User:Whitney Morales/Notebook/Biology 210 at AU
February 27, 2014
DNA sequencing from lab 3 (microbiology)
M3-T5-1
GCTACTCTCACGAGAGTAGGTTTATCCCTATACAAAAGAAGTTTACAACCCATAGGGCCGTCGTCCTTCACGCGGGATGGCTGGATCAGGCTCTCACCCATTGTCCAATATTCCTCACTGCTGCCTCCCGTAGGAGTCTGGTCCGTGTCTCAGTACCAGTGTGGGGGATCACCCTCTCAGGCCCCCTAAAGATCGCAGACTTGGTGAGCCGTTACCTCACCAACTATCTAATCTTGCGCGTGCCCATCTCTATCCACCGGAGTTTTCAATACCGAATGATGCCATCCAGTATATTATGGGGTATTAATCTTCCTTTCGAAAGGCTATCCCCCAGATAAAGGCAGGTTGCACACGTGTTCCGCACCCGTACGCCGCTCTCAAGATTCCGAAGAATCTCTACCGCTCGG
- Genus: Chryseobacterium Species: iMTI10 - a genus of Gram-negative bacteria
Lab 6- Embryology & Zebrafish Development
First time (2/20/14)
Procedure 1. Observe zebrafish embryos carefully and determine their developmental stage. 2. Set up the control group (water treated) and the test group (nicotine treated) in covered petri dishes. 3. Use 20 mls of Deerpark water and 20 healthy translucent embryos per dish. Use a dropper to transfer the eggs to the dishes with the appropriate water. 4. Count the number: of dead eggs, living embryos still in egg cases, number of living hatchlings (larvae), number of dead hatchlings.
Control Embryos (1-20)---Development stages (by the hour)
1. 16 cell (1.5 h) 11. 64 cell (2h)
2. germ ring (5.7h) 12 75% epiboly (8h)
3.shield (6h) 13.18-somite (18h)
4. 14-somite (16h) 14. 18-somite (18h)
5. 14-somite (16h) 15.germ ring (5.7h)
6. 18-somite (18h) 16. germ ring (5.7h)
7. oblong (3.7h) 17. 14-somite (16h)
8. 10-somite (14h) 18. 30% epiboly (4.7h)
9.dome (4.3h) 19. bud (10h)
10. 30% epiboly (4.7h) 20. 90% epiboly (9h)
Due to the zebrafish embryos treated in regular water (control) opposed to nicotine, the following day (2/28/14) upon observation it’s safe to assert that normal development occurred within those 24 hours; therefore 24hours were added to each embryos development stage taken the prior day.
- Observations of embryo development were made using microscope.
Second time (2/27/14)
Control Embryos (1-20)---Development stages (by the hour) 1. 60+ hours “pec fin stage” 11. 60+ hours “pec fin stage” 2. 60+ hours “pec fin stage” 12 60+ hours “pec fin stage” 3. 60+ hours “pec fin stage” 13. 60+ hours “pec fin stage” 4. 60+ hours “pec fin stage” 14. 60+ hours “pec fin stage” 5. 60+ hours “pec fin stage” 15. 60+ hours “pec fin stage” 6. 60+ hours “pec fin stage” 16. 60+ hours “pec fin stage” 7. 60+ hours “pec fin stage” 17. 60+ hours “pec fin stage” 8. 60+ hours “pec fin stage” (bent tail) 18. 60+ hours “pec fin stage” 9. 60+ hours “pec fin stage” 19. 60+ hours “pec fin stage” 10. 60+ hours “pec fin stage” 20. deceased (still in larva)
The week following the initial observations of the zebrafish embryo development, on average the development stage was 60 hours “pec fin stage”. The only exception was the lone larva un-hatched due to the fact that the zebrafish embryo had deceased; making the survival rate 19/20 of the initial count. Another observation made, was the bent tail of one of the hatchlings. All of the other hatchlings had a straight long tail while this one’s tail was bent, it was still able to swim at the rate of the other fish, but it was peculiar to see that it had this morphological defect being that it’s part of the control group (water).
