User:Student 55/Notebook/Biology 210 at AU
Thursday, February 25, 2016
Purpose The purpose of this experiment was to use PCR (polymerase chain reaction) and primer to identify a 16S rRNA gene. We took samples from two nutrient rich and deficient agar plates one of which contained the antibiotic tetracycline. We found the gene sequence that would be a resistant (this is the species that still grew on the agar plate containing tetracycline). The bacteria we identified was a commonly occurring form of bacteria.
Materials & Methods 2 PCR tubes Add 25 micro-liters of primer concentration to PCR tube. Mix the two until dissolved forming the PCR bead. 4. Take a toothpick and swab the solution in the PCR tube (make sure to get a sample of bacteria from the colony on the toothpick) 5. Put tube into the PCR machine. 6. Repeat steps 1-5 using the second PCR tube and a new toothpick Next Lab… Pipette 5 micro-liters of the PCR from last class into the agarose gel. Place agarose gel underneath lamp. Observe and record results. Next Lab… Identifying 16s Sequence 1. 16S Nucleotide Sequence. 2. use this website to “balst” your sequence: https://blast.ncbi.nlm.nih.gov/Blast.cgi?PAGE_TYPE=BlastSearch 3. Copy and paste the sequence into the “Blast.”(use sequence 30 characters in from the left and right of each sequence) 4. observe and record results
Results:
Nucleotide Sequence CTTGCTGCTTCGCTGACGAGTGGCGGAC GGGTGAGTAATGTCTGGGAAACTGCCTGATGGAGGGGGATAACTACTGGAAACGGTAGCTAATACCGCATAACGTCGCAA GACCAAAGAGGGGGACCTTCGGGCCTCTTGCCATCAGATGTGCCCAGATGGGATTAGCTAGTAGGTGGGGTAACGGCTCA CCTAGGCGACGATCCCTAGCTGGTCTGAGAGGATGACCAGCCACACTGGAACTGAGACACGGTCCAGACTCCTACGGGAG GCAGCAGTGGGGAATATTGCACAATGGGCGCAAGCCTGATGCAGCCATGCCGCGTGTATGAAGAAGGCCTTCGGGTTGTA AAGTACTTTCAGCGGGGAGGAAGGCGATGTGGTTAATAACCACGTCGATTGACGTTACCCGCAGAAGAAGCACCGGCTAA CTCCGTGCCAGCAGCCGCGGTAATACGGAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGCACGCAGGCGGTC TGTCAAGTCGGATGTGAAATCCCCGGGCTCAACCTGGGAACTGCATCCGAAACTGGCAGGCTTGAGTCTCGTAGAGGGGG