User:Student 64/Notebook/Biology 210 at AU
March 3 2016 Zebrafish
Purpose to examine and compare a controlled versus treated group of zebrafish when exposed to 7.5ppm fluoride through the water over a span of 2 weeks.
Methods and Materials - prepare a controlled environment by filling 24 wells with 2mL distilled water - prepare the treated water by creating a solution of 7.5ppm NaF (about 500mL) - collect 48 fertilized eggs in a Petri dish - drop one egg in each well - clean and feed the fish daily by taking out the egg shell, food waste, and also changing the water. - feed the fish 15μL of food daily once hatched
Observation and Data Controlled 4 controlled fish have died and one did not hatch. They had similar hatching rates compared to the treated, however, more controlled hatched in the span of 72 hours than treated. tend to have a darker complexion. spend more time in the middle of the well then the edges. Smoother movements
Treated 3 dead one did not hatch 4 empty wells (possibly unfertilized eggs) most hatched similarly to control but some were delayed. tend to be smaller and lighter in complexion had two with orange eyes one had a bend in the spine (looked like an S shape) jolted movements
Conclusion So far fluoride has not affected the death rate of the zebrafish, but, it has affected the size and color of the zebrafish. This might mean that they are able to adapt to their enviroment but it takes more energy and develop less in other areas to counter the environment.
--Michaela Nieuwenhuizen 16:28, 3 March 2016 (EST)
March 3 2016 PCR Sequencing
Purpose to adequately sample bacteria colonies from serial dilution agar plates and perform PCR on the 16S sequence from the sample. Thereafter, the aim is to use the results to adequately identify the bacterial species found to be living within the borders of Transect One.
Materials and Methods - Using samples from the serial dilution bacteria plates labeled Agar + Tetracycline 10^-6 (13A) and Agar + Tetracycline 10^-2 (13D) that were originally collected from the Hay Infusion Cultures of Transect One, DNA was isolated into a 100uL of water in a sterile tube. For ten minutes, the tube was incubated in a heat block at 100 degrees Celsius, immediately being centrifuged for five minutes at 13,400 rpm. A total of 20uL of primer/water mixture was added to a PCR tube, after which the PCR-bead dissolved. Lastly, 5uL of supernatant from the centrifuge was added to the 16S rRNA reaction and the tube was placed in a PCR machine. Data and Observations After one week, the PCR products were run on an agarose gel and the DNA was purified for sequencing File:PCRtransect1.JPG
CAATCCTGGCGGCGAGTGGCGAACGGGT GAGTAATACATCGGAACGTGCCCAATCGTGGGGGATAACGCAGCGAAAGCTGTGCTAATACCGCATACGATCTACGGATG AAAGCAGGGGATCGCAAGACCTTGCGCGAATGGAGCGGCCGATGGCAGATTAGGTAGTTGGTGAGGTAAAGGCTCACCAA GCCTTCGATCTGTAGCTGGTCTGAGAGGACGACCAGCCACACTGGGACTGAGACACGGCCCAGACTCCTACGGGAGGCAG CAGTGGGGAATTTTGGACAATGGGCGAAAGCCTGATCCAGCCATGCCGCGTGCAGGATGAAGGCCTTCGGGTTGTAAACT GCTTTTGTACGGAACGAAACGGCCTTTTCTAATAAAGAGGGCTAATGACGGTACCGTAAGAATAAGCACCGGCTAACTAC GTGCCAGCAGCCGCGGTAATACGTAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGTGCGCAGGCGGTTATGT AAGACAGTTGTGAAATCCCCGGGCTCAACCTGGGAACTGCATCTGTGACTGCATAGCTAGAGTACGGTAGAGGGGGATGG AATTCCGCGTGTAGCANTGNAATGCGTAGATATGCGGAGGAACACCGATGGCGAANGCAATCCCCTGGACCTGTACTGAC GCTCATGCACGAAAGCGTGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCACGCCCTAAACGATGTCAACTGGTTGTT GGGTCTTCACTGACTCA
Results Variovorax paradoxus strain 57-Y925 16S ribosomal RNA gene, partial sequence
February 22 2016 Exercise VI: Embryology and Development
Purpose to perform and experiment with a controlled and treated sample of zebrafish and determine their developmental stages and provide an observation the effect of the chemical on development and viability. Fluoride can be harmful because it can penetrate the fetal-maternal membrane, and therefore, affect a fetus. We are looking at whether or not it will affect the size nervous response and skeletal functions of zebrafish. We had to use a very small amount because studies have shown with doses of 15ppm that their treated group died and did not have nay leftover to study.
