User:Sithara Thalluri/Notebook/Biology 210 at AU
PCR DATA
Purpose: The purpose of this lab was to isolate DNA samples of bacteria from our transect, and use primers and PCR to amplify the 16S rRNA gene in order to correctly identify bacteria.
Materials and Methods: Students transferred a single colony of bacteria to a 100 μL of water in a sterile tube. The tubes were then incubated at 100°C for 10 minutes in a heating block, floating in water. Samples were then centrifuged for 5 minutes at 13,400 rpm. While samples were centrifuged, 20 μL of a primer and water solution was added to a labeled PCR tube and mixed to dissolve the PCR bead. Then, 5 μL of the supernatant from the centrifuged samples were transferred to the 16S PCR reaction and placed in to the PCR machine. A week later, students ran PCR products on an agarose gel. Then samples were sequenced and able to be identified.
Data and Observations
>MB47-For_16S_G06.ab1 NNNNNNNNNNNNNNNNNCTNNNCNTGCAGCCGAGCGGTAGAGATTCTTCGGAATCTTGAGAGCGGCGCACGGGTGCGGAA CACGTGTGCAACCTGCCTTTATCAGGGGAATAGCCTTTCGAAAGGAAGATTAATGCCCCATAATATATCATATGGCATCA TTTGATATTGAAAACTCCGGTGGATAAAGATGGGCACGCGCAGGATTAGATAGTTGGTAGGGTAACGGCCTACCAAGTCA GCGATCCTTAGGGGGCCTGAGAGGGTGATCCCCCACACTGGTACTGAGACACGGACCAGACTCCTACGGGAGGCAGCAGT GAGGAATATTGGACAATGGGTGAGAGCCTGATCCAGCCATCCCGCGTGAAGGACGACGGCCCTATGGGTTGTAAACTTCT TTTGTATAGGGATAAACCTACCCTCGTGAGGGTAGCTGAAGGTACTATACGAATAAGCACCGGCTAACTCCGTGCCAGCA GCCGCGGTAATACGGAGGGTGCAAGCGTTATCCGGATTTATTGGGTTTAAAGGGTCCGTANGCTGATGTGTAANTCANTG GTGAAATCTCACANCTTANCTGTGAAACTGCCNTTGATACTGCATGTCTTGAGTGTTGTTGAANTANCTGGAATAANTNN GTANCAGTGAAATGCCTANATATTACTTNNANCACNANGTGCTAANGCANGTTGGTANNCCNCNACTGACNCTGATNGAG NAAANCNTGGGNNAGCGAACANAANTNNATACCCTGGGGNNGTNNNCNNNAANNAANCTNANTCNNTTTTTNTCTTTCTC TTNCNNATACANNNNNANCCGANAAGNTNGCCNNCTNCCGGGTGGTGTTCTCCNTNNTNNNGATGNNNTCNNCTNNNNNN NNNNNCNGCCCCCCCNCAANNATTTNTANANNNNTATANNNTNNNANANCNNGCGGCCCCCTNTNTAANNGGNNNNGGGG GAGNNNNNNGNNNNNGTTTTCTATTATATNTNNNNCTNTNNNCCNCNNGNNCNGGGGGGGTTGTNTCTCCCNNCCAGAAC NNAANGANANTNTNCNNCANCAGCCNNNN
Chrseobacterium (above): MB 47
>MB48-For_16S_H06.ab1 NNNNNNNNNNNNNNGCNNANNNTGNNANNNNNGCGGTANGANGGGANGCTTGCTCTNNGATTCAGCGGCGGACGGGTGAG TAATGCCTAGGAATCTGCCTGGTAGTGGGGGACAACGTTTCGAAAGGAACGCTAATACCGCATACGTCCTACGGGAGAAA GCAGGGGACCTTCGGGCCTTGCGCTATCAGATGAGCCTAGGTCGGATTAGCTAGTTGGTGAGGTAATGGCTCACCAAGGC GACGATCCGTAACTGGTCTGAGAGGATGATCAGTCACACTGGAACTGAGACACGGTCCAGACTCCTACGGGAGGCAGCAG TGGGGAATATTGGACAATGGGCGAAAGCCTGATCCAGCCATGCCGCGTGTGTGAAGAAGGTCTTCGGATTGTAAAGCACT TTAAGTTGGGAGGAAGGGCATTAACCTAATACCTTGGTGTTTTGACGTTACCGACAGAATAAGCACCGGCTAACTCTGTG CCAGCAGCCGCGGTAATACAGAGGGTGCAAGCGTTAATCGGAATTACTGGGCGTAAAGCGCGCGTANGTGGTTTGTTAAG TTGGATGTGAAAGCCCCGGGCTCAACCTGGGAACTGCNTTCNAAACTGNCNAGCTAGAGTATGGTANAGGGTGGTGGAAT TTCCTGTGTAGCNNTGAAATGCGTAGATATANGAANGAACACNNNGTGGCNAAGCGGACCACCTGNACTGATACTNACAC TGANNTGCGAAANNNTGTGGANCAAACANNATTANGATNNCCTNNAGTCCACNGCCNGTANACNNNNTCAACTANCNNNN NNAGCNCTTNANNTGTTANTGNCGCNNCTAACNCATTAANTNNCNNCGCTGGNTNNGTAGNAGNCCNCGNCCGTTAGNNC TNNNNNNGGAGTTNANNGGNGCCNNGCACAAGCNACTGNAGCAGGGNGGGNTGTAGTTCCNAANNNNNNACNAAAAANNN NNACCCNGNCCCTNGGNATNNAANNNAGNNNNNGAGGNNNNNNAANNNGNGNNNNGGNTGNNNNCNNNGGAAANNNNACC ANNNGNNNATGGTNGGNNNNNNNCNNCANNNNNNCNANCCCNNNNNNN
Pseudomonas MB 48
Conclusions
In conclusion, the 16s sequencing which identified the bacteria matched with previous attained observations, although generally poor in quality.