GTAGAATTCCAGGTGTAGCGGTGAAATGCGTAGAGATCTGGAGGAATACCGGTGGCGAANGCGGCCCCCTGGACGAAGAC TGACGCTCANGTGCGAAAGCGTGGGGAGCAAACAGGATTAGATACCCTGGTAGTCCACGCCGTAAACGATGTCGACTTGG AGGTTGTGCCCTTGAGGCGTGGCTTCCGGAGCTAACGCGTTAAGTCGACCGCCTGGGGAGTACGGCCGCAANGTTAAAAC TCNNTGAATTGACGGGGGCCCGCACAAGCGGTGGAGCATGTGGTTTAATTCNATGCAACGCGAAGAACCTTACCTGGTCT TGACATCCACAGAACTT
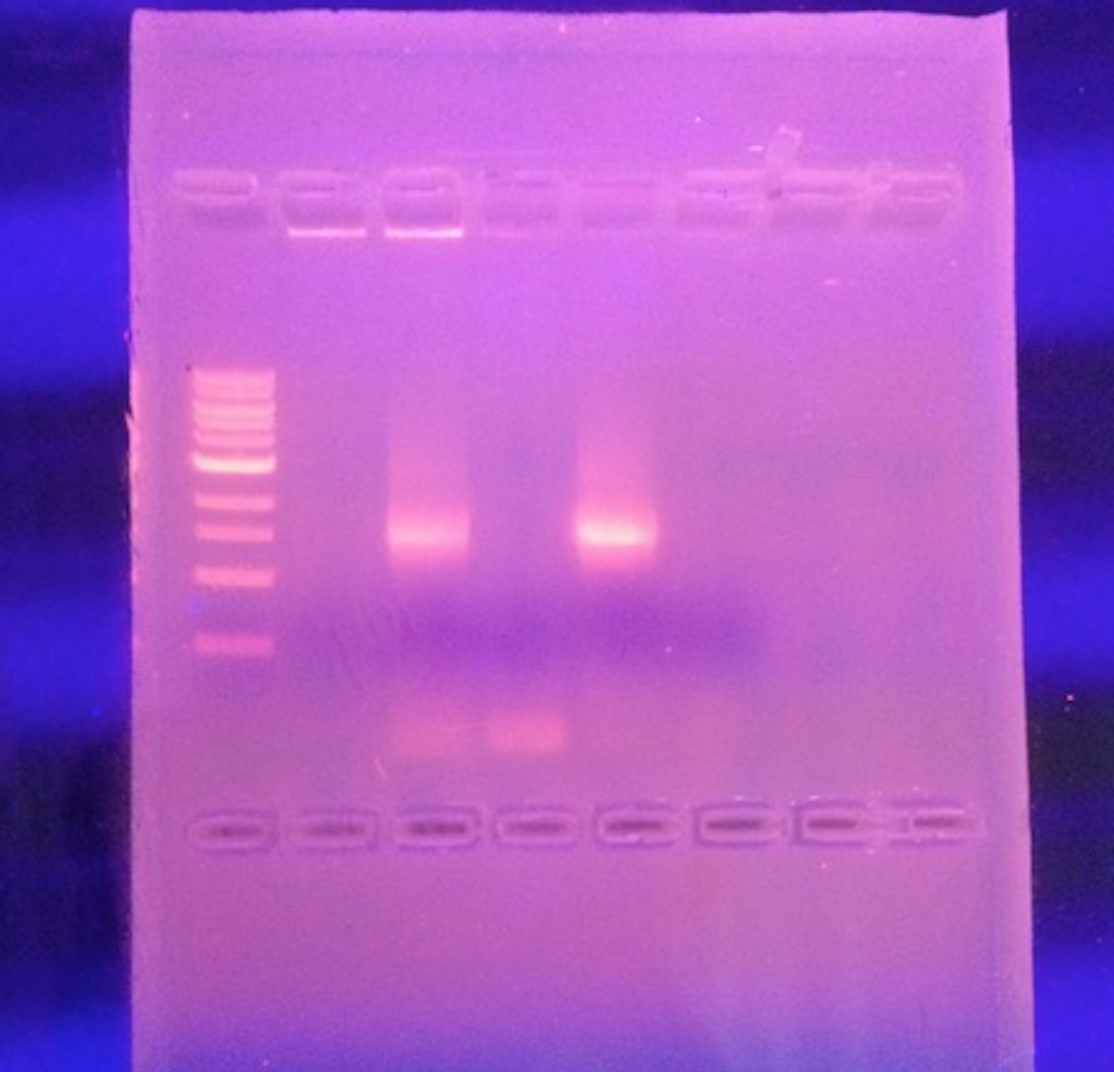
This sequence was a match with Salmonella bongori BS
Thursday February 25, 2016
Purpose The purpose for this experiment is to determine the physical characteristics of the zebrafish (that have grown into adults) in the two samples. One sample is composed of the control which is just water, where the second sample is the experimental sample which contains 1.5% ethanol concentration. By placing twenty-four zebrafish in each solution we can determine the effects of ethanol on early development of the zebrafish.
Materials & Methods 1.Identify and differentiate living and dead zebrafish
a. If dead preserve each dead fish in a small vial containing 0.02% tricane solution and 4% formaldehyde (label each vial)
2.Remove any egg shells to prevent mold growth/contamination 3.Add 2mL of either control or tested solution to each to refill 4.Observe any changes using a microscope 5.Test heart rate of the zebrafish 6.Feed the fish two drops of paramecium
Data & Observations We observed the physical differences between the two groups. The control group presented normal sized eyes, where the control group had larger protruding eyes. The control group had normal swimming patterns, the control group presented impaired motor skills and some of the zebrafish swam sideways. It was evident that there had been mental and physical effects of 1.5% ethanol concentration on the tested group. We observed the tested ethanol group to have grown legs and the control group had not. BS
Thursday, February 18, 2016
Purpose The purpose for this experiment is to identify and compare the characteristics of the zebrafish of the control group vs the tested group.
