User:Xing Zhang
Contact Info

- Xing Zhang (John), PhD candidate
- Department of Biomedical Engineering, University of Alabama at Birmingham (UAB), Birmingham, AL, USA.
- Mailing Address: CBSE 100, 1530 3rd Ave. S., Birmingham, AL 35294
- E-mail: xzchina at uab.edu
Objective
Currently seeking a post-doc/research associate position in a laboratory of tissue engineering or biomaterails.
Highly experienced in cardiovascular tissue engineering, including scaffolding, physical and biomedical characterizations, and engineering of a functional tissue.
Education
- 2010, PhD (expected) in Biomedical Engineering (cardiovascular tissue regeneration), University of Alabama at Birmingham (UAB), AL, US
- 2008, M.S in Biomedical Engineering (cardiovascular mechanics), University of Alabama at Birmingham (UAB), AL, US
- 2006, B.S in Biological Sciences, Wuhan University, Hubei, China
- 2005, B.Eng in Materials Sciences, Wuhan University of Technology, Hubei, China
Research interests
My research interests primarily center around the regeneration of vascular tissues (blood vessels), ranging from the fabrication of prosthetic scaffold, cell interaction with the scaffolding materials, the development of a novel bioreactor and quantitative analysis of tissue formation process. I am also interested in the biological mechanism of angiogenesis, the formation and differentiation of blood vessels.
Scaffolding
To fully regenerate a functional tissue and ultimately an organ, a prosthetic scaffold structurally mimicking the native extracellular matrix (ECM) is required to provide an appropriate temporal micro-environment for the tissue to re-grow. A satisfactory scaffold should possess comparable properties typically associated with the ECM, such as compliance, tensile stress/strain, porous structure, degradation and remodeling profiles.
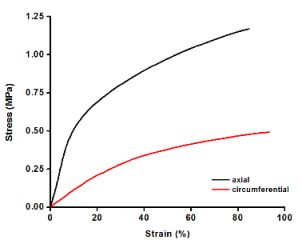
In my research, electrospinning is employed to fabricate a highly porous multi-layered composite scaffold (porosity~80%) composed on nano-fibers (fiber diameter~600nm). Physical characterizations show that this scaffold possess sufficient mechanical strength as an arterial substitute and suitable micro-structure for cellular adhesion and motility.
Details can be found in (2) in Journal Publications.
Fig. 1 From left to right: The digital image of the tubular scaffold (ID=4 mm); SEM images of the inner layer; SEM images of the outer layer; The uni-axial stress-strain curves
Regeneration of a Human Endothelium
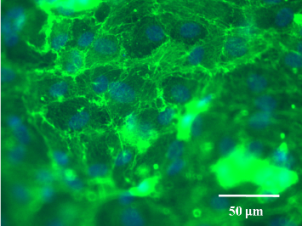
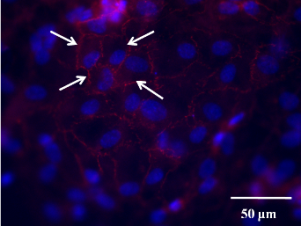
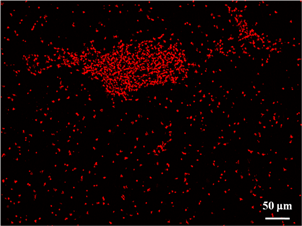
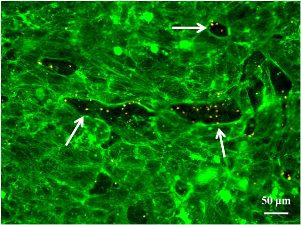
After the successful construction of an ECM-like scaffold, primary human aortic endothelial cells (HAECs) were used to evaluate the scaffold's biocompatibility and its capacity to support the regeneration of a functional endothelium. Our results show that HAECs could easily adhere to the scaffold's luminal surface and form a confluent mono-layer within 24 hr, given an appropriate seeding concentration. In addition, tight junctions between adjacent HAECs were detected within the same time window. This endothelial mono-layer almost completely prevented human platelet adhesion while the bare scaffold induced widespread platelet adhesion and aggregation.
Fig.3 From left to right: HAECs confluent mono-layer; Tight junctions (white arrows) between adjacent HAECs; Platelet adhesion and aggregation on bare scaffold; Platelet adhesion on exposed scaffold (white arrows) in an endothelial mono-layer. No platelet adhesion/aggregation on HAECs mono-layer.
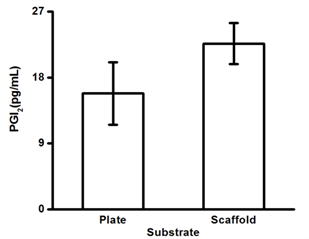
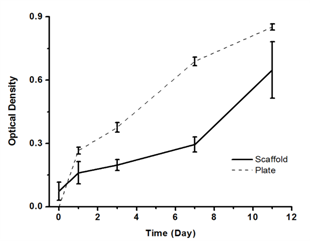
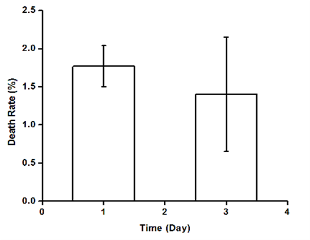
Furthermore, we determined that HAECs were able to produce prostacyclin, a multi-functional factors secreted by native endothelium to prevent thrombosis. The proliferation profile shows that HAECs possessed a strong growth capacity and that the degraded scaffolding materials did not jeopardize the viability of HAECs. The death rates of HAECs within initial 3 days were extremely low, exhibiting no increasing trend.
Fig.4 From left to right: prostacyclin secretions of HAECs on scaffolds and tissue culture plates (positive control); Proliferation profiles of HAECs on scaffolds and tissue culture plates (positive control); Death rates of HAECs on scaffolds within 3 days after seeding.
Details can be found in (1) in Journal Publications.
Journal Publications
<biblio>
- under_review Zhang X, Thomas V, Xu Y, Bellis S, Vohra YK. The in vitro regeneration of a functional human endothelial on an composite nano-fibrous electrospun scaffold.
- paper2 pmid=19902335
- paper3 pmid=19575442
- paper4 pmid=18780360
- paper5 pmid=18458479