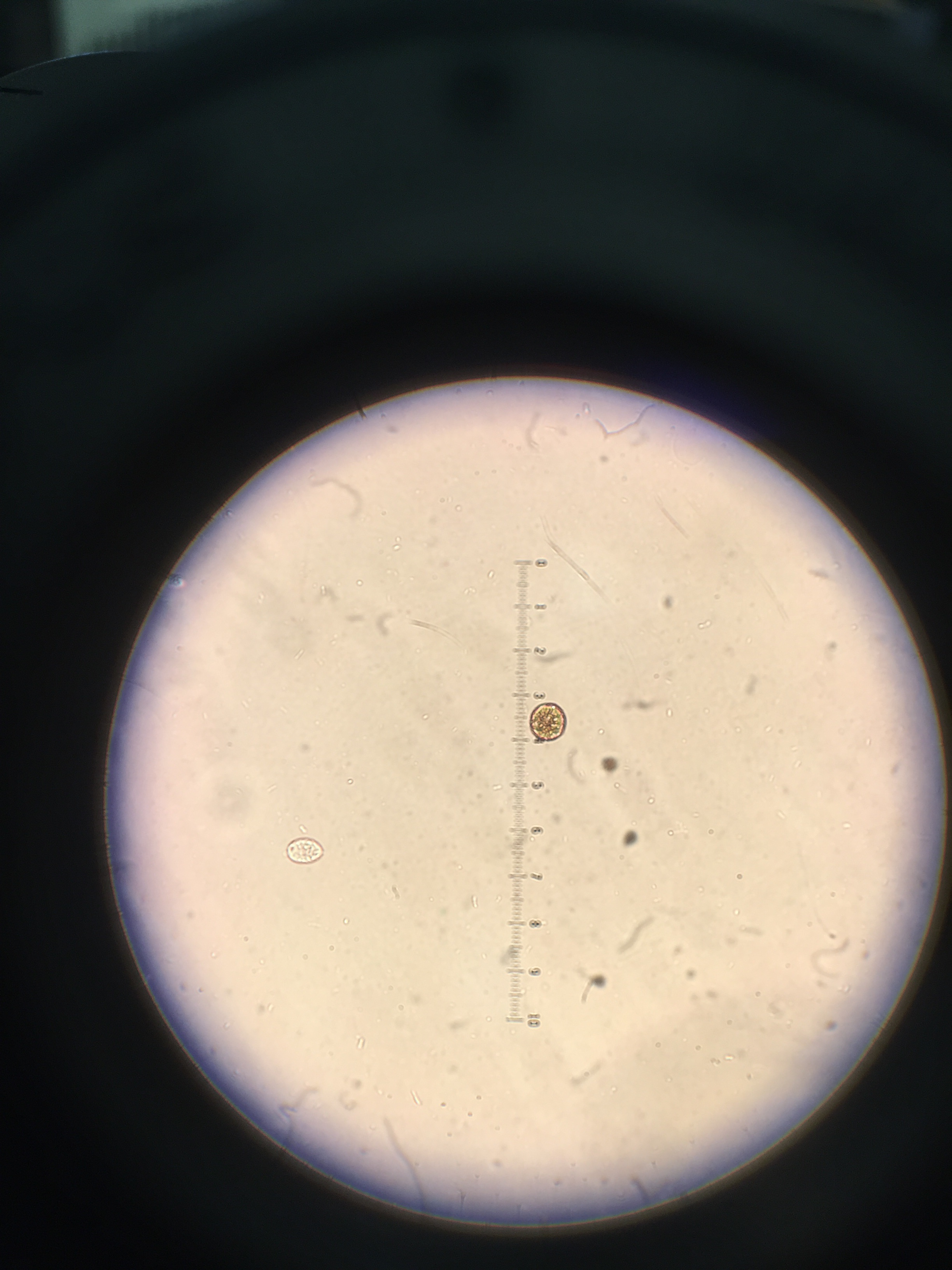
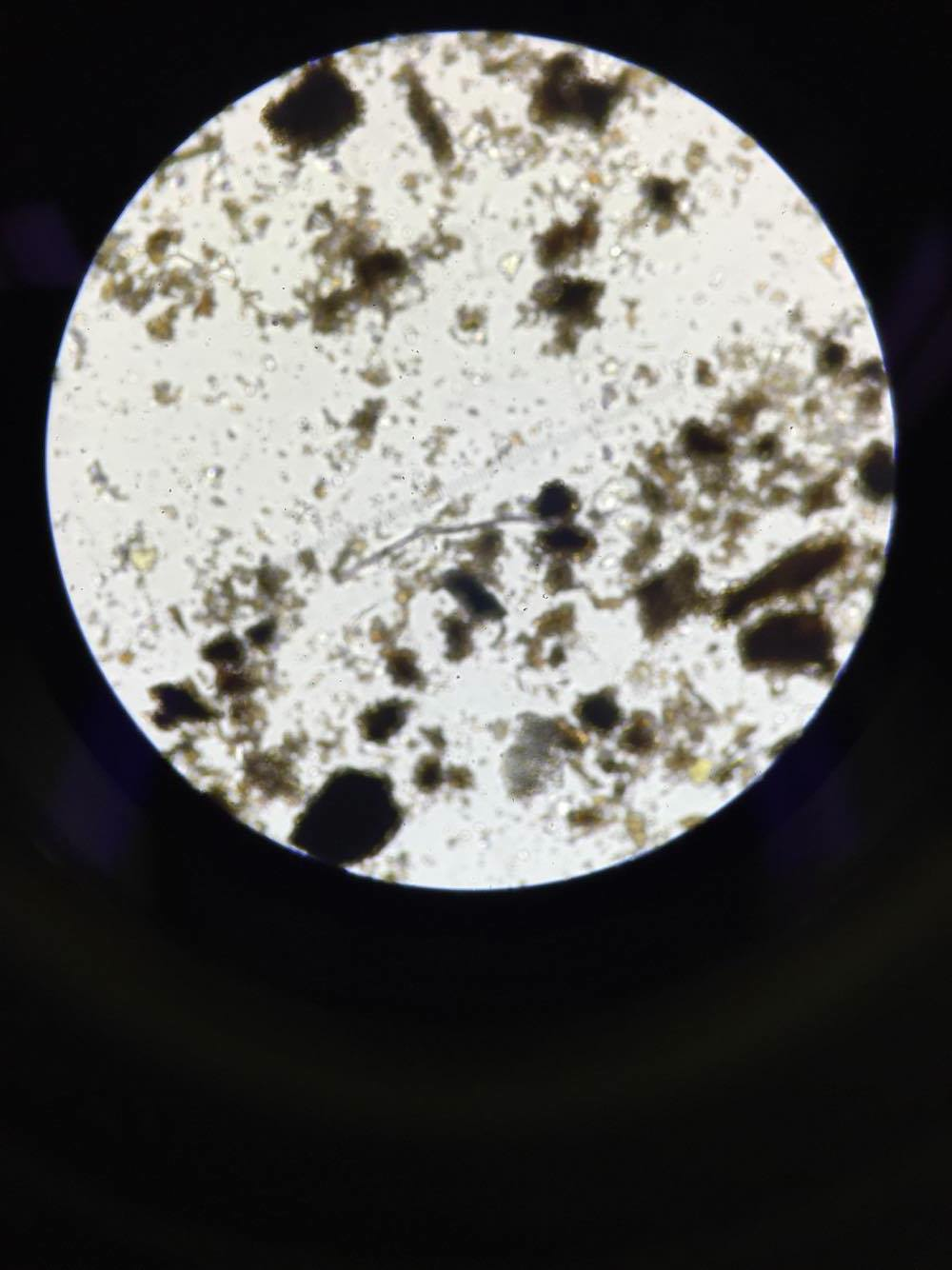
The six invertebrates found in the Berlese Funnel collections include: 3 Phylum: Nematoda; Class: Enoplia, 1 Phylum: Arthropoda; Class: Diplopoda, 1 Phylum: Arthropoda; Class: Insecta Collembola, and 1 Phylum: Arthropoda; Class: Homoptera, as shown below in Table 1. Note that the first five invertebrates listed in the table came from Transect 4, and the last one came from our Transect 1. The size range of the organisms were from 2mm to 22.5mm. One of the Nematoda Enoplia is the largest organism at 22.5mm, and the Arthropoda Homoptera measured the smallest at 2mm. The most common organisms in the leaf litter were Nematoda Enoplia, and also three different types of Arthropods. The top and bottom samples from Transect 4 are different in that the top sample contained Nematoda Enoplia, and the bottom sample contained arthropods. The top sample from Transect 1 contained an arthropod as well.