Materials & Methods 1. Put 2mL of 1.5% concentrated ethanol solution into each of the 24 slots of the dishes 2. Put 2mL of water into each of the 24 slots of the second dish 3. Use a pipette to get 48 zebra fish eggs 4. Place the eggs into a petri dish with some of the water from the original bowl of fish eggs 5. pipette 1 fish egg into each of the 24 slots of the control(water) 6. Pipette 1 fish egg into each of the 24 slots of the ethanol solution
Data & Observations We observed that the clear fish eggs were the fertilized ones. Those were the ones we would be using in this experiment. We then have to wait for the fish to hatch to observe the deformities caused by the ethanol concentration. We anticipate the size of the eyes of the zebra fish to be much larger than the fish of the control group. BS
Thursday, February 11, 2016
Purpose
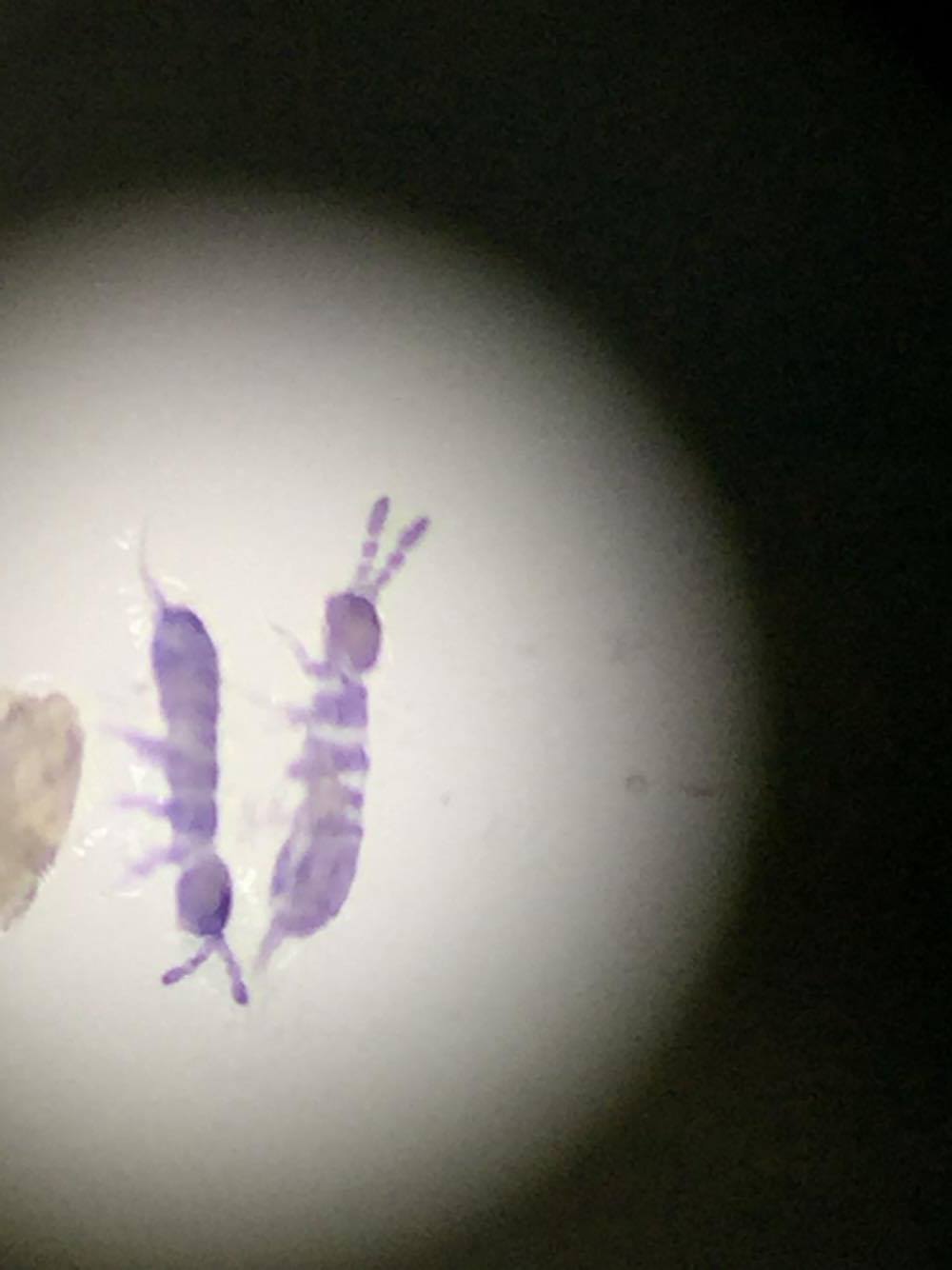
The purpose for this experiment was to observe the organisms living within our transect. We used Berlese Funnels to collect falling debris and organisms. From this we identified the invertebrates collected from the Berlese Funnels.