For the first sequence, it is determined that the species of bacteria was Chryseobacterium. This is in agreeance with previous observations: chryseobacterium is gram negative, circular, and slow motility. The colors of the bacterium is variable, but includes the white color observed.
In the second sequence it was determined that the bacteria was Pseudomonas aeruginosa. Pseudomonas are fairly motile, bacillus or coccus in shape and are gram negative - this is in accordance with previous observations as well.
Lab #6 Zebrafish
Purpose: The purpose of this lab is to learn the stages of embryonic development, compare embryonic development in different organisms, and set up an experiment to study how environmental conditions affect embryonic development.
Materials and Methods: Students first read a published paper about the affect of their given treatment, in this case - nicotine, on zebrafish embryos. From these papers we were able to make predictions, hypotheses, and experimental plans. On the first day of the experiment, students set up two groups of zebrafish, a test group and a treated group - thus tested only one variable, nicotine. To do so, two petri dishes were acquired. One dish was filled with distilled water, and the other filled with pre-made nicotine solution. Then 20 healthy translucent embryos were placed in each petri dish. An observation schedule was arranged.
Data and Observations
| Control | Day 0 (2/19) | Day 1 (2/20) | Day 4 (2/23) | Day 7 (2/26) | Day 11 (3/2) | Day 13 (3/4) | |||||||
| # of Dead Eggs | 0 | 0 | 4 | 1 | 0 | 0 | |||||||
| # of Live Eggs | 20 | 20 | 8 | 0 | 0 | 0 | |||||||
| # of Live Hatchings | 0 | 0 | 5 | 10 | 9 | 9 | |||||||
| # of Dead Hatchlings | 0 | 0 | 0 | 1 | 1 | 0 | |||||||
| Stages of Development | Zygote | Zygote | 8-->25-somites, 5--> high spec | 10--> protuding mouth | - | 0 | |||||||
| # Disappeared | 0 | 0 | 3 | 0 | 0 | 0 | |||||||
| Degree of Body/Tail Pigmentation | - | - | - | - | yellow, with black dots, straight | bits of yellow by head, blacks spots down body, striaght | |||||||