Table 1: Invertebrate Description Table
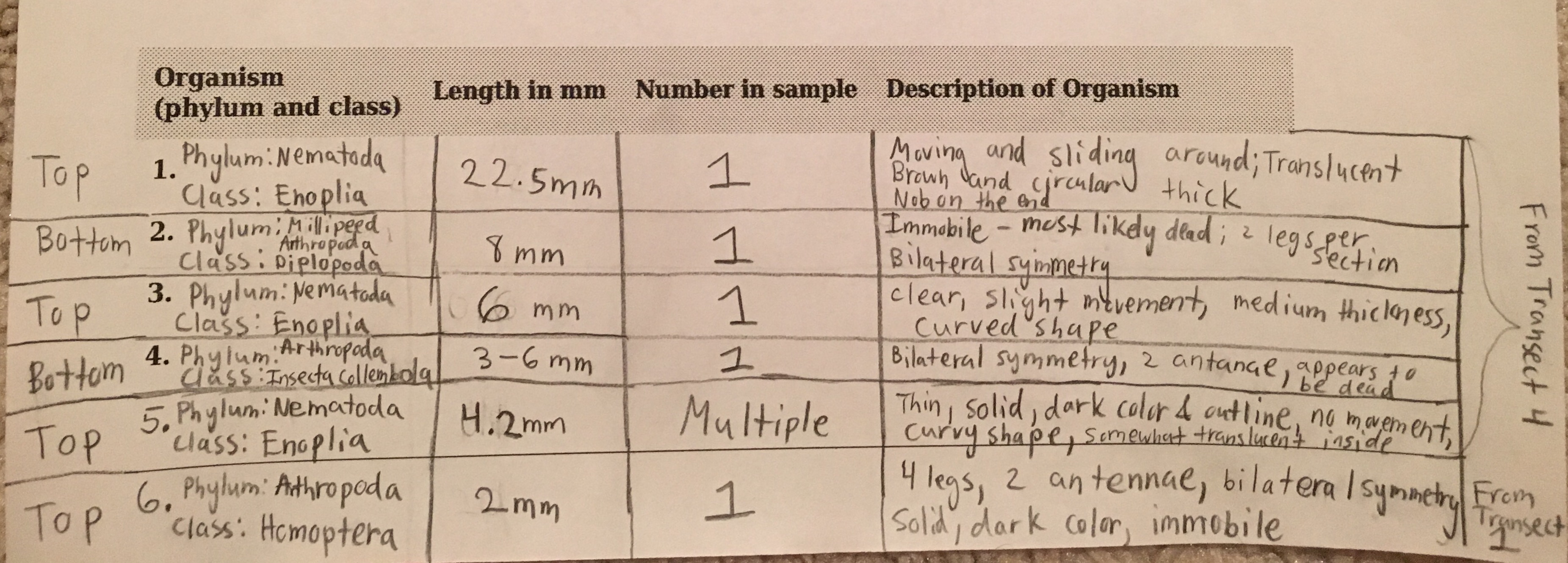
Phylum: Nematoda; Class: Enoplia

Millipede: Phylum: Arthropoda; Class: Diplopoda

Phylum: Nematoda; Class: Enoplia
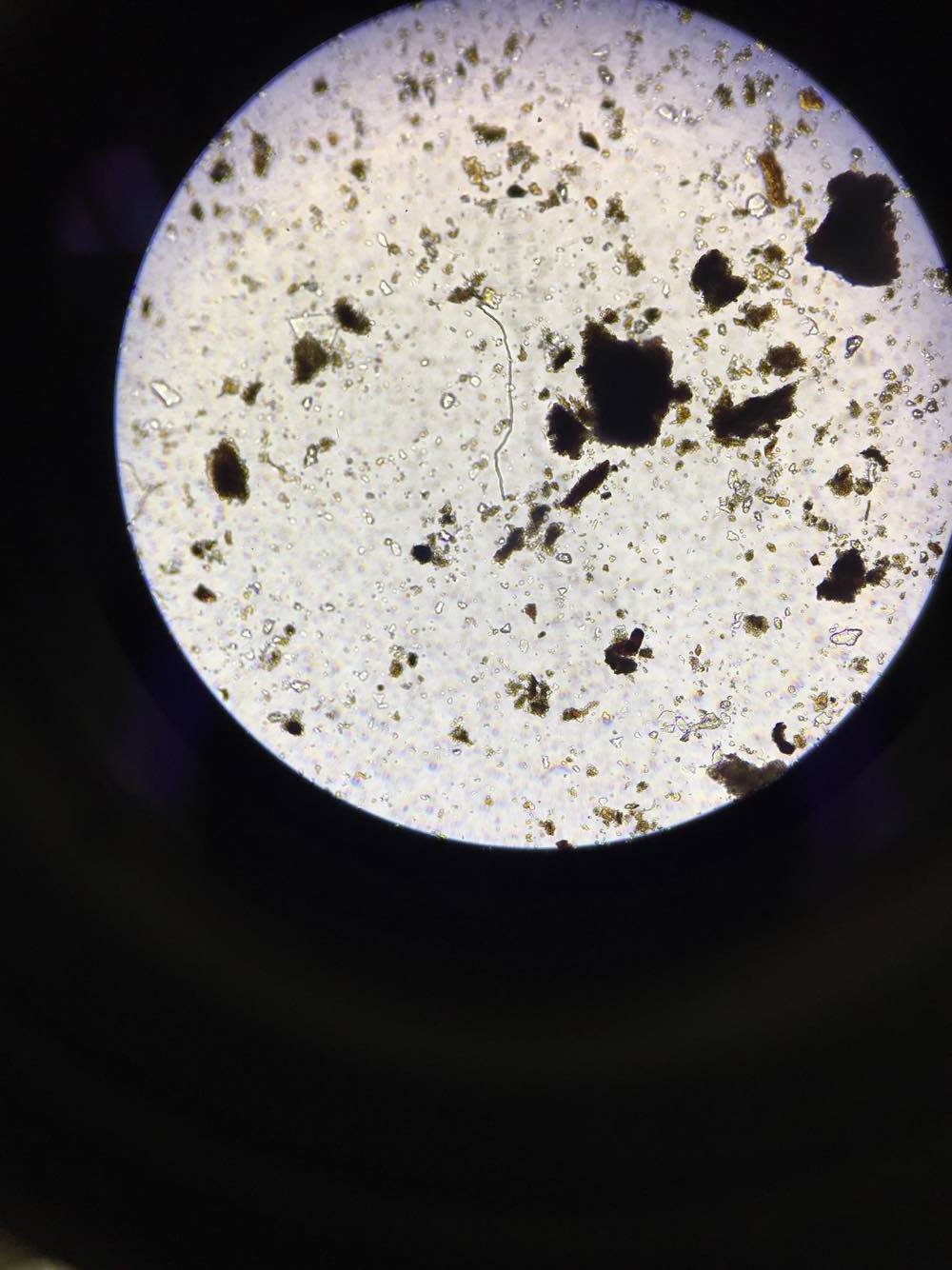
Phylum: Arthropoda; Class: Insecta Collembola

Phylum Nematoda; Class Enoplia (Multiple)
Phylum: Arthropoda; Class: Homoptera

Conclusions and Future Directions
Based on the results of this experiment, there are a large variety of invertebrates within the transect, but many of them are quite similar in that they come from the same phylum or class. This was demonstrated in how half of the invertebrates found were from Nematoda Enoplia, and how the other three were some form of arthropod.
Y.J.T.F.
2/12/16
Plantae and Fungi Lab
Purpose The purpose of the lab experiment is to collect plant samples from our transect, and to analyze their characteristics as well as try to identify what they are exactly. We had to select five different plant samples that we found in different places throughout the transect. We are performing this experiment to not only determine the different plant life in our transect, but to understand how each plays a role in the ecology of the transect. Hypothesis: Plants from the transect influence the ecological environment of the transect and the organisms that inhabit it.
Materials and Methods -Collected 5 or more plant or fungal samples from different parts of our transect -Observed the 5 plant samples that we found under the dissecting scope -Used razor blades or scissors to cut open and analyze the inside of the plant materials and observed under dissecting scope again -Identified certain characteristics such as vascularization, specialized structures, mechanisms of reproduction, shape, and size, and recorded that information in the plant characterization table -Determined if the plants were monocots or dicots using the key provided in the lab -Used a field guide from the library to try to identify what the plant samples were (Source cited down below)
Data and Observations
The five plants that were found in this transect include a dandelion leaf, a branch from a Virginia pine tree, a common milkweed, a white clover leaf, and half a walnut, as shown in Table 1. The dandelion leaf was found between one of the sprinkler heads near some tall grass, the branch from a Virginia pine tree was found near the pine/fern trees nears the back wall of the transect, the common milkweed was found near the front of the transect growing around some tall grass, the white clover leaf was found on the soil underneath the tall oak tree, and the half of a walnut shell was found near the lamp post on the western side of the transect.