Materials & Methods 1. Collect insects from Berlese Funnel a. collect sample from top b. collect sample from the bottom 2. Using a microscope and a dichotomous key identify each organism in the sample 3. From the text book identify other organisms in the transect (vertebrates)
Data & Observations
We identified 4 organisms that lived in our funnel, they were: Flies Diptera, Fleas (Siphonaptera), Termites (Isoptera)
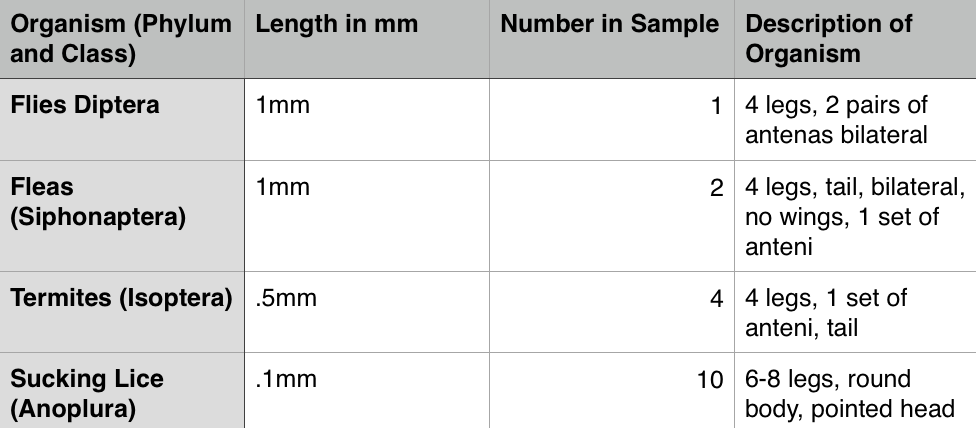
From this data we concluded the Berleses Funnel contains various types of invertebrate These organisms range from .1mm to 1mm there are 10 of the smallest organisms in the sample. This organism is the Sucking Lice (Anoplura). The largest organism in the sample was the Fleas (Siphonaptera) and the Flies Diptera these both were 1mm in length. All of these being insects, they were the most common organisms of the sample. The two samples we observed both contained insects they were relatively similar with the organisms living on the top sample and the bottom sample.

Five vertebrates that might inhibit this transect are Robins(Chordata>Aves>Passeriformes>Turdidae>Turdus>Turdus Migratorious) Cardinals(Chordata>Aves>Passeriformes>Cardinalidae>Cardinalis>C.Cardinalis) Field Mice(Chordata>Mammalia>Rodentia>Muroidea>Muridae>Murinae>Mus>M.Musculus) Squirrels (Chordata>Mammalia>Rodentia>Sciuridae>Sciurus>Carolinensis>Gmelin) and Garter Snakes(Chordata>Vertebrata>Reptilia>Squamata>Serpentes>Colubridae>Thamnophids>Thamnophis Elegans) The birds of this transect could feed off the worms and insects living in the soil and leaves, The squirrels can feed off of nuts and seeds from plants in the transect, the snakes can eat the insects and the rodents or field mice living around the transect. The field mice will look for food and plants to eat the biotic factors in the transect such as plants and soil will keep fresh and producing because of the organisms living in that location. Not many changes will happen to the abiotic lamp posts, benches, sidewalks, and rocks. These characteristics will mostly be stagnant in the transect. Food webs are a way to summarize energy flow and to record complex trophic interactions within an ecosystem.