Methods and Materials
-prepare container with 2mL of the controlled solution (distilled water) in each section. - prepare treated solution of 7.5ppm NaFl and fill the container with 2mL in each section -collect 48 fertilized eggs (clear) with a pipette
--Michaela Nieuwenhuizen 20:50, 23 February 2016 (EST)
February 17 2016
Exercise V: Invertebrates and Vertabrates
Purpose To be able to use a dichotomous key to identify invertebrates found in your transect and give examples of which germ tissues give rise to. Understand the different mechanisms for mobility, different symmetries, and digestive systems.
Methods and Material -identify five invertebrates from your Berlese funnels collections, and record your results. - concluded the range of organisms you had in your transect - identify five invertebrates that might inhabit the transect, and identify their classification from phylum down
Observation and Data
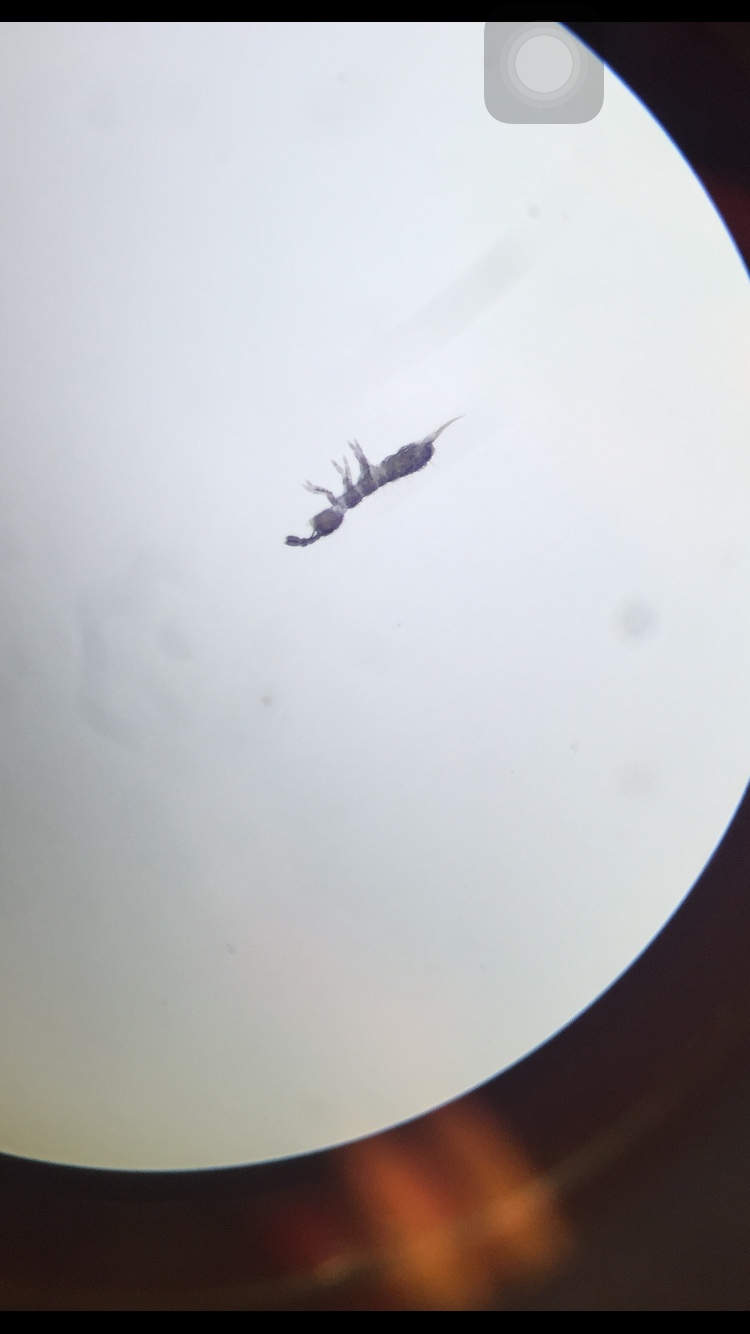
- 1. Springtail: we found 2 organisms in the sample about 2mm in length. It had 6 legs, wings by its side and was a light brown/beige color, and bilateral symmetry
- 2. Biting lice: found only one in the sample that was about .5mm long. It was orange, with bilateral symmetry, and 6 legs
- 3. Tick: found four in the sample that was about .2mm long. They were small with a hard shell, black in color, 6 legs, and no wings
- 4. Termite: white in color with legs and only .5mm in length. Only one was found in the sample
- 5. Ant: found 2 in the sample about 1.5mm in length. It was brown in color with 6 legs and a bead-like antenna. it had eyes
Conclusion
Febuary 10 2015
Exercise IV: Plantae and Fungi
Purpose be able to characterize plants that have evolved through evolution and describe the definitive characteristics of fungi and understand the difference from plant. Know the difference between angiosperms and bryphytes and draw the schematics. To understand the structure and function of a plant reproduction system.