Along with observing the hatchlins’ further development, we had a water changed. We removed 5 mls of old water and replaced it with 5 mls of new water. We also “sacrificed" two of the remaining 19 fish fixing them in 0.2% tricaine solution in order to anesthetize them. 4% formaldehyde was then transferred to the .2% tricaine solution. This was done by simply using a dropper to suck up the two hatchlings into a small glass case filled 25% with .2% tricaine solution and then the later addition of 4% formaldehyde. Next week 3/6, we will observe these anesthetized fish to view its morphology in greater detail.
February 20, 2014
Lab 5- Invertebrates
I. Observing Acoelemates, Pseudocoelamtes and Coelmates
- Under the microscope, the movements of Planaria, Nematodes and Annelida were observed and how their movement relates to their body structure.
Planaria (Acoelemate): Have an enclosed body cavity. Slow movement, glides along its belly. They move by way of having cilia, slime or even rhythmical muscle spasms.
Nematodes (Pseduocolemate): Have an enclosed body cavity partially lined with mesoderm. Move a little faster than planaria because of its lightly lined coelem of mesoderm.
Annelida (Coelemate): Have an enclosed body cavity completely filled with mesoderm. Because the annelida has a coelem lined with mesoderm, this means it has the ability of muscle function therefore faster movement than that of its counterpart planria because it lacks mesoderm. Annelida kind of had this contracting and elongating movement.
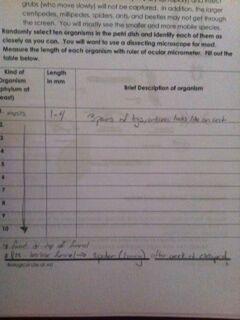
II. Analyzing the invertebrates collected with the Berlese Funnel
After randomly selecting ten organisms in the petri dish collected from the Berlese Funnel a week ago, the organisms were identified as closely as we can under the microscope. In our case the was only one type of organism found in our petri dish but surprisingly ten individuals of that species. That organism found fits the description of Springtail X (primitive insect). We measured the length of each individual with the ruler of ocular micrometer.
III. Vertebrates and Niches
Vertebrates that may inhabit and pass through our prairie transect observed by their droppings, footprints etc., are:
1. Squirrels 2. Beetles 3. Spiders 4. baby pigeon chic 5. sparrow
February 6, 2014
Lab 4- Plantae and Fungi
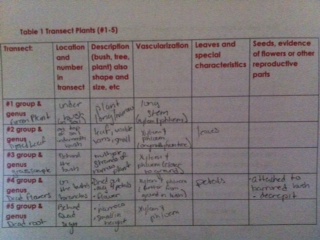
I. Collecting five plant samples from the transect
1. Our prairie transect, we collected about 500g of litter into a zip lock bag. This sample will be used to set up a Berlese funnel for collecting invertebrates next week (2/20). 2. Take representative samples from five plants to represent the plant diversity of the transect.
II. Plant Vascularization - - Majority of the plants on the prairie transect were terrestrial plants containing vascular tissue (roots_ to carry water and nutrients from the soil to the leaves. They also contained rhizoids (hair like projections) which helps them anchor to the ground enabling them to grow and spread along the ground. Most of our transect plants contain stems that serve as both as a support factor and means to transport water and nutrients to all parts of the plant. Within the stem there are xylem and phloem layers specifically responsible for for the transport of water and nutrients in an up and down motion.
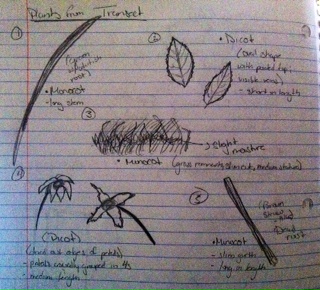
III & IV. Plant Specialization
Here is a diagram briefly describing the shape size and cluster arrangement of the leaves from the transect plants along with identifying their seeds as monocot or dicot.