BS
Thursday, February 4, 2016 Purpose The purpose for this experiment is to identify unique characteristics about plants that live in certain transects. By describing the characteristics of the organisms we collected such as vascularization, special structures, and the way they reproduce, we can then determine what genus the plants belong to that are living in this area. We can then specify the different types of plants and invertebrates and contrast them to fungi. From these samples we will make a Berlese Funnel to collect invertebrate.
Materials and Methods 1. Collect leave samples from transect location. a. 20 dead leaves b. 5 plant samples 2. Identify hight and vascularization of the plants 3.Determine seeds to be either monocot or dicot 4. Explain the fungi sporangia and its importance
Data and Observations
Characteristics of plants collected from the transect
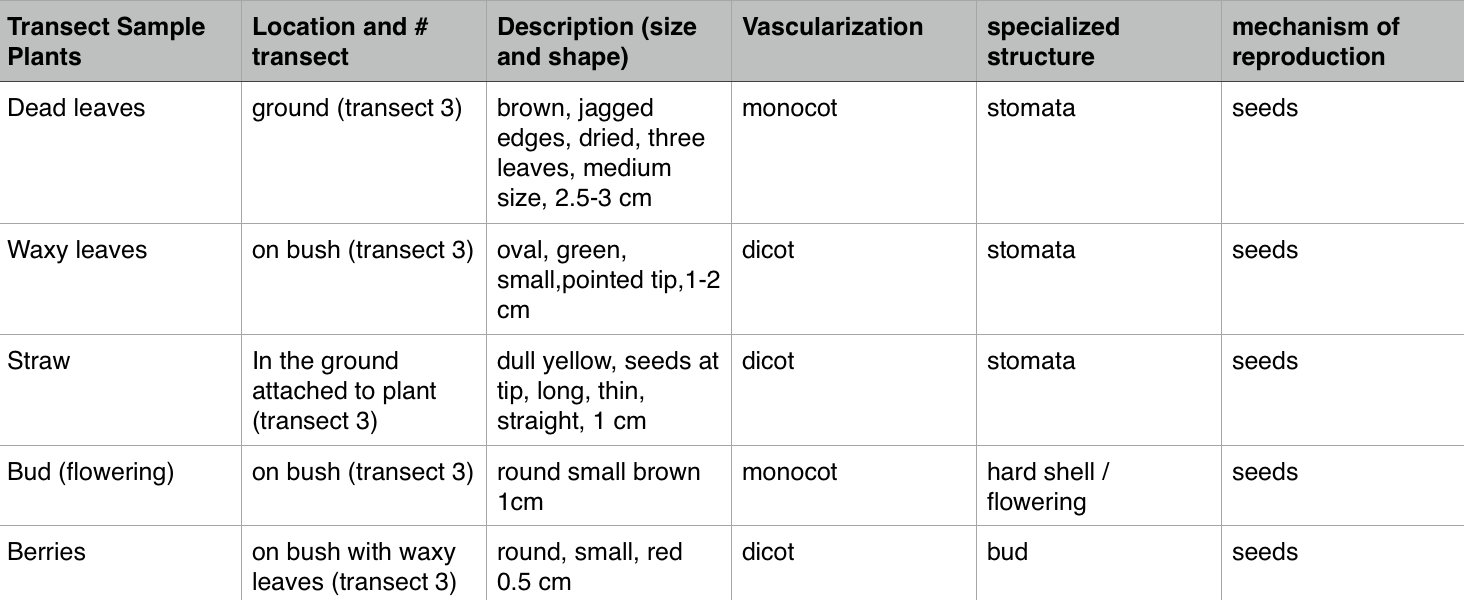
Fungi Sporangia(black mold): This type of mold is classified as a mycelia this type of mold creates spores, this is crucial to the development of fungi.