Plant Identifications and Descriptions: After thoroughly analyzing the plant samples and recording the data in the Plant Characterization Table (Table 1 below), we went to the AU library and examined a field guide consisting of plants, animals, and other organisms around Washington D.C. The first identification was of the dandelion leaf. The leaf itself is much longer than it is wide, and its has rather sharp cut pointy edges. When examining the field guide, the this leaf found in the transect matched the dandelion leaf pictured and described in the guide exactly. Next, the the fern or pine branch that was found in the transect has many similar characteristics of the Virginia pine tree that was found in the field guide. There are some rather distinct differences between the branch sample found in the transect and the picture of the pine found in the field guide, but it appears that they are most likely closely related with similar short and smooth needles as well as detailed pinecone structures attached. Prior to examining the field guide in the library, we identified that the twig like plant with several smooth spherical berries was most likely a milkweed. After examining the field guide, we determined that the features of the plant found in the transect and the picture and description in the guide were very similar in their features. Regardless of whether or not this plant is a milkweed, it seems to be very closely related. The next plant sample was a very small, light green leaf that was made up of multiple round leaves and pointy leaves. After examining the field guide, there are some similarities between the leaf sample found in our transect and the white clover leaf pictured and described in the field guide. There are several distinct features between the sample and the field guide picture in terms of leaf shape and slightly different coloring, but overall it appears that perhaps the leaf we found in the transect is part of the clover family. Lastly, we identified in lab that the brown half of a shell found in the transect best resembles a walnut. We could not find this walnut in the field guide, but after asking about it in class, we determined that it most likely is some form of a walnut. It has very elaborate details on the outside shell, and it is somewhat hollow on the inside where there most likely used to be a nut or seed inside.
Plant Descriptions and Images:
Dandelion Leaf: This is the dandelion leaf that was found between one of the sprinkler heads near some tall grass in the transect.

Branch from Virginia Pine Tree: This is the Virginia pine tree branch that was found near the multiple pine/fern trees near the back wall of the transect.

Common Milkweed: This is the common milkweed that was found growing near the front of the transect surrounded by tall grass in the transect.

White Clover Leaf: This is the white clover leaf that was found underneath the tall oak tree in the transect.

Walnut: These are two pictures of the walnut shell that was found near the lamp post on the western end of the transect. The first picture is of the elaborate front of the shell, and the second is of the inside where a seed most likely used to exist.


Table 1: Plant Characterization Table

Conclusions and Future Directions After thoroughly collecting and examining the plant samples in our transect, we can conclude that there is a lot of plant diversity that makes up part of the living things in our transect. In particular we noticed several different types of weeds and wild flowers (milkweed, clover, and dandelion leaf) and a couple of plant materials fallen from the trees. These plants have the ability to reproduce throughout the transept and grow new plants, which therefore allows us to conclude from our hypothesis that the plants in our transect do indeed influence the environment of the transect.
Field Guide Source: Youth, Howard. 2014. Field Guide to the Natural World of Washington, D.C. Johns Hopkins University Press: Baltimore 285-302.
Y.J.T.F
2/5/16
Final Observations of Hay Infusion Culture: Water has evaporated, There is a thick film at the top, Minimal smell, rather earthy smell, Water is much darker, Thicker consistency, Hypothesis for change in smell/appearance: Water evaporation each week in the Hay Infusion culture causes a change in smell and appearance. Archaea potentially could have grown on our agar plates, but a reason they most likely would not is that they typically grow in extreme environments such as hot springs or at the bottom of the ocean.
Serial Dilution Results Table:
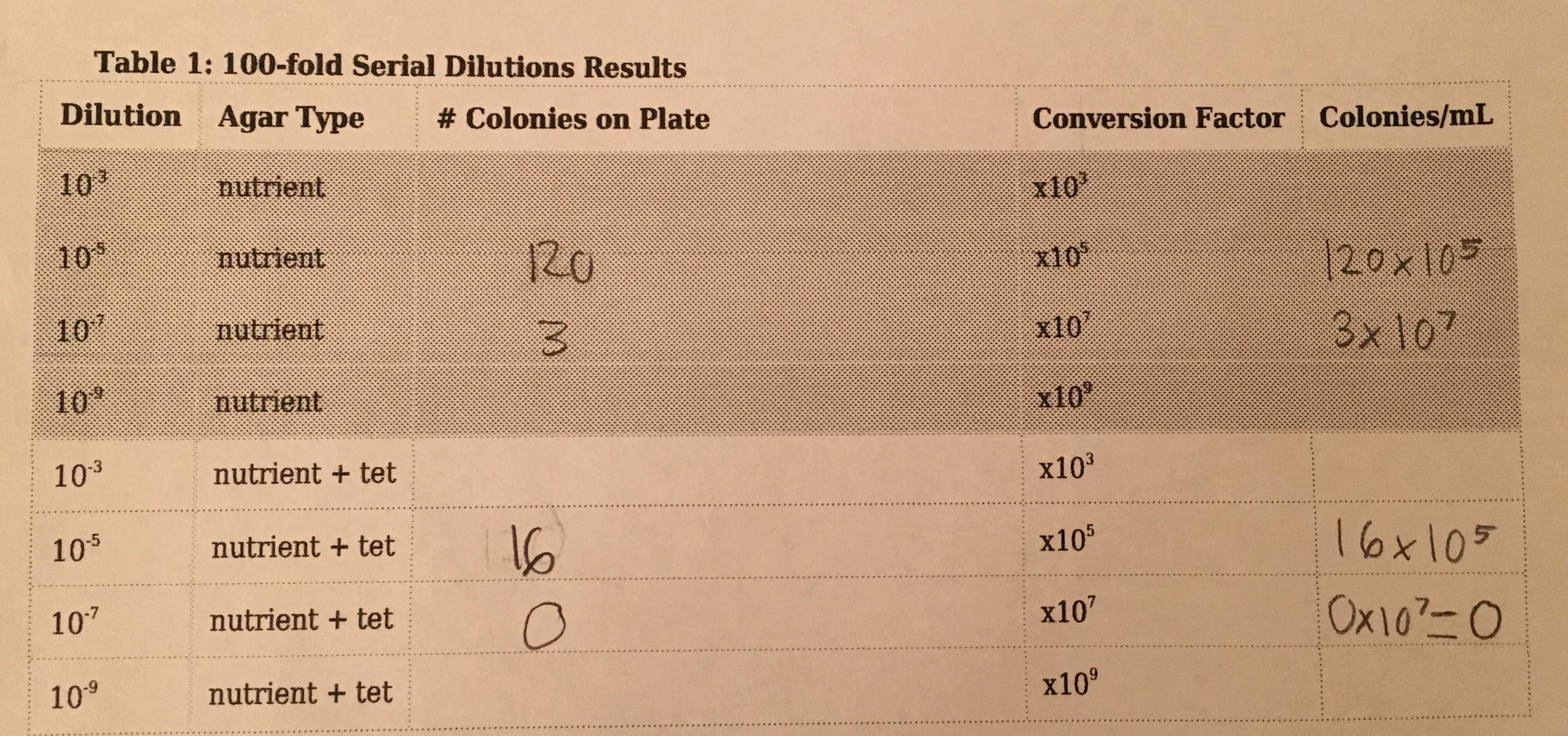
The agar plates that had tetracycline had much less bacterial growth in general compared to the agar plates without antibiotics. There were much fewer types of colonies formed on the plates that had the tetracycline, and one of these plates actually had no bacterial growth. This indicates that most of the bacteria were affected by the antibiotic, but a few species are resistant to tetracycline. There was no fungal growth on any of the plates, so we did not see the impact tetracycline has on the growth of fungi. The agar plates without tetracycline had a variety of different colors and sizes of bacterial colonies on the plates. The agar plates with tetracycline however, had about only one or two types of colonies grown, so approximately 1-2 species of bacteria are unaffected by tetracycline.
According to primary research, the mechanism of action for tetracycline is a ribosomal binding mechanism, and the types of bacteria that are sensitive to tetracycline include chlamydiae, mycoplasmas, rickettsiae, as well as protozoan parasites. (Chopra & Roberts, 2001)
Chopra, I., Roberts, M. 2001. Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiology and Molecular Biology Reviews. 65: 232-260.
Bacteria Characterization Table:

Materials and Methods The Gram Stain procedure began by sterilizing a loop with a flame from a bunsen burner, which was used to scoop some bacterial growth from the agar plate. Next the bacterial growth was smeared onto a microscope slide and a drop of water was added on top. A red wax pencil was used to draw a circle around the bacterial smear sample. With a set of tongs, the microscope slide was passed through the flame to evaporate the water on the bacterial sample. After fully cooling, a drop of Gram's iodine mordant was placed on top of the sample for one minute and then rinsed off with water. 95% alcohol was added on top for ten to twenty seconds to decolorize the area around the bacterial smear. Safranin stain was then added on top of the smear for twenty to thirty seconds and then rinsed off with water. Any excess water was removed with a kimwipe and the microscope slide was set aside to air dry. Lastly, the bacterial sample was observed under the microscope, and photographs were taken under 40x magnification. The bacteria were characterized, and this whole process was repeated for a total of four agar petri dishes.