Methods and Material - find 5 different plants from your transect and organize the data by classification. Identify the major group and genus for each. - characterize the vascularization of each plant and record the height. The observe a cross section of your plant. - now describe the shape, size, and cluster arrangement of the leaves from your plants. if there are no leaves examine the evidence of if there is (attachment site, or leaf litter) - identify the seeds you brought back as either monocot or dicot. - describe what fungi sporangia are and their importance. decide if they are fungi and which of the three groups they belong to.
Observation and Data



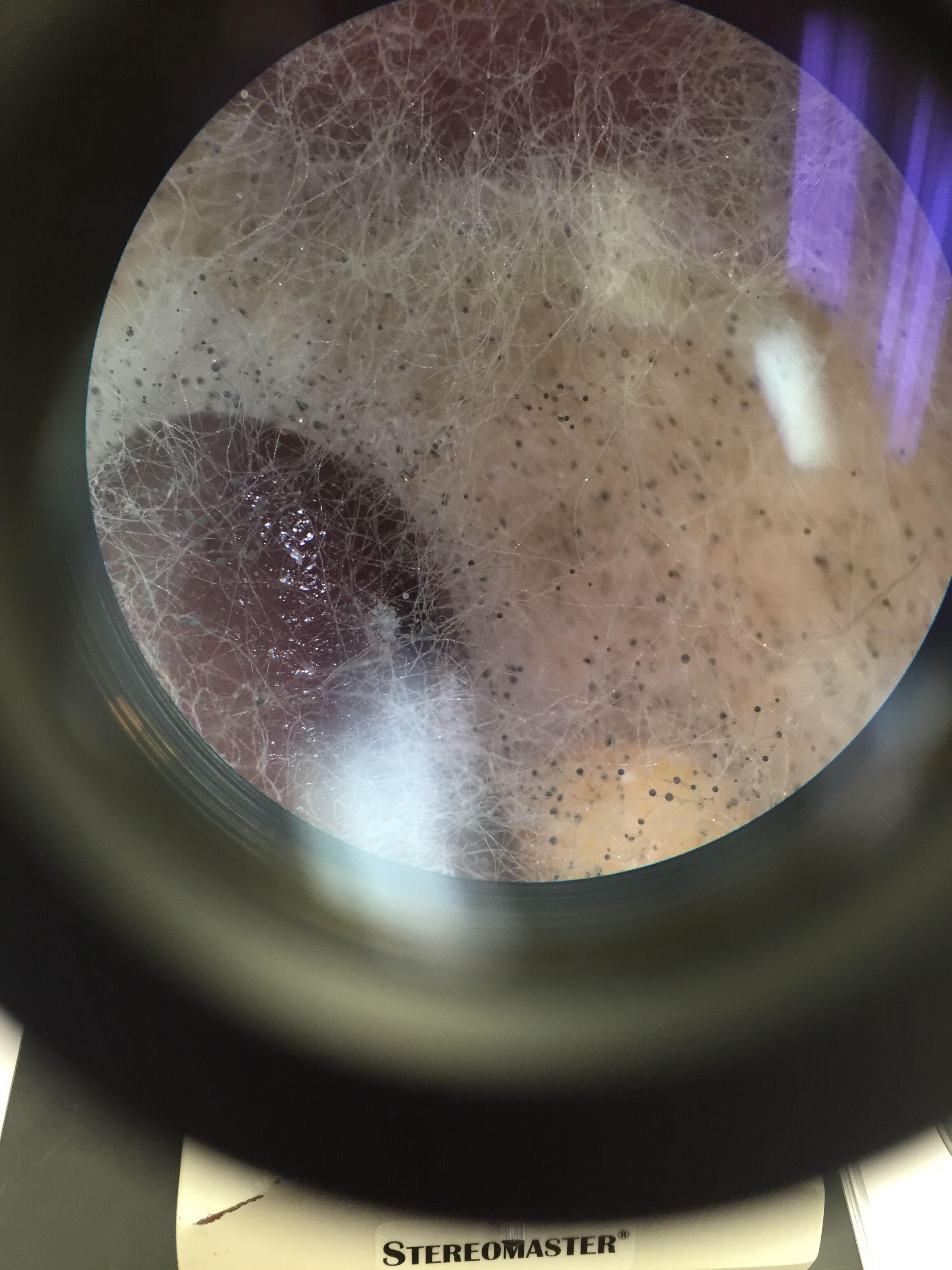
Conclusion the black bread mold is a fungi sporangia because it can reproduce asexually because it has spores that you can see under the microscope. It is classified under the Zygomycota phylum.