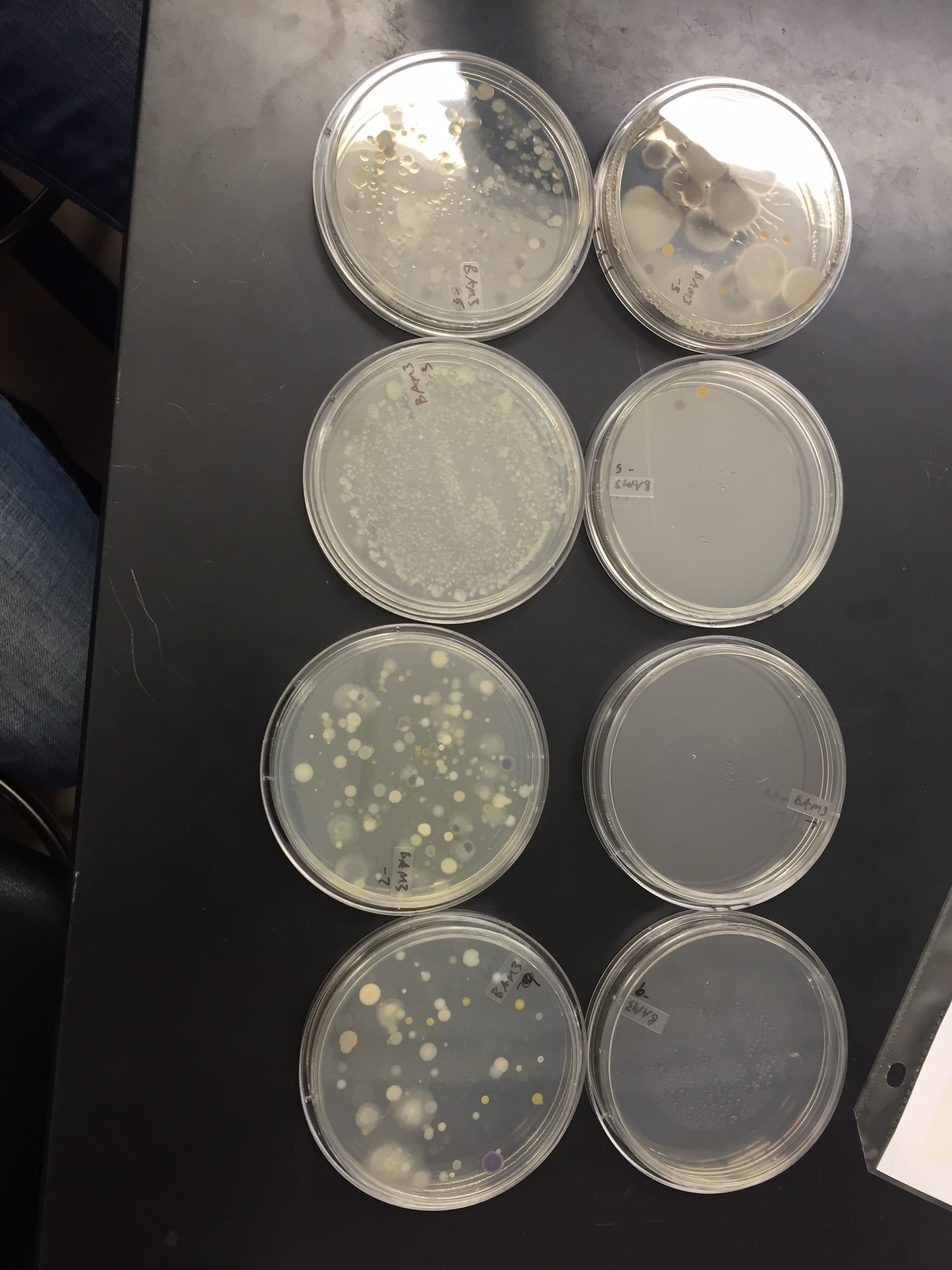
Thursday, January 28, 2016 The Hay Infusion Culture water is a clear/yellow ish color. The water has brown sediments of dirt on the bottom, and a film of bacteria, mold, and algae on the top. There is less water in the jar and the smell has become increasingly worse. The water smells like sewer or pond water. These changes are caused by microorganisms in the infused culture. The smell and appearance of the Hay Infusion Culture is hypothesized as a result from the growth of algae and mold on the top layer of the jars. From week to week this could chance due to the water and plant life incorporated in the jars. The archaea species will grow on the agar plates because single cells can become isolated on these plates allowing them to grow.