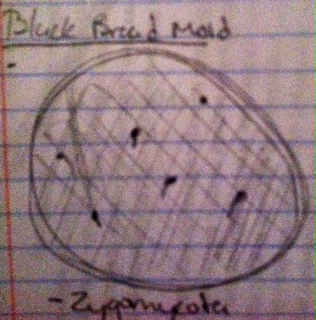
V. Observing Fungi
- Fungi sporangia is a fungal species containing spores. It can be composed of a single cell or can be multicellular. Sporangia can produce spores by process of mitosis, but in majority of many fungi, sporangia are the site of meiosis and can produce genetically distinct haploid spores.
VI. Setting up the Berlese Funnel to Collect Invertebrates
For next week’s lab, we will observe the invertebrates that reside in our prairie transect using the Berlese Funnel method.
Procedure:
1. Pour about 25 ml of the 50:50 ethanol/water solution into the flask 2. Fit a piece of the screening material into the bottom of the funnel. Tape the sides of the screen if necessary, so that leaf litter doesn’t fall into the preservative. 3. Place the funnel into the neck of the square-sided bottle. 4. Carefully put the leaf litter sample in the top of the funnel. Place a lighted 40 watt lamp above the funnel with the incandescent bulb about 1-2 inches from the top of the leaf litter. Cover everything with foil. 5. Leave the lighted setup on the lab bench for a week.
Set-up of Berlese Funnel (Picture).
•Note: When pouring the soil contents into the funnel, we found a live spider that burrowed its way under the leaves. An invertebrate that would have been interesting to observe the following week after having been preserved for a week, it sadly escaped.
January 30, 2014 (Lab 3)
Water level in hay infusion culture slightly decreased, possibly due to evaporation. Smell wasn't as repugnant as it the week prior probably due to the lack of nutrients in the jar compared to their initial habitat. Water albeit its brownish tint appeared a little more translucent. The three niches as described in Lab 2, soil based bottom, liquid water middle and film like layer on surface, remained in tact with the exception that the film layer top disintegrated and therefore wasn't visible to the eye.
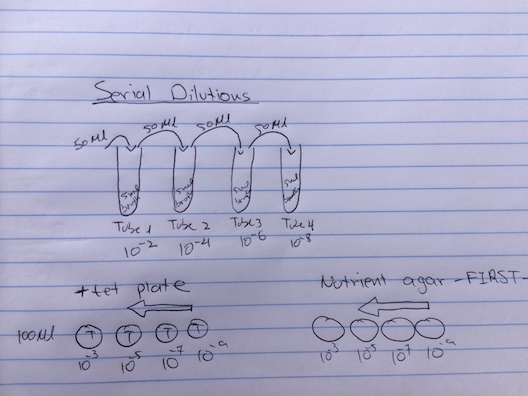
Microbiology and identifying Bacteria with DNA Continuing where we left off in lab 2 with spreading samples of our serial dilutions from the hay infusion cultures on petri dishes with nutrient agar and tetracycline, bacteria and possibly some fungi might have grown on the surfaces. Multiple bacteria that form together to make one bacterium form what is called a colony, which is very visible to the eye. Colony morphology is a characteristic for each species. We counted the total number of colonies on each plate where depending on the dilution. there would be a range of bacteria colonies on each plate.
Table 1 100-fold Serial Dilutions Results
Dilution (Plate Table) Agar Colonies Counted Conversion Factor Colonies/ml 〖10〗^(-3) Nutrient 200 X〖10〗^3 200.000 ml 〖10〗^(-5) Nutrient 6 X〖10〗^5 600,000 ml 〖10〗^(-7) Nutrient 0 X〖10〗^7 0 ml 〖10〗^(-9) Nutrient 0 X〖10〗^9 0 ml 〖10〗^(-3) Nutrient +Tet 107 X〖10〗^3 107,000 ml 〖10〗^(-5) Nutrient +Tet 2 X〖10〗^5 200,000 ml 〖10〗^(-7) Nutrient +Tet 0 X〖10〗^7 0 ml
- In the case of hay culture infusion bacteria, they seem to have been antibiotic resistant bacteria as they still formed colonies in the presence of tetracycline. Comparing the colonies size of that in nutrient agar with that in nutrient + tet agar, the colony size are close with few exceptions due to the dilution of those samples. In the most dilute samples 10^-9 (nut) and 10^-7 (nut + tet) there was no bacteria present even with the addition of tetracycline as it should. Tetracycline is supposed to work by preventing the growth and spread of bacteria; however in our case there was growth on those agar plates with tet indicating that our bacteria species in the prairie transect is resistant to it.