| tail= 28.7 5μm body=41.25 μm | |||||||||||||
| Eyes/Eye Movement | - | - | - | - | strong, fast | rapid eye movement, eyes= 2.5 μm | |||||||
| Heart Rate | - | - | - | - | - | 56 bpm | |||||||
| General Movement | - | - | - | - | jittery movement, fast reaction time (1sec) | constantly moving, fast reaction time (1sec) | |||||||
| Feces | - | - | - | - | mild, few piles (5) | 4 piles | |||||||
| - | |||||||||||||
| Treated with Nicotine | Day 0 (2/19) | Day 1 (2/20) | Day 4 (2/23) | Day 7 (2/26) | Day 11 (3/2) | Day 13 (3/4) | |||||||
| # of Dead Eggs | 0 | 6 | 5 | 0 | 0 | 0 | |||||||
| # of Live Eggs | 20 | 20 (added 6) | 1 | 1 | 0 | 0 | |||||||
| # of Live Hatchings | 0 | 0 | 10 | 9 | 8 | 5 | |||||||
| # of Dead Hatchlings | 0 | 0 | 0 | 0 | 0 | 0 | |||||||
| Stages of Development | Zygote | 17-somites | 1--> 21 somites, 10--> high spec | 1-->25-somite 9--> protuding mouth | 0 | 3 | |||||||
| # Disappeared | 0 | 0 | 4 | 1 | 2 | 0 | |||||||
| Degree of Body/Tail Pigmentation | - | - | - | - | either much more yellow w/ black dots or not yellow with black dots | body bent, minimum yellow,most clear with black dots down back | |||||||
| body was bent | tail=25 μm body= 40 μm | ||||||||||||
| Eyes/Eye Movement | - | - | - | - | slow, delayed | no eye movement, eyes=2.75 μm | |||||||
| Heart Rate | - | - | - | - | - | 60 bpm | |||||||
| General Movement | - | - | - | - | float around, very slow reaction time (3 sec) | floating, not much movement, slow reaction time(3), one was crazy | |||||||
| Feces | - | - | - | - | Lots of piles (20) | lots of piles (25)
|
Tail | Body | Eye | General Observations | |||
| Fixed Control | 25 μm | 37.5 μm | 3.75 μm | furry, clear no black dots, moldy? | |||||||||
| Fixed Treated | 25 μm | 42.5 μm | 2.75 μm | bent, black dots on back |
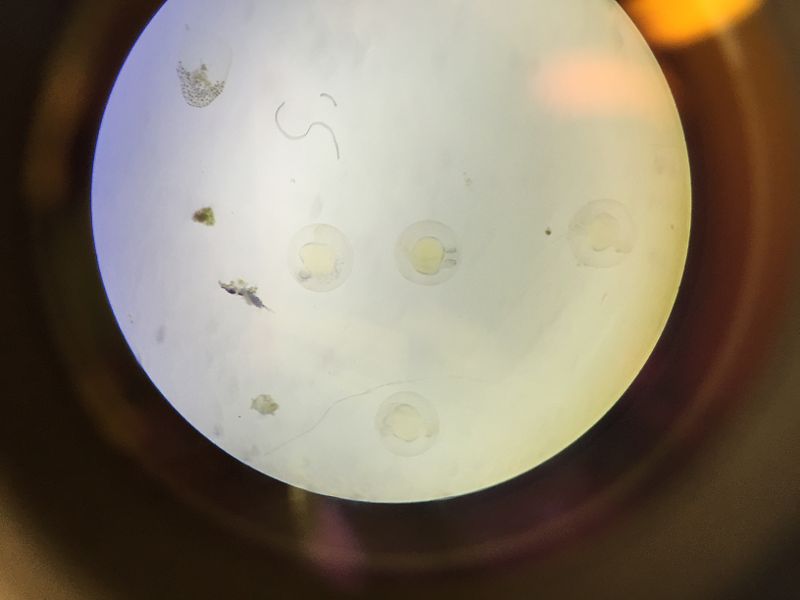
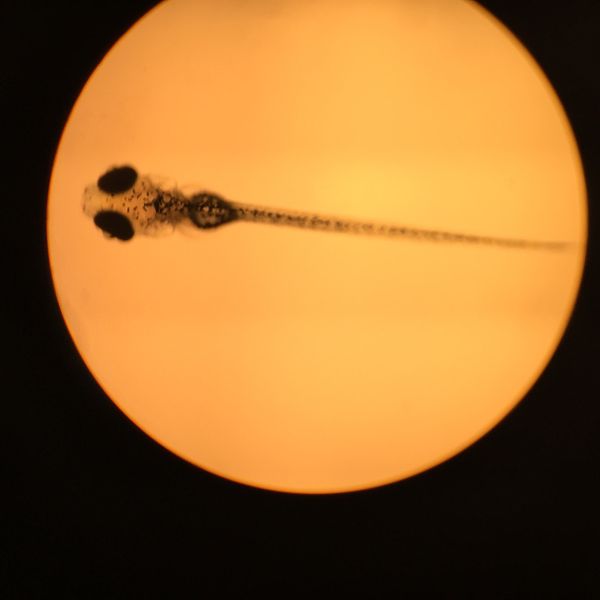

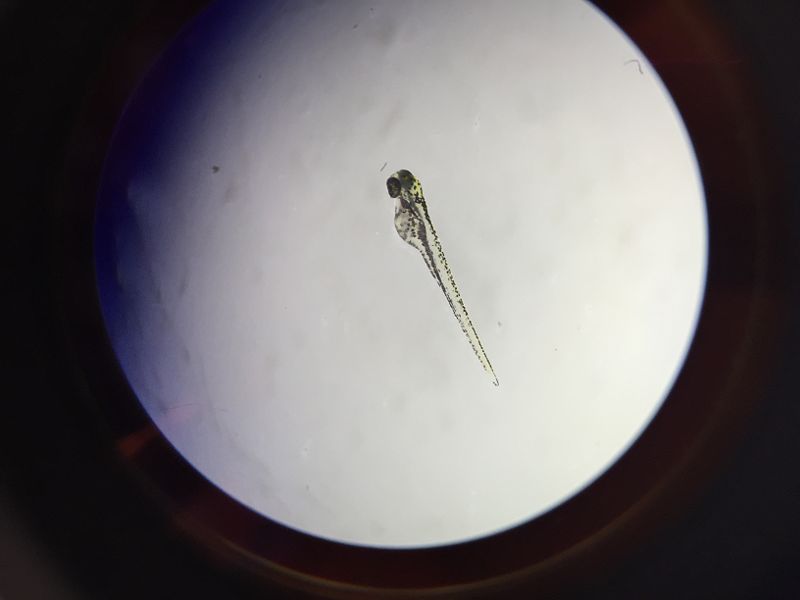