The number of colonies differ in how much bacteria was presented on certain plates. It was observed that plates containing the tetracycline had more resistance to the growth of bacteria and fungi colonies unlike the agar plates that did not contain the antibiotic tetracycline. Plates that did not contain tetracycline grew many colonies varying in size and color. The two groups contained serial dilutions ranging from 10^-3 to 10^-9 (only counting odd numbers) The first group did not contain the tetracycline and their colonies were 10^-3=880 10^-5=213 10^-7=150 and 10^-9=80. From this data we observe that the larger the dilution number the more resistant the bacteria and fungi are to growing on the agar plate. The second group did contain the tetracycline and their colonies were 10^-3=28 10^-5=1 10^-7=0 and 10^-9=0. From this data we can conclude that the lower the tetracycline dilution was the higher the chance of forming colonies on the agar plates. Overall two of the species were unaffected by the tetracycline and two were not.
Tetracyclines are broad-spectrum agents, exhibiting activity against a wide range of gram-positive and gram-negative bacteria, atypical organisms such as chlamydiae, mycoplasmas, and rickettsiae, and protozoan parasites.Tetracyclines inhibit bacterial protein synthesis by preventing the association of aminoacyl-tRNA with the bacterial ribosome.
Chopra,I. June 2001 "Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance". Journal of Microbiology and Molecular Biology Review. Volume 65(2).
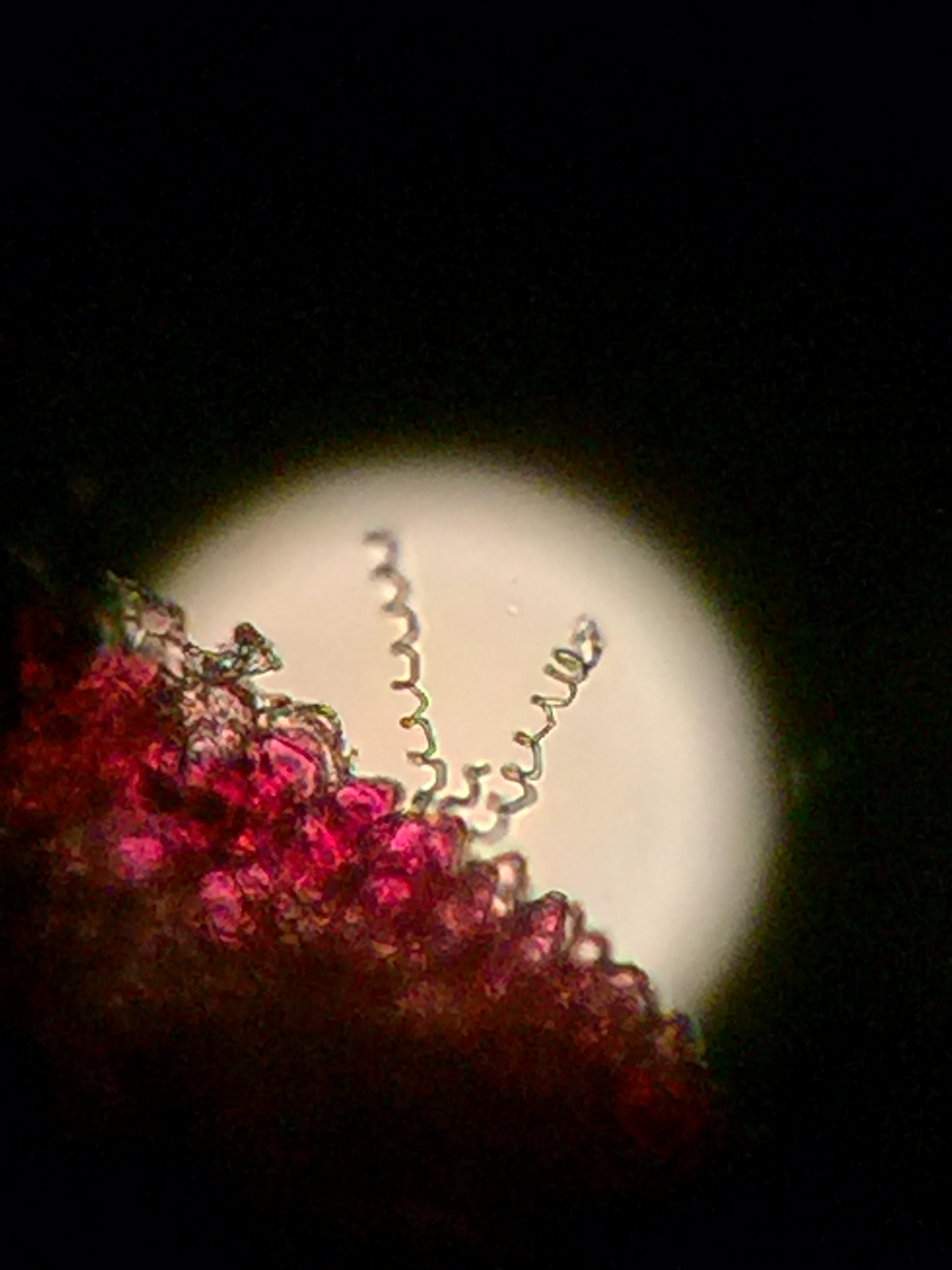
The 4 cells were similar and different. The first cell was 10^-7 this cell did not contain tetracycline the cell was pink and spotted, it was non-motile, circular, gram positive and it was toward the edge of the slide. this cell was 20x2.5 ocular spaces. The second cell was 10^-9 this cell did not contain tetracycline the cell was purple and had many colonies, it was non-motile, circular, gram positive and this cell was 10x2.5 ocular spaces. The third cell was 10^-5 this cell contained the tetracycline. The colony was light to dark