--Michaela Nieuwenhuizen 10:57, 10 February 2016 (EST)
Febuary 3 2015
Exercise lll: Microbiology and Identifying Bacteria
Purpose: To characterize and identify bacteria based on motility, gram stains, colony morphology and sequencing of the 16s ribosomal subunit gene.
Method and Materials:
Observing Hay infusion culture after 2 weeks noting any changes in smell or appearance procedure I: Observation of growth on agar plates that were made last week from the hay infusion culture. Identify the number of colonies and what it appearance looks like procedure II: look at the agar plates that contain tetracycline and compare the difference of bacteria growth and how antibodies might help accelerate bacteria growth or inhibit it. procedure III: observe both a wet mount and gram stain of two well-defined colonies from both the nutrient agar plate and two from the tetracycline plate. wet mount, place a drop of water on a clean slide and use a sterile stick to transfer the bacteria from the agar plate onto the slide. gram stain, mix the bacteria from the agar plate onto a clean slide with a drop of water. get rid of the water by passing the slide over a flame with the bacteria side up. do not place it in the flame or too close to the flame. Cover the bacteria smear with crystal violet for 1 minute the rinse the stain off using a wash bottle of water. decolorize by using 95% alcohol for 10-20 seconds. Cover the smear with safranin stain for 20-30 seconds. Rinse the stain off using a wash bottle of water. Blot the excess water off with a kimwipe and allow to air dry. procedure IV: prepare a PCRf or 16S amplification by taking a tiny bit of bacteria from the agar plate into the primer. Mix to dissolve the PCR bead. place in the PCR machine.
Observations and Data: Hay Infusion: the smell of the hay infusion has decreased from last week. The smell is no longer potent. It smells just like dirt. This could be because the dirt had soaked up some water. The water level went down from the previous week. All the sticks and leaves looked moist and soggy. They were no longer stiff and had a green plant growing on them. This could be the algae we found last week that has now grown more.
File:Agarplates.jpg File:10-5agarplate.jpg File:10-3agarplate.jpg
Conclusion:
- Michaela Nieuwenhuizen 11:38, 3 February 2016 (EST):
No serial dilution table or bacteria characteristics table. -Pragati
January 27 2016 - Exercise II: Identifying Algae and Protists
 Figure 1. Hay Infusion side view
Figure 1. Hay Infusion side view
 Figure 2. Hay Infusion Top View
Figure 2. Hay Infusion Top View
The Hay Infusion of our transect smelled like mold. It had a wet mild smell that was not pleasant. As you can see in the pictures above the water was a light brown, dirt settled to the bottom and there was a thin film on top of the water, which could mean life growing. There was also sticks leaves and grass in the Hay Infusion.
