User:Sydney Murphy/Notebook/Biology 210 at AU
March 15
Zebrafish Chart
| Date/ Type | # living | # dead | stage of development | Color | Eye Color | Size | |
| 2/18 ex | 20 | 0 | 17-20 Somites | Clear | N/A | N/A | 1 mm |
| 2/18 c | 20 | 0 | 17-20 Somites | Clear | N/A | N/A | 1 mm |
| 2/20 ex | 20 | 0 | 31 h | clear w/ black speckling | black | N/A | 1 mm |
| 2/20 c | 8 | 7 | 31 h | clear w/ black speckling | black | N/A | 1 mm |
| 2/20 c2 | 20 | 0 | 31 h | clear w/ black speckling | black | N/A | 1 mm |
| 2/23 ex | 20 | 0 | 60 h | slightly yellow w/ black speckling | black | fast/agile/freeze in light | 4 mm |
| 2/23 c | 8 | 0 | 60 h | clear w/ black speckling | black | fast/agile/freeze in light | 4 mm |
| 2/23 c2 | 20 | 0 | 60 h (4 mishappen) | clear w/ black speckling | black | fast/agile/freeze in light | 4 mm |
| 2/25 ex | 19 | 1 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 2/25 c | 8 | 0 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 2/25 c2 | 16 | 3 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 2/27ex | 18 | 0 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 2/27 c | 8 | 0 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 2/27 c2 | 6 | 10 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 3/2 ex | 11 | 7 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 3/2 c | 7 | 1 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
| 3/2 c2 | 16 missing | 16 assumed | N/A | N/A | N/A | N/A | N/A |
| 3/4 ex | 0 | 11 | N/A | N/A | N/A | N/A | N/A |
| 3/4 c | 2 | 5 | juvenile | slightly yellow w/ black speckling | black | fast/agile | 5 mm |
Sydney Murphy
To see photos of the fish throughout the experiment please visit: [[1]]
February 17, 2015
Invertebrate Identification: February 11
This lab allowed us to identify the invertebrates that were found in the leaf litter in our transect. It was also the last lab of our transect. I was not expecting to find much in the Berlese Funnel, due to the pine needles and the temperature of the ground they were found in.
We first separated the collection tube from the leaf funnel and collected a sample from the bottom and a sample from the top to observe. We observed each organism through the dissecting microscopes and classified them by structures. We observed the different types of worms and the way that they moved: the flatworms moved in a fluid swimming motion through water and the roundworms move in a corkscrew motion through the agar.
| Organism | length | number | Description |
| Ixodida Ixodoidea (tick) | 1 | 1 | File:Sydney Murphy tick.jpg |
| Isopoda (pillbug) | 3 | 1 | 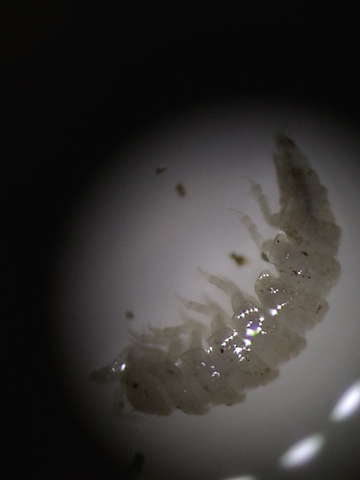
|
| Unidentified insect | 2 | 1 | 
|
| Springtail X | 1 | 6 | File:Sydney Murphy bugg.jpg |
| Proturan X | 2 | 2 | 
|
This chart shows five of the organisms found in the Berlese Funnel, one of which could not be identified using a dichotomous key. Though they were all quite similar in size, the organisms still contained a lot of diversity. The pillbug was the largest, around 3 millimeters in length with most of the others around the same size, 1 millimeter in length. The Springtail X was the most common in the litter, with 6 specimens in just the amount of liquid we pulled out.
Other organisms that likely visit our transect include birds, humans, squirrels, and other wild animals common to the American University campus.
Sydney Murphy
February 10, 2015
Plant and Fungi Identification: February 3
This lab was set up for us to begin classifying the types of life in our transects,from plants to protists. It is entirely likely that we find a great variety of plants in our transect, as it is a raised farmland style plot.
We first set up our gel electrophoresis with the Hay Infusion liquid and allowed it to run while we collected plants from our transect. We first collected our leaf litter from next to the bushes, where not all of the leaves were dry. Then we collected leaves from the spinach plot, kale plot, brussel-sprout plot, bush near the gates, and next to the spinach box. We classified and described each of the types of plants these leaves came from. Lastly, we observed fungus and set up a Berlese Funnel to catch the invertebrates collected in our leaf litter.
| Location | Description | Vascularization | Specialized Sturctures | Mechanisms of Reproduction |
| Kale Plot | green, small, leafy | yes | dicot | flowering |
| Brussel-sprouts plot | green, round leaves, thick/hard stems | yes | dicot, no visible stomata | flowering |
| Spinach Plot | green, leafy, flimsy | yes | dicot, stomata | flowering |
| Bush near gates | purplish, reddish, green, rough | yes | dicot, no visible stomata | flowering |
| next to spinach box | green, small, 3-leaf clusters, round leaves | yes | monocot, stomata | flowering |
This chart is the classification of each of the leaves we collected from our transect, we collected a Kale leaf, Spinach leaf, Brussel-sprout leaf, Clover leaf, and Bush leaf. They all presented with vascularization and most with dicot tendencies.