For the DNA isolation and PCR amplification process, bacterial colony growth was scooped out of two agar plates with sterile toothpicks. One agar plate had tetracycline, and the other did not. 20μL of primer mixture was added to two PCR tubes and were mixed thoroughly to dissolve the beads. The two bacterial colonies were then placed in the PCR mixtures of separate tubes, and were placed in the PCR machine. The process was performed to amplify the 16S rRNA gene and allow the bacterial colonies to be further characterized.
Pictures of Colonies:
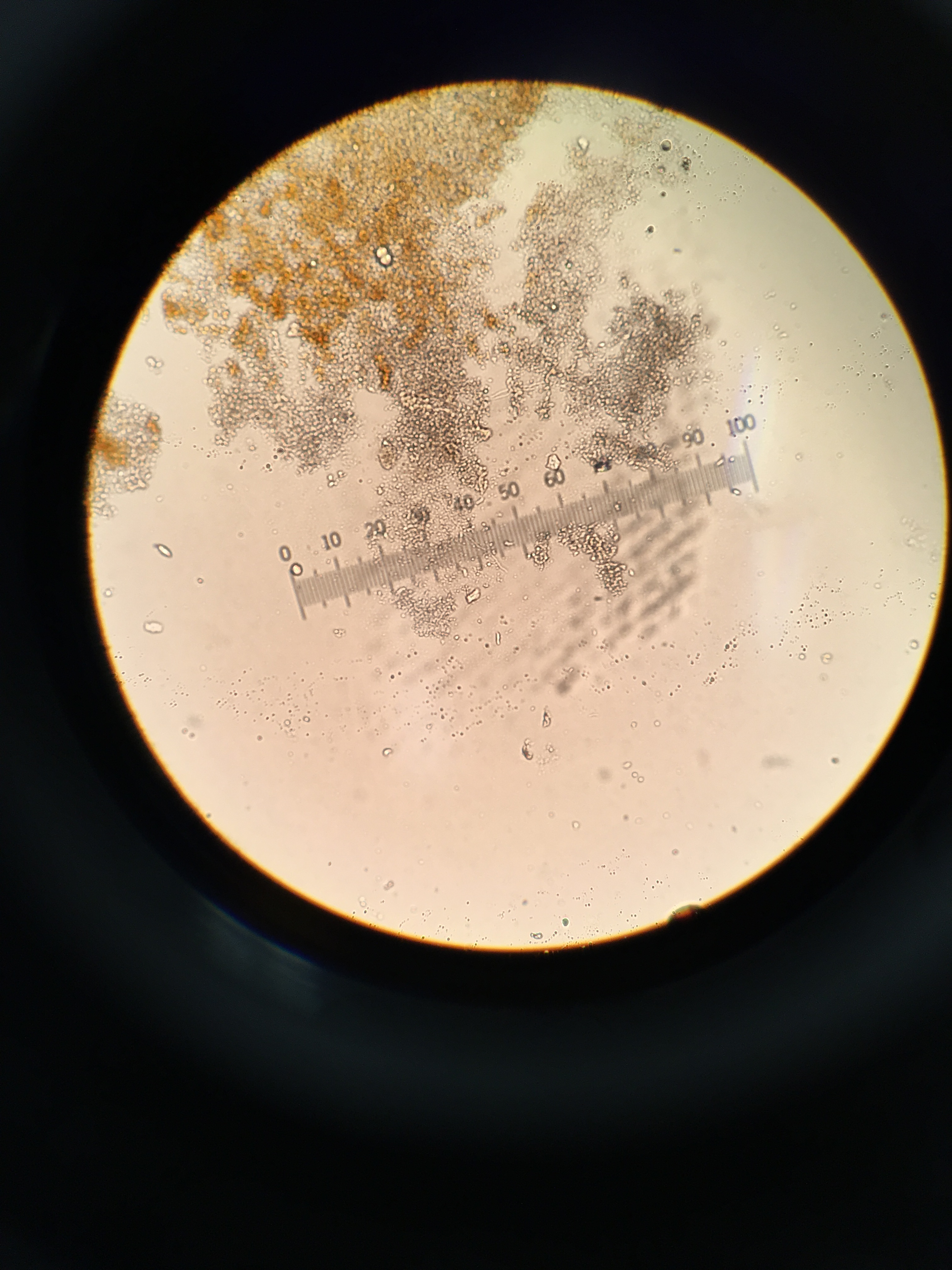