purple, non-motile, oval/circular, gram positive and was roughly 1/2x2.5 ocular spaces. The fourth cell was 10^-3 this cell did not contain tetracycline. The colors were light purple and clear, the cell was non-motile, had jagged edges and contain rod shaped structures. this cell was gram positive and was roughly 1x2.5 ocular spaces BS
Thursday, January 21, 2016 The Hay Infusion Culture smells like a sewer. Its appearance is moldy; mucky; cloudy. On the top of the jar there is a film of mold. It is apparent that organisms live closer to the surface because theres oxygen and further away from the plant matter because it was submerged in solution. The protist and algae that are present are, (In the top layer) Gonium at 12.5 ocular spaces is a motile algae organism. Pandorina at ... ocular spaces is a motile algae organism as well. Colpidium at ... ocular spaces, is a non motile protozoan organism. (In the middle layer) there are Volvox at ... ocular spaces, this organism has characteristics of both protozoa and algae. This organism is motile by the use of flagella. Paramecium Bursaria at ... ocular spaces, is a motile protozoan organism. Spirostomum at ... ocular spaces, is a motile, protozoa, organism that uses cillia. (In the bottom layer) there are Blepharisma at ... ocular spaces that is a motile protozoa. Didinum Cyst at ... ocular spaces are protozoan organisms. Chilomonas Sp. at ... ocular spaces, is a protozoan organism. If the Hay Infusion Culture were to grow for another two months we would find many forms of life growing on the surface of the jar. We would also have other forms of protozoa and algae living off of the plant life in the jar. The carrying capacity for certain organism could exceed others creating extinction of some organisms and over population of others. BS
Thursday, January 14, 2016 The 20 x 20 foot dimension transect we observed as in the garden located adjacent to the Butler Pavilion. There was many abiotic factors such as rocks, dirt, and twigs. The biotic factors included Grass, plants, leaves, and small organisms in the soil. The items were scattered throughout the transect. BS