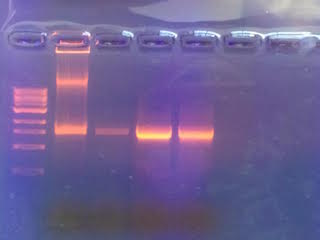
This image represents the gel of the bacteria found in our transect. The left-most line is the ladder, then the four samples to the right of it.
This image is a close up of the sporangia of a fungus, which help the fungus in reproducing sexually.
Sydney Murphy
February 2, 2015
Microbiology and Bacteria Identification: January 28
This lab allowed us to examine our serial dilution plates and the bacteria that grew on them over the past week. It is entirely likely that the weaker the concentration the less bacteria will grow; the plates with tet will likely have even less than their non-tet equivalent. It is extremely unlikely that any archea will be found in our samples as the environment was not extreme enough to perpetuate their existence.
We began the lab by observing our Hay infusion Culture and serial diffusion plates. The Hay infusion still reeked of mold, though no more appeared to be growing; most of the water had evaporated from the Culture. The plates that were treated with the tet had less growth, though the 10^-9 dilution had very few in both scenarios. We then made slides of colony samples from four of the eight plates, 10^-5 non-tet, 10^-5 get, 10^-7 non-tet,10^-7 get, and observed each for cell descriptions. Another sample was taken from the same plates and gram stained, to test for peptidoglycan. Lastly, we used another sample from the same colony to begin PCR to isolate the 16S rRNA gene for use next week. The agar plates were placed upside-down for another week of growth.
| Dilution | Agar Type | Colonies Counted |
| 10^-3 | no tet | lawn |
| 10^-5 | no tet | 480 |
| 10^-7 | no tet | 17 |
| 10^-9 | no tet | 1 |
| 10^-3 | tet | 370 |
| 10^-5 | tet | 14 |
| 10^-7 | tet | 1 |
| 10^-9 | tet | 0 |
This chart shows that as the concentration decreases the number of colonies decreased as well. Tet clearly caused a decrease in the amount of colonies present.
| Plate Type | Colony Description | Cell Description | Gram +/- |
| 10^-5 no tet | 10^-7 no tet | 10^-5 tet | 10^-7 tet |
| yellow, undulate, raised | filamentous, flat, whitish | curled, convex, yellow | dark green, round, umbonate |
| budding | streptobacilli | coccus, singular | diplococci |
| neg | pos | neg | pos |
Though our scope was not the best and had trouble focusing on 40x, we were able to mostly figure out which bacterium were present.
The most common type of bacteria present was coccus, or a form of it. The gram staining came out with an even 2 positive and 2 negative. Next week, it will be easier to distinguish the different strains with their DNA samples.
Great job! Make sure you post pictures to help document your findings as well! ML
January 27, 2015
Protist Identification and Using a Dichotomous Key: January 21, 2015
This lab introduced us to using Dichotomous keys and allowed us to find and classify the protists in our Hay Culture Infusions. It is likely that the Culture will contain a large number of protists throughout the span of the jar, due to the different types of soils collected.
Great Job Sydney! I like your lab notebook so far. I would like more photos that show the diversity you are seeing in your transect. Your descriptions are very complete and your rationale has a logical progression. Keep up the good work! ML
We began by making a wet mount of a mix of known organisms and classifying two using a dichotomous key. Our slide contained Paramecium Multimicronucleature at 250 micrometers and Colpidium at 50 micrometers. We then moved onto out Hay Infusion Culture, which had separated, was beginning to grow mold on the surface, and was giving off a pungent odor. We took a sample from the center bottom, the center top, and the center middle next to a leaf and made wet mounts of the three, identifying two organisms from each area. When this was finished we set up next weeks experiment by making serial dilutions, beginning with 100 microliters of Hay Culture: 10 milliliters Broth then diluting 100 microliters of this solution into 10 milliliters of Broth. This continued two more times, leaving us with 4 tubes of decreasing concentration. We then spread 100 microliters of each solution on two agar plates, one with tetracycline and one without. The plates were then set to grow agar side up for a week.