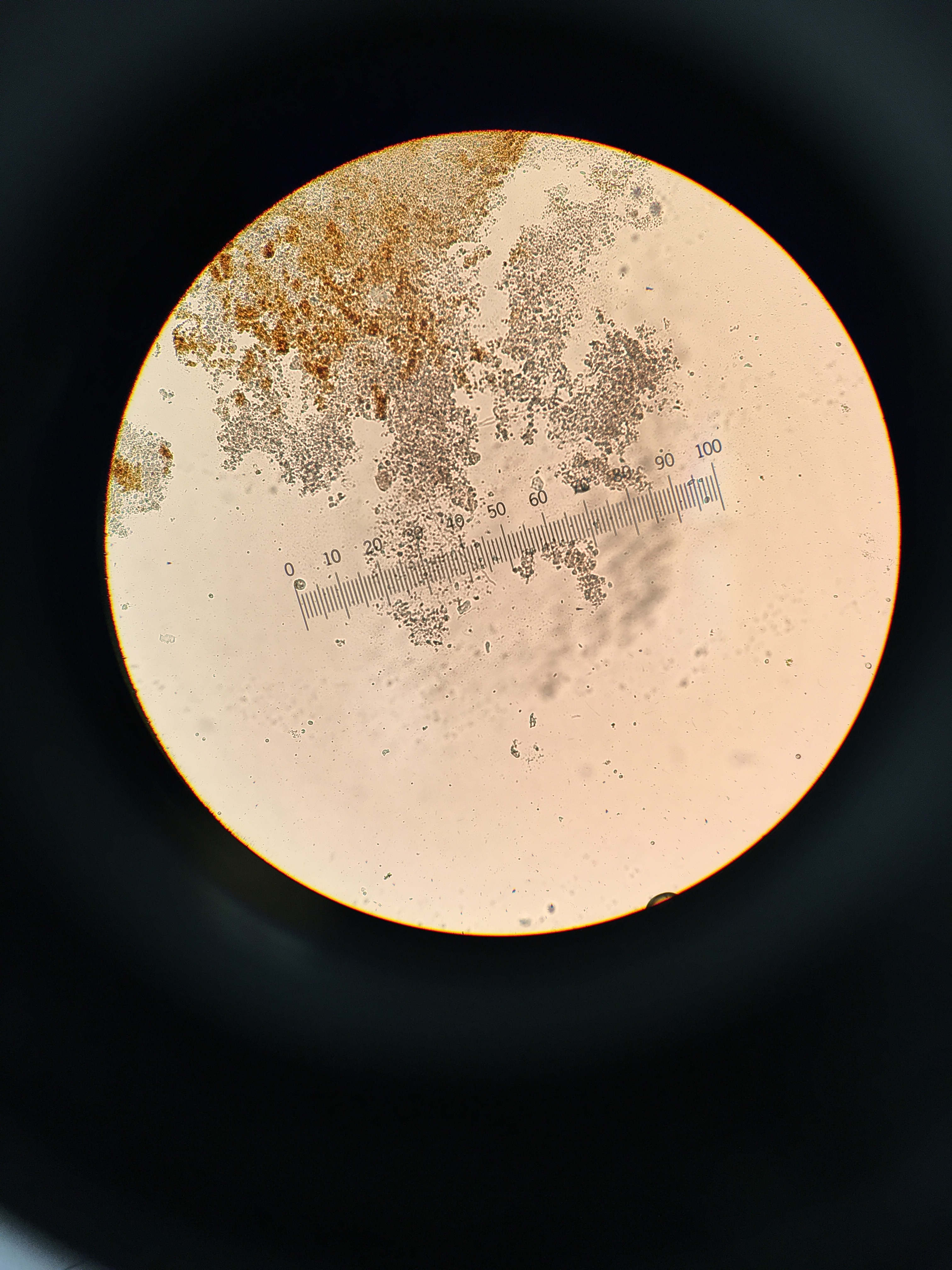
Figure 1. Gram Stain 10-5 tet (-); 40x
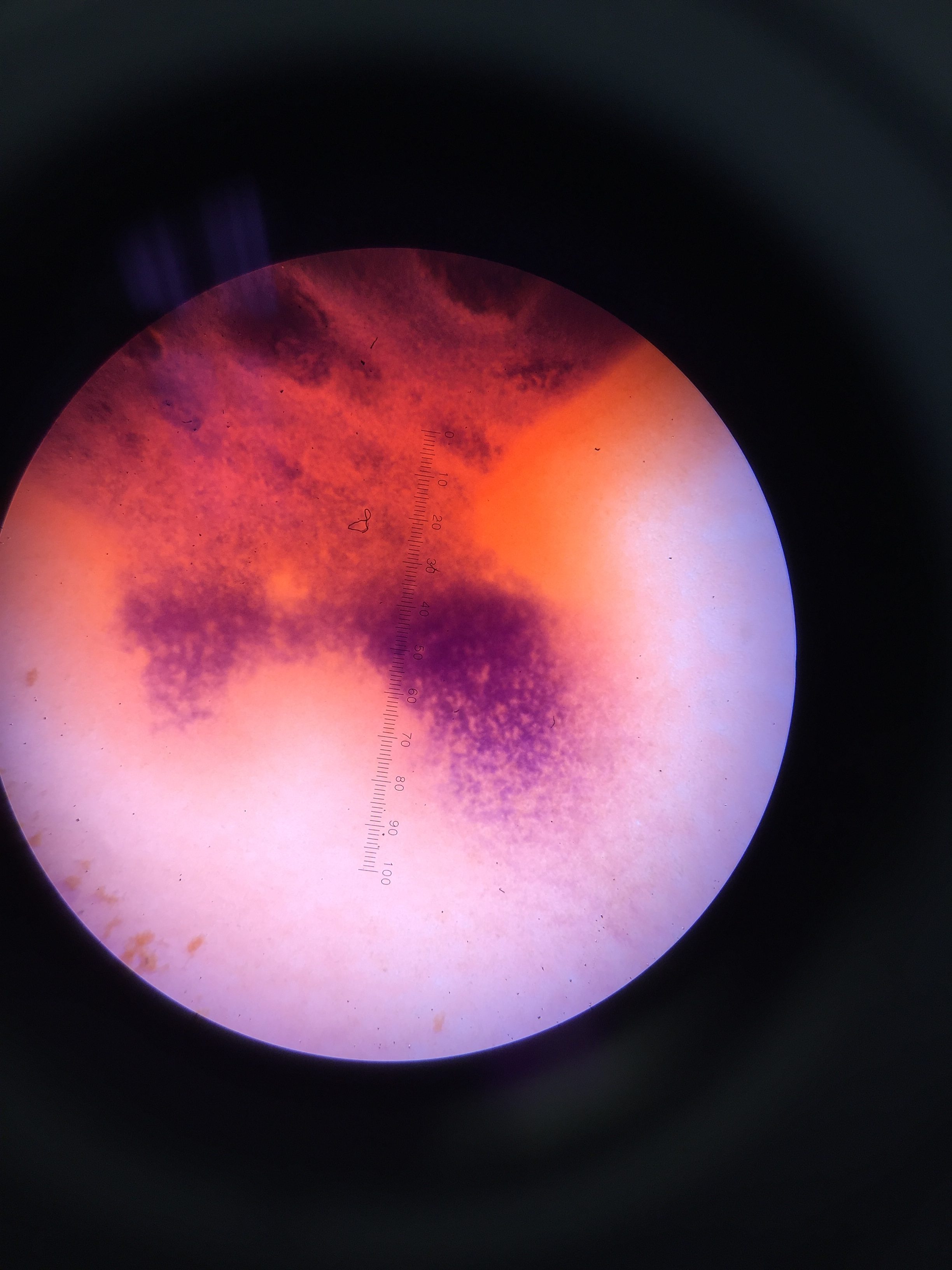
Figure 2. Gram Stain 10-5 tet (+); 40x
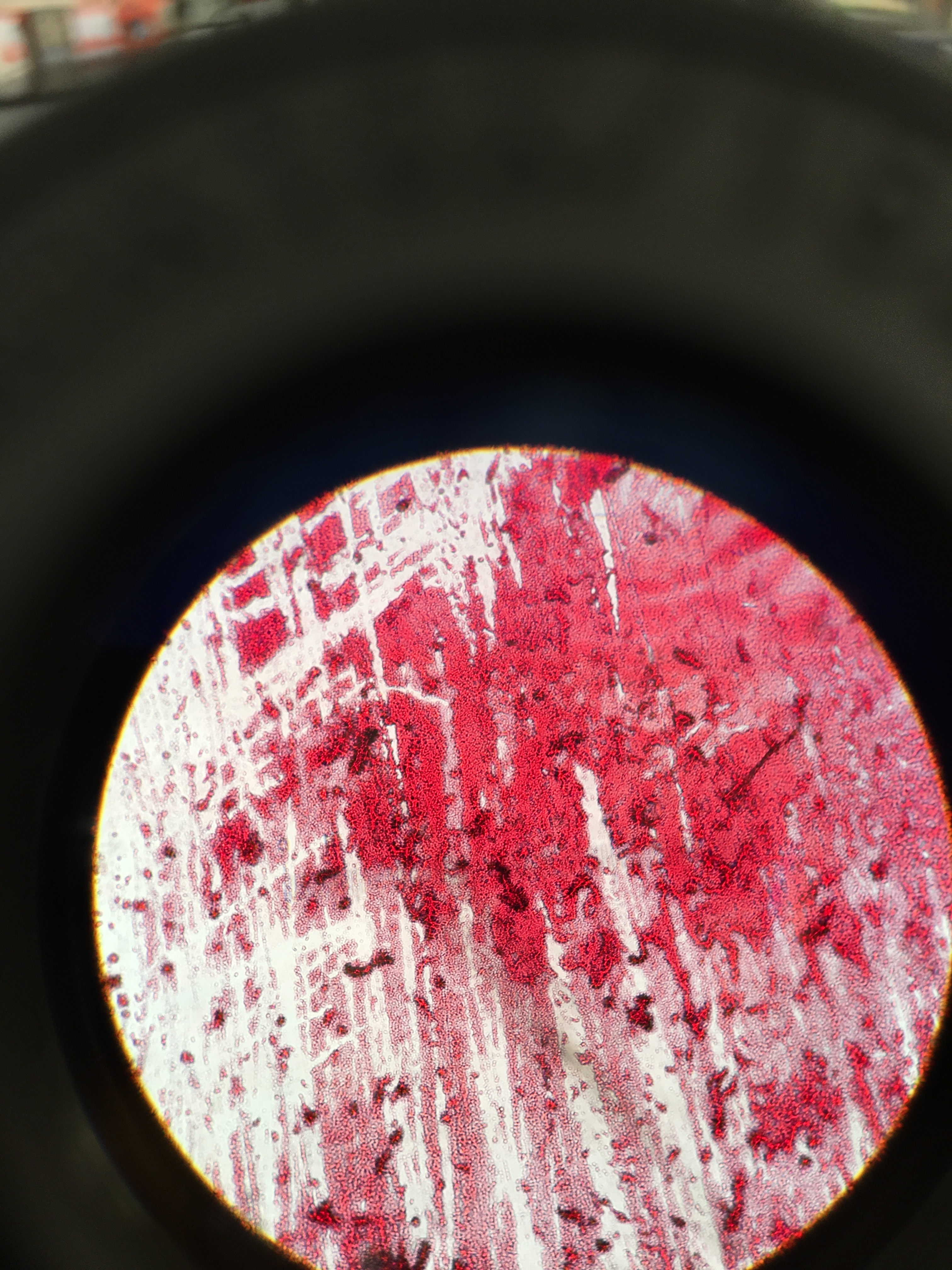
Figure 3. Gram Stain 10-7 tet (-); 40x
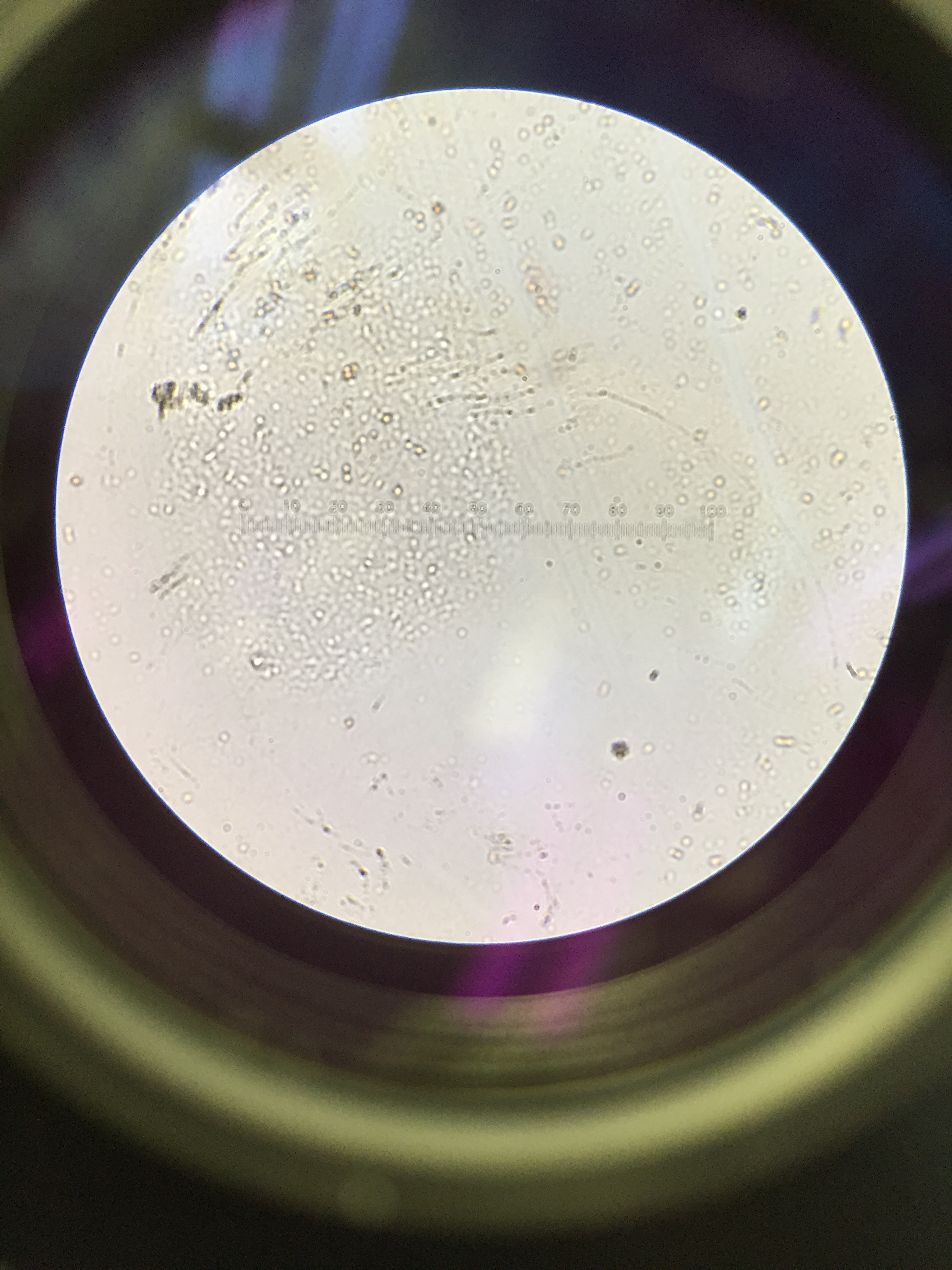
Figure 4. Gram Stain 10-7 tet (+); 40x
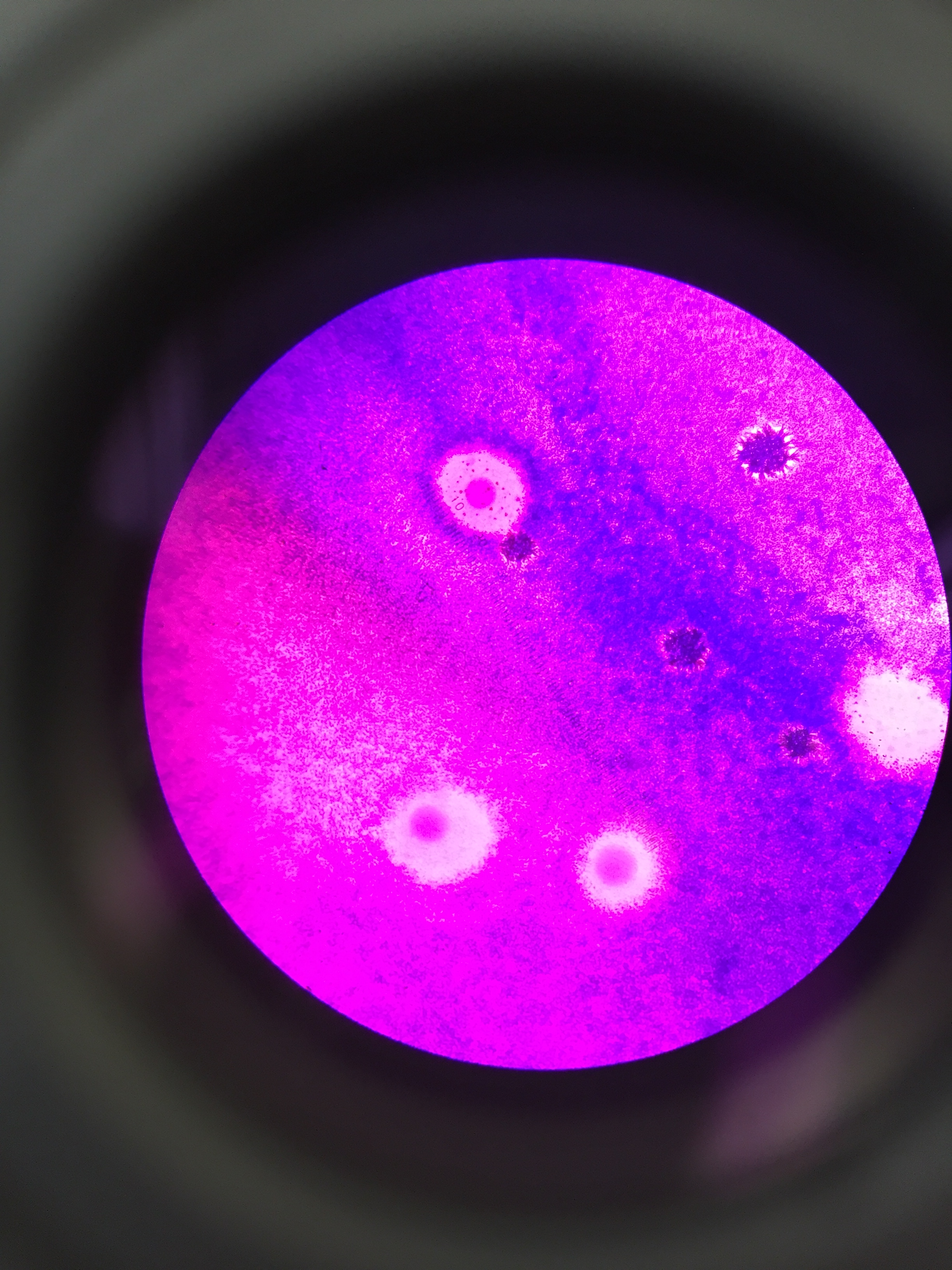
Figure 5. No Gram Stain 10-5 tet (+); 40x
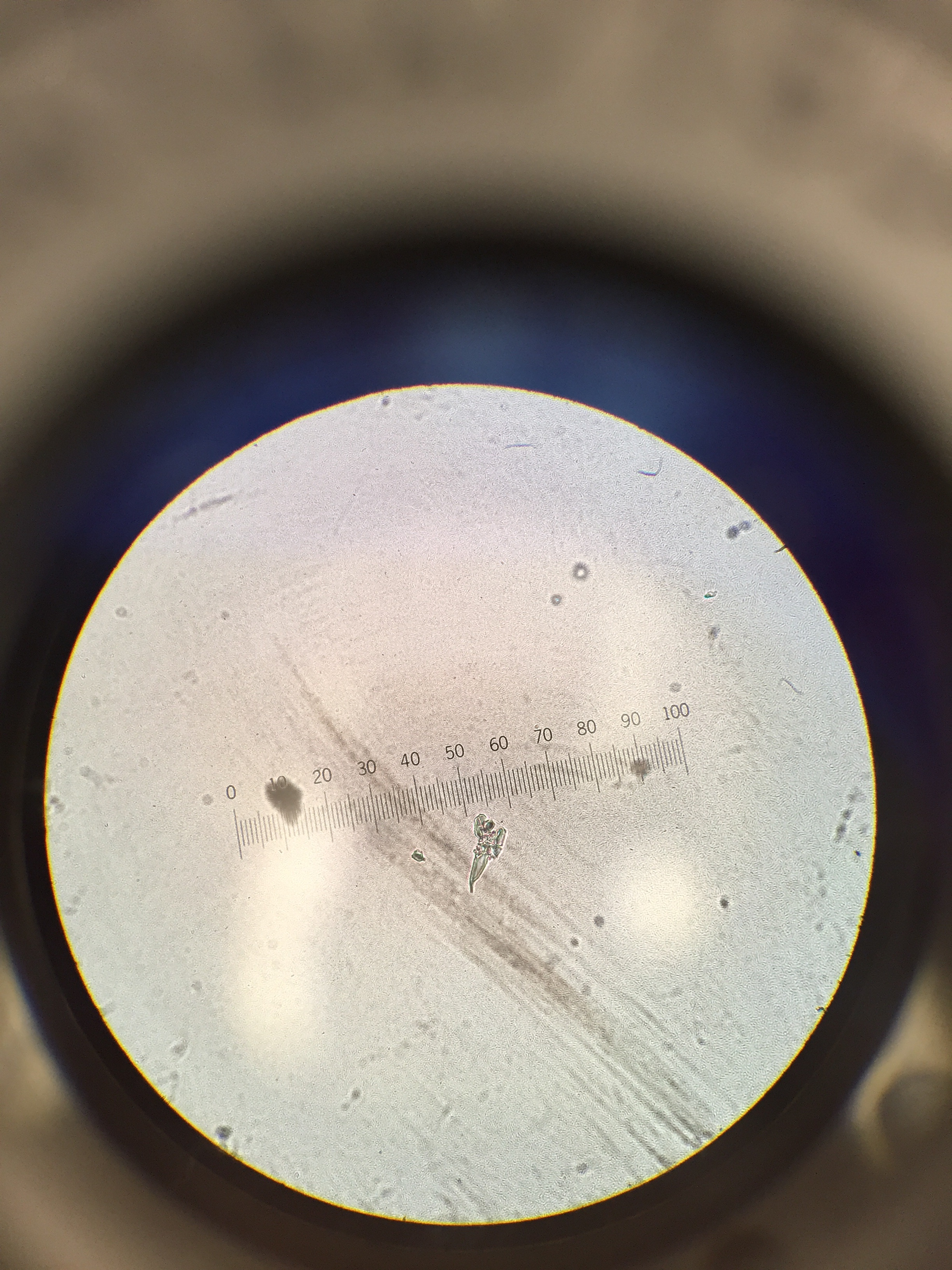
Figure 6. No Gram Stain 10-7 tet (-); 40x

Y.J.T.F
1/29/16
Description of Hay Infusion and Initial Observations: The Hay Infusion culture was filled with fallen leaves, acorns, small pine cones, twigs and smaller branches, and some soil. After adding the deerpark water and the dried milk and waiting one weak, the culture contained a very brown colored solution inside. The collected biotic materials from the transect are distributed unevenly throughout the culture. There was a rather sulfuric or rotten egg like smell in the culture, but also a rather earthy smell. There were no green shoots on top of the liquid, but there appeared to be some mold growth and possibly the beginnings of fungal growth. The mold and fungus will probably grow much more overtime if we let the culture remain.