Bacteria Cell Morphology Observations - We observed both a native wet mount preparation and a gram stain of two well defined colonies taken from the nutrient agar plate and one taken from the tetracycline plate. We used the 10^-3 and 10^-5 nutrient agar and 10^-3 tetracycline plate to observe.
Wet mount preparation: 1)scoop a tiny amount of growth from the surface of the agar, and mix it into a drop of water on a slide. 2)place a cover slip over the drop before observed with microscope
Gram stain preparation: 1)Label the slides 2) heat fix the air dried side by passing it through a flame with the bacterial smear side up three times 3) cover the smear with crystal violet for one minute over a staining tray 4) rinse the stain off with bottled water 5) cover smear with Gram's iodine mordant for 1 minute. Rinse gently. 6) decolorize by flooding smear with 95% alcohol for 10-20 seconds. Rinse gently.(when solvent flows colorlessly from the slide, decolorization has occurred) 7) cover smear with safranin stain for 20-30 second. Rinse gently. 8) blot excess water carefully with paper towel and air dry. 9) observe the gram stain under 40x using the microscope.
Observations made using wet mount and gram stain File:Observations
PCR Preparation for DNA Sequence Identification A)Select a bacteria from each of the three plates that has the best characterization. B)DNA from the bacteria will be isolated using two primer sequences (27F and 519R) to selectively amplify the 16S rRNA gene.
1) transfer a single colony of bacteria to 100 microliters of water n a sterile tube. Incubate at 100 degrees Celsius for 10 minutes and then centifuge. 2)The following week the PCR products will be ran on an agrogose gel and if it looks good, the PCR products will then be purified using a Qiagen kit and sent to Genewiz Inc for sequencing--- DNA sequences would then be analyzed.
January 23, 2014 (Lab 2)
As a preparation in identifying unknown organisms in our prairie transect, wet mounts were made with known organisms provided in lab where they were observed under the microscope. Using the Dichotomous Key, we were able to identify the known organisms according to observations made based on their size, shape, movement, and color.
After having a sample of our prairie transect in a jarred container with deer park water and powdered milk, the following: - semi- thick film on surface of water - some underlying mold-like substance underneath globs of soil that float on top of water's surface - greenery remained intact in the plant stem - 3-apparent niches
1) a soil based bottom
2)liquid water middle (water pretty transparent)
3) a thin film layered surface
- water in the container again is transparent with a brownish tint - mildew surrounding the perimeter of the jar at the surface layer
Using the Dichotomous Key just as we did in preparation, we were able to identify unknown organisms in our hay infusion of prier transect. I specifically focused on the possible unknown organisms found at the bottom of the jar of our hay infusion. That area being a soil based niche in regards to the other niches mentioned earlier (Liquid water middle and thin film layered surface).
Organisms found in soil based bottom niche
1) Paramecium Vorticella (40x) - 6oc(ocular spaces)=15 um -small cell with elongated body -swims rapidly in a corkscrew fashion -body covered entirely with cilia
2)Colpidium (40x) - 10oc= 25 um -white/colorless -small body, oval shaped with small mouth -fast swimmer
3)Amoeba (40x) - 1oc= 2.5 um -small -creeps using false feet -single disk-shaped nucleus
4)Gonium (40x) -14oc=35 um -colony flat, disc-shaped, usually containing 16 cells -organ of locomotion is long whip-like flagella -white/colorless
- Amoeba, one of the four organisms found in the soil based niche of our hay infusion demonstrates the qualities of being "alive" as: it acquires energy from its by means of using its pseudopods, it's single-celled, processes information (genes), is cable of replicatingasexually (binary fission), and are a product of evolution
- Some selective pressures that may have affected the composition of our samples may be the lack of or increase in sunlight exposure, which in turn has an affect on how much/little energy is acquired by the organisms. As well as the lack/ increase of oxygen(air), where the organisms that are aerobic would have definitely been affected.