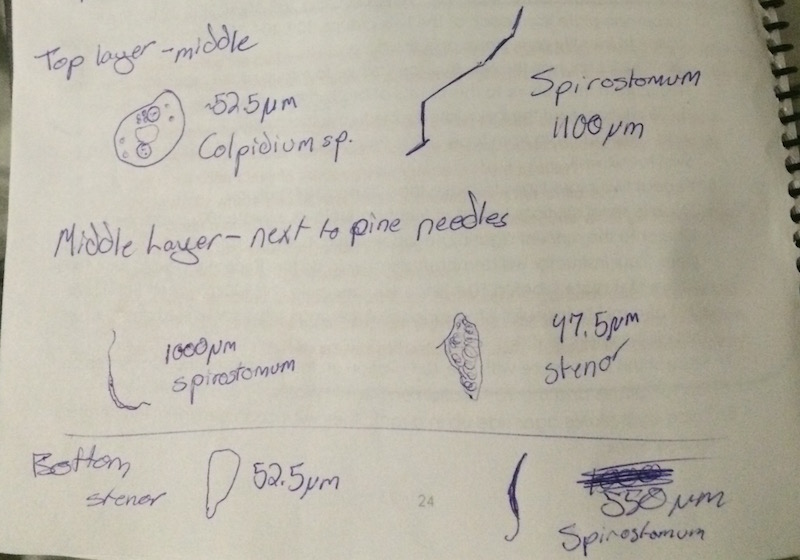
Within the Hay Culture Infusion there was little variation between the layers. Each layer consisted of Spirostomum varying in length from 550 Micrometers to 1100 micrometers. The two lower levels both consisted of Stenor, ranging from 47.5 micrometers to 52.5 micrometers. The top also consisted of a Colpidium at 52.5 micrometers. The lack of diversity within the Culture is likely due to the cold weather and the fact that the soil they were collected from was frozen when the samples were taken. There will likely be more diversity when the weather heats up and the ground thaws out.
Sydney Murphy
January 27, 2015
Volvocine Line and Transect Introduction: January 14, 2015
The purpose of this lab was to reintroduce us to microscopes and making wet mounts, as well as getting acquainted with our transects. By taking the first day to adjust to being back and introducing us to our transects it allows us to successfully transition into the class and get to know our partners better.
We began the lab by making wet mount slides of the three examples of the Volvocine line: Chlamydomonas, Gonium, and Volvox. We then observed the samples under a microscope, searching specifically for colony size, cell specialization and motility mechanisms. After, we walked around campus and were introduced to our transects, which we mapped, drew samples of and listed biotic and abiotic factors. When we returned we made a Hay Infusion Culture by combining 12 grams of our soil, 500 mLs of water, .1 grams of dried milk in a jar, they were mixed together and left to sit open for a week on the back table.
| Characteristic | Chlamydomonas | Gonium | Volvox |
|---|---|---|---|
| Number of cells | 1 | 4 | 1 |
| Colony Size | 10 | 17.5 | 200 |
| Specialization? | No | Yes | Yes |
| Motility Mechanisms | Flagella | None | Flagella |
| Iso or Oogamous? | Isogamous | Oogamous | Oogamous |
The side by side comparison of the Volvocine line shows the evolution over time, from a single-celled organism, to a colony, to an advance organism.
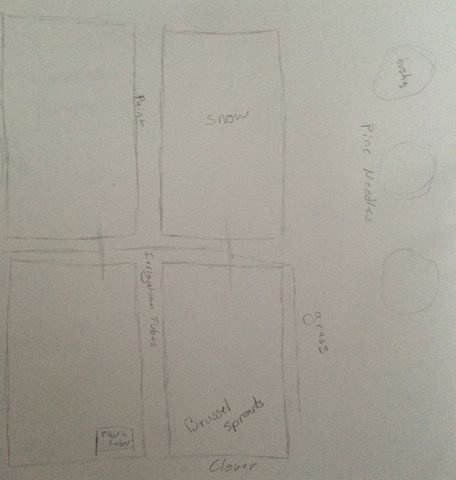
| Abiotic | Biotic |
| Snow | Brussel Sprouts |
| Rocks | Clover |
| Irrigation Tubes | Pine Needles |
| Plastic label | Bushes |
| Paint | Grass |
These are some of the prominent Abiotic and Biotic factors from our Transect. Transect four is found within the community garden near the soccer fields towards the back edge of campus. It is a fairly flat area with four raised plant beds on the one side and three bushes on the other side, closer to the fence and gate.
Overall, this experiment worked well; we were able to observe our transect and collect a variety of soil for our Hay Infusion Culture. Though, the ground was frozen and the protists we collected may end up reflecting this in their diversity, or lack thereof. Hopefully it will be warmer next time we collect from our transects, allowing us more variety in protists collected.
Great description! I liked the aerial drawing and from your description had a great idea of what the environment looked like. In the future, including other photos would also be helpful. Great Job! ML
Sydney Murphy
January 22,2015
This is my test message for lab! Hope it works well!