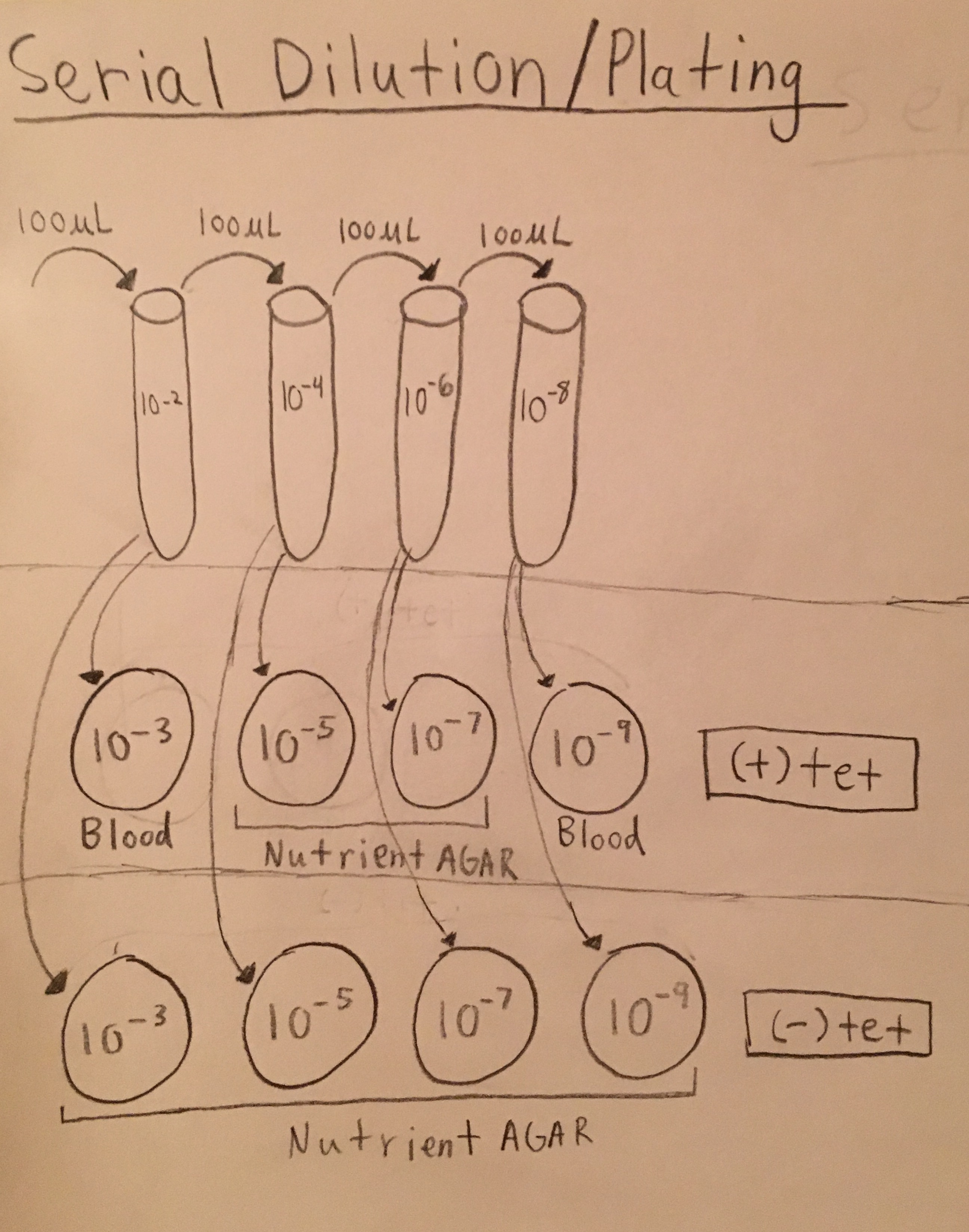
Serial Dilution/Plating Description: Four tubes were filled with 10mL of broth. The four tubes were labeled: 10-2, 10-4, 10-6, 10-8. 100μL of solution from the Hay Infusion Culture were pipetted into the 10-2 tube, then 100μl from 10-2 solution pipetted into the 10-4 tube, 100μL from 10-4 solution pipetted into the 10-6 tube, and finally 100μL from 10-6 pipetted into the 10-8 tube, as shown in Figure 1 below. Next the 100μL from each tube were placed on two sets of four petri dishes. The first set of plates contained tetracycline, and two plates were filled with blood while the other two were filled with nutrient agar. The other set of plates each contained nutrient agar but did not contain tetracycline. The plates were labeled: 10-3, 10-5, 10-7, 10-9. In both sets of plates, 100μL from the 10-2 tube was pipetted onto the 10-3 plate, 100μL from 10-4 tube was pipetted onto the 10-5 plate, 100μL from 10-6 tube was pipetted onto 10-7 plate, and 100μL from 10-8 tube was pipetted onto 10-9 plate, as shown in Figure 1 below. The petri dishes were set aside for one week to observe the bacterial growth.
Serial Dilution/Plating Diagram - Figure 1:
Niche Samples: The samples used for making wet mounts came from the Top Zone and the Middle Zone
 Depending on whether the organisms are algae or protists will determine whether or not they will be close to or away from plant matter. Algae has the ability to make its own food through photosynthesis, so it may not necessarily be found near plant matter. Protists however do not perform photosynthesis but they need to consume nutrients. They need a food source which is why they will most likely be found near plant matter.
Depending on whether the organisms are algae or protists will determine whether or not they will be close to or away from plant matter. Algae has the ability to make its own food through photosynthesis, so it may not necessarily be found near plant matter. Protists however do not perform photosynthesis but they need to consume nutrients. They need a food source which is why they will most likely be found near plant matter.