Niche #1 - Top of Hay infusion A. Stramenopila algae, it was a golden brown color, it was about 60um, and nonmotile. File:Stramenopila.jpg Figure 3. Organism 1
B. Pelomyxa protist, clear/colorless, floated (non-motile), not spherical in shape (shape changes), and was about 30um. File:Pelomyxa.jpg Figure 4. Organism 2
C. Chlamydomonas protist, circular in shape, unicellular, non-motile and about 47um
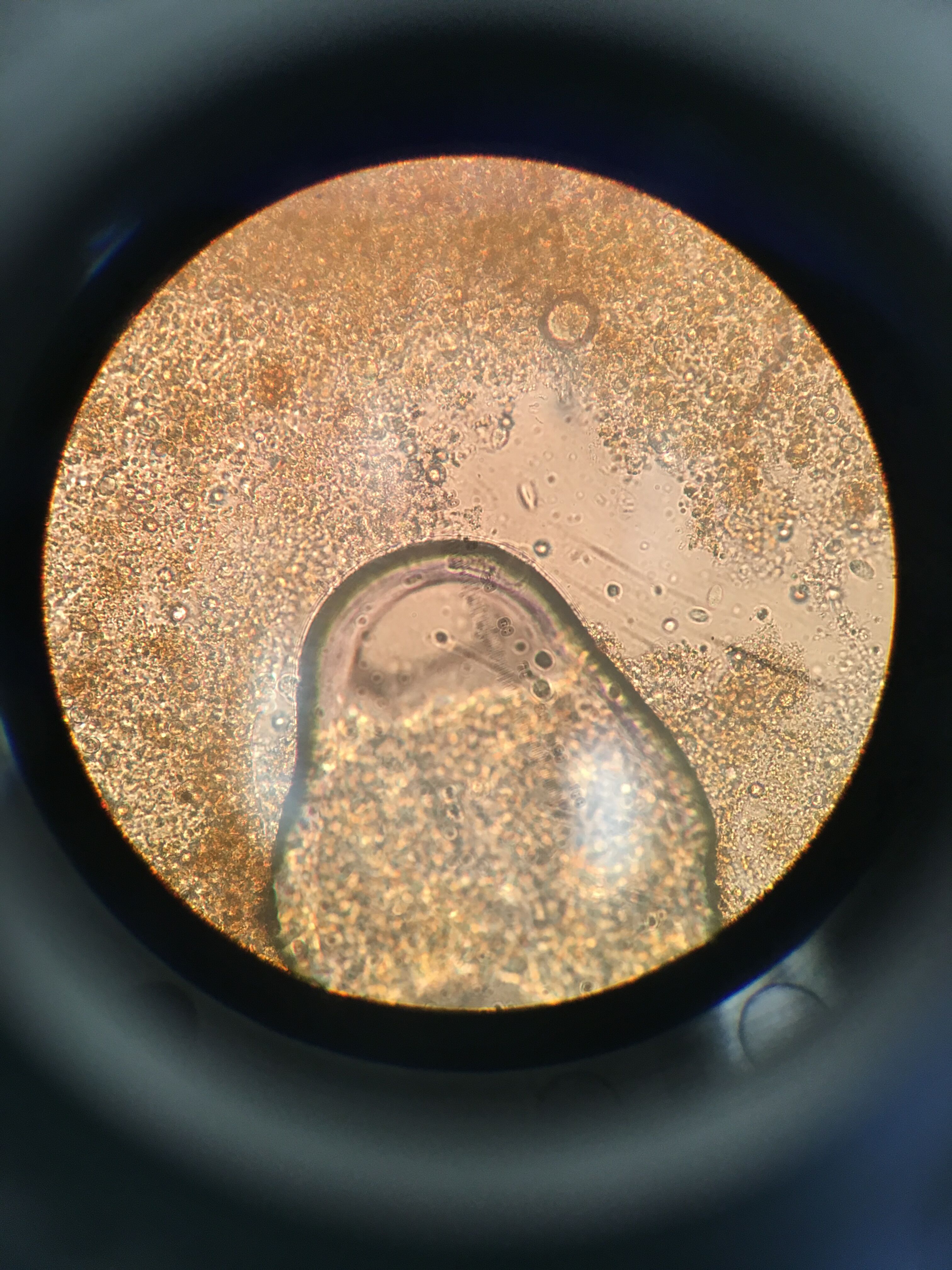 Figure 5. Organism 3
Figure 5. Organism 3
Niche #2 - Middle (near top of grass) A. Pandorinaprotist, uniform, circular in shape, clustered, pinkish membrane with green insides, between 25-40μM, non-motile, and single nucleus File:Pandorina.jpg Figure 6. Organism 4
B. Euglena algae, green in color, single motile cell, one flagella, elongated in shape, about 10μM File:Euglena.jpg Figure 7. Organism 5
C. Volvox algae, spherical in shape, colony, green in color, non-motile (float)
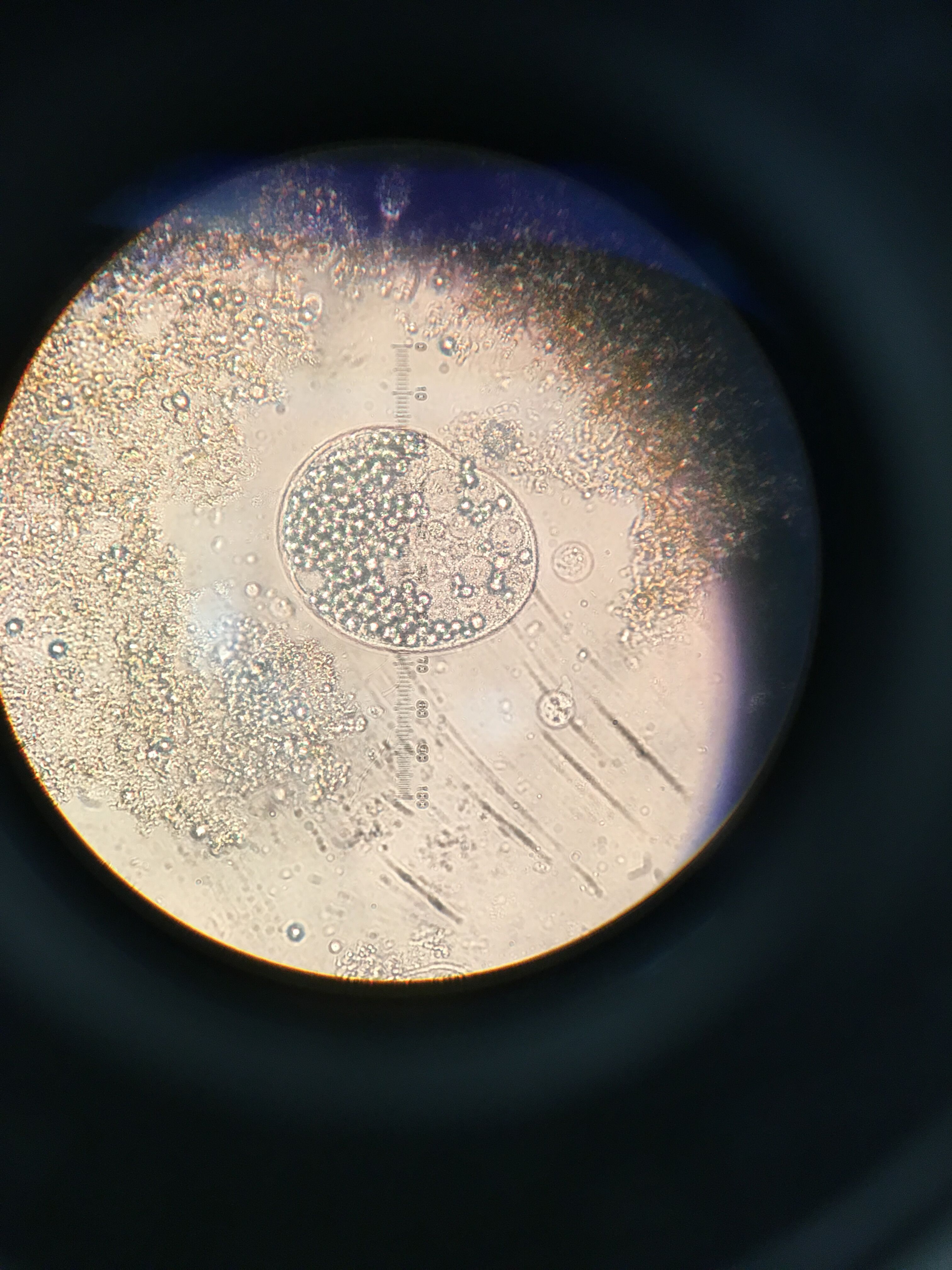 Figure 8. Organism 6
Figure 8. Organism 6
File:Hay infusion serial dilution.jpg Figure 9. Hay Infusion Serial Dilution Diagram
--Michaela Nieuwenhuizen 18:42, 27 January 2016 (EST)
January 27 2016 - Exercise I: Examining Biological Life at AU
 Figure 1. Transect 1 looking from east side
Figure 1. Transect 1 looking from east side
My transect is located beside the Hurst building. It is a 20 by 20ft piece of land that contains, a large tree, dirt, a lamp post, sprinklers and more. The soil has multiple things planted and some plants have been cut for winter, but their roots are still visible. There is evidence of weeds and grass growing in the soil but not in large quantities. Garbage was found in the transect which could be a result of being next to a sidewalk. Abiotic factors of this transect is the light, garbage, location and sunlight, and sprinklers Biotic factors are the plants growing, the soil, and the animals living there.
--Michaela Nieuwenhuizen 16:27, 27 January 2016 (EST)