Preparing Serial Dilutions Procedure: 1) Four tubes were obtained each containing 10 mls of sterile broth and labeled 2,4,6,8 respectively. 2)Obtained four nutrient plates with agar labeled (10^-3, 10^-5, 10^-17, 10^-9)and three nutrient plates with agar and the addition of tetracycline labeled (10^-3 T, 10^-5 T, 10^-17T), *the T representing tetracycline 3)Swirled the hay infusion order to mix up all the organisms and then took 100 microliters using a micropipette from the mix and aseptically added it to the to mls of sterile vroth in test tube 2 (10^-2 dilution). The inoculated tube was then swirled 4)100 microliters of broth fro test tube 2 was then taken and inserted in test tube 4 (10^-4 dilution). 5)The process was then repeated to make 10^-6 and 10^-8 dilutions. 6) 100 microliters from test tube 2 were taken ans easptically placed on the surface of the nutrient agar plate (10^-3). the sample was then carefully spread on the surface. 7) The procedure (step 6) was repeated exactly with the nutrient agar plate containing tetracycline (10^-3 T). 8) The procedures taken place in steps 6 and 7 were then repeated using test tube 4 for the 10^5 plates, and test tube 6 for the 10^-7 plates. 9) Finally the agar plates were incubated at room temperature over the next week.
After snowfall that occurred on 1/21/14, our prairie transect was naturally covered with snow
January 16, 2014 (Lab 1)
- Prior to defining our niche on campus, samples of green algae were looked at in determining the Volvecine Line - Compared the following green algae using microscopes: 1)Chlamydomonas 2)Gonium 3)Volvox - When comparing the evolutionary specialization of members of the volvocine line we looked at: number of cells, colony size, any functional specialization of cells, and reproductive specialization - classifying these cells according to their special functions (ie reproduction) provided insight on the evolution of green algae in the volvocine line by observing an differences or similarities between unicelluar algae such as Chlanydomonas and muticellular green algae such as Volvox.
Group 5: Prairie
Goal/objective: Analysis of 20 by 20 transect of prairie land
- Determining what what set of specific requirements collectively make the niche prairie land. - What interactions, if any, are there between the organisms within the community inhabiting prairie land? - what organisms are there to even begin with? Is there much biodiversity? - How does weather/ change in season affect the prairie transects topography and the organisms that reside there?
Materials Used: - 50 ml conical tube - 10 grams of sample of prairie transect - open jar for hay infusion culture - 0.1gram dried milk - 500 ml deer park water
Procedure: Exactly as followed from Lab 1:Biological Life at AU Step 1: describe the general characteristics of the prairie Step 2: take a sample of soil and vegetation from transect and put it in the 50 ml conical tube. Step 3: make a hay infusion culture
- weigh 10 grams of soil sample, place in jar with 500 mls of deerpark water
- add 0,1 gram dried milk into the jar containing at this point water, and soil sample
- place jar cover on and mix up all the components for 10 seconds
- remove the jar lid and let it sit
- The fact that the jar lid was not left on, poses the question of how oxygen will affect the hay infusion culture over a period of time. Will our initial prairie niche stay in tact? is there the possibility hat other niches may form? will there be any new organisms?
Characteristics of Prairie land
Abitotic Elements:concrete benches, walkway stones, metal lining surrounding soil, rocks pebbles
Biotic Elements:soil/fertilizer, bushes/shrubs, leaves, flowers, grass, birds (chicks)
- Area mostly consists of biotic elements with the implementation of man made factors such as the concrete benches and walkway stones (abiotic elements) - Due to the current winter season, the plot of land is essentially barren. Dead flowers on the bushes/shrubs, little to no grass directly on the soil. - On this particular day, there was a good amount of sunlight (source of energy for any potential growth of the biotic factors)