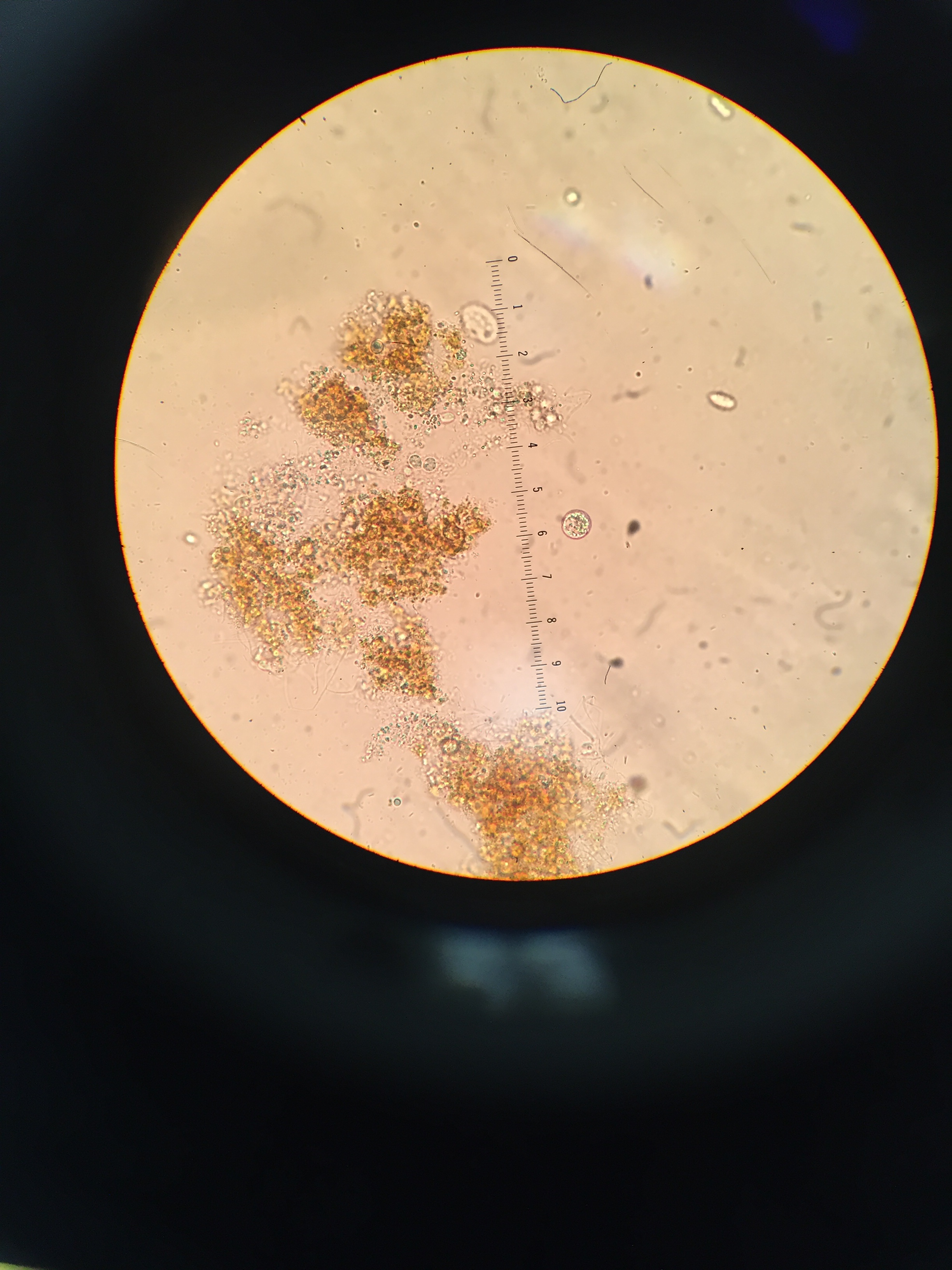
Protists and Algae Found in Sample: Top Zone: Gonium: 30μm in diameter; Green in color; flat; not completely round, rather curvy edges; non-motile; algae; photosynthesizing Pandorina: ~25μm in diameter, colorless; appears to be 16 cells inside; quite round in shape; flat; non-motile; algae; photosynthesizing Peranema sp.: 40μm in length, colorless; appears to be one flagellum; very oval like in shape; motile, protist; not photosynthesizing
Middle Zone:
Paramecium: 210μm in length; colorless; flat; quite large compared to other organisms found; oval like in shape; non-motile, protist; not photosynthesizing
Eudorina:12μm in diameter; slight coloring; flat; smaller compared to other organisms identified; non-motile; algae; photosynthesizing
Euglena: 40μm in length; Greenish color; round, oval like in shape; flat; non-motile; protist; not photosynthesizing
Images of Protists and Algae Under Microscope:
Top Zone:
Meeting All the Needs of Life (based on page 2 of Freeman text)
Organism: Gonium
Energy: Gonium is a form of algae that has the ability to produce the energy necessary to survive by photosynthesizing
Cells: As seen under the microscope, Gonium organism that forms colonies of multiple green cells
Information: Gonium is going to respond to the information if finds in its environment. For example, in the Hay Infusion Culture, the Gonium may avoid being surrounding by leaves and other biotic material that may conceal it from natural light. It wants to position itself so that it can get sunlight to be able to photosynthesize.
Replication: Gonium is isogamous and sexually reproduces offspring that is morphologically similar to the parent cell.
Evolution: The textbook explains that the organisms we have today are a result of evolution, so therefore Gonium has evolved into the algae that it is today, and may continue to evolve overtime to adapt to potential influences such as the environment.
2 Months Later If we let the Hay Infusion Culture grow for another two months, there would probably be significant mold growth and fungal growth especially in the top zone niche. Some of the biotic materials such as the leaves may be very soggy or broken down due to being in liquid for a longer period of time. This may result in leaf matter or other biotic material moving around and forming a gunk like material at the top of the culture. These selective pressures may influence the communities of the sample based on how the organisms react to mold and fungus, and also many protists may have to change location to get nutrients from the plants that have moved location, and then the algae may move around as well away from the plants as they photosynthesis for nutrients.
Y.J.T.F
1/22/16
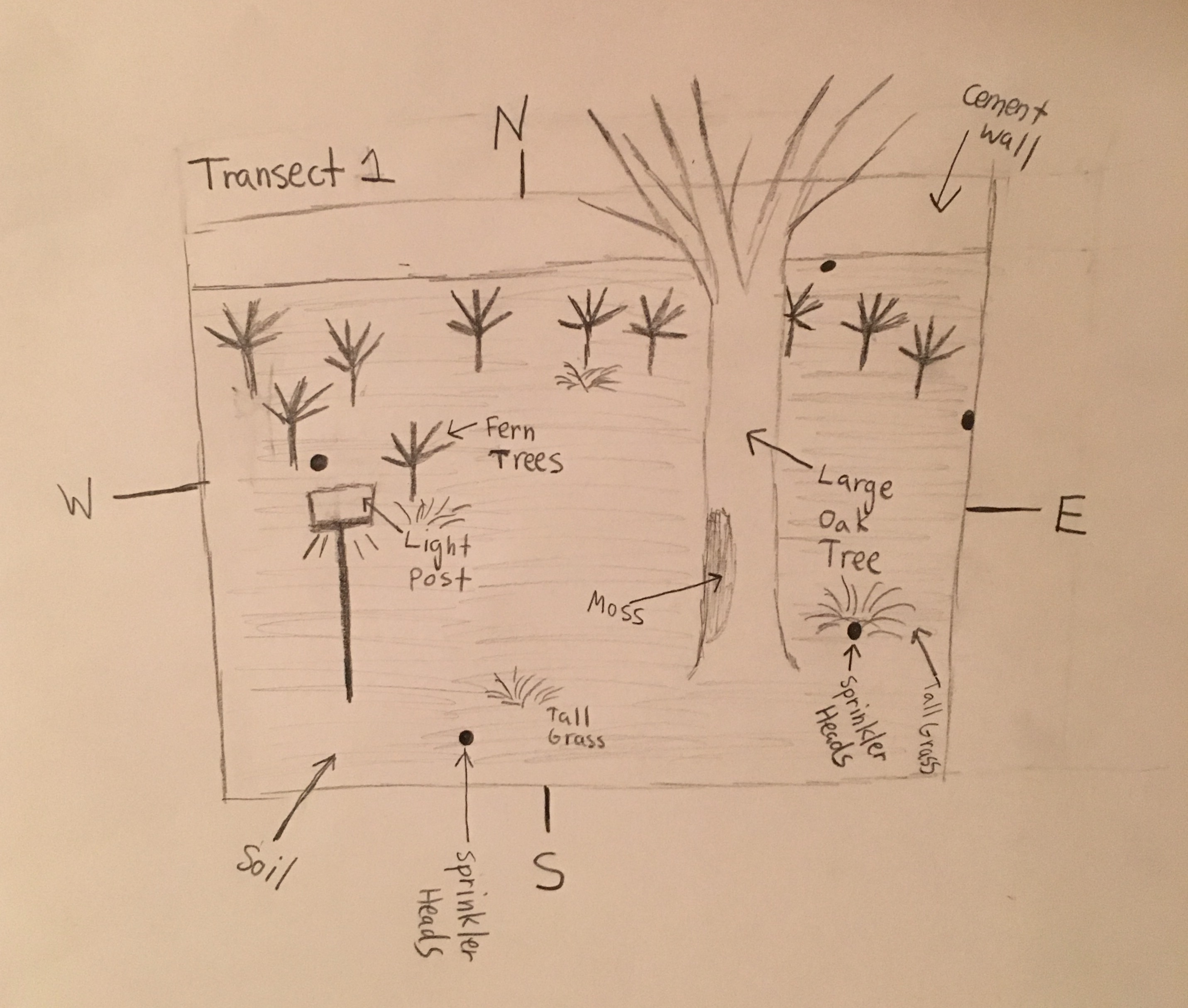
Abiotic Factors: Light Post, Soil, Sprinkler Heads
Biotic Factors: Large Oak Tree, Smaller Fern Trees, Tall Grass, Fallen Leaves and Acorns, Fallen Oak and Fern Tree Branches, Squirrel, Moss
Description: The aerial diagram above shows the basic layout of Transect 1, which is a large soil plot consisting of a tall oak tree, ten smaller fern trees, small scattered sections of tall grass, a light post, as well as several sprinklers throughout the plot. Scattered all around the transect there are fallen branches/twigs, leaves, and acorns from the oak tree, and some fallen branches from the fern trees. There is a bit off moss grown on the trunk of the oak tree, and a squirrel was seen eating fallen acorns that surround the oak tree. Just outside the north border of the transect there is a cement wall.
Y.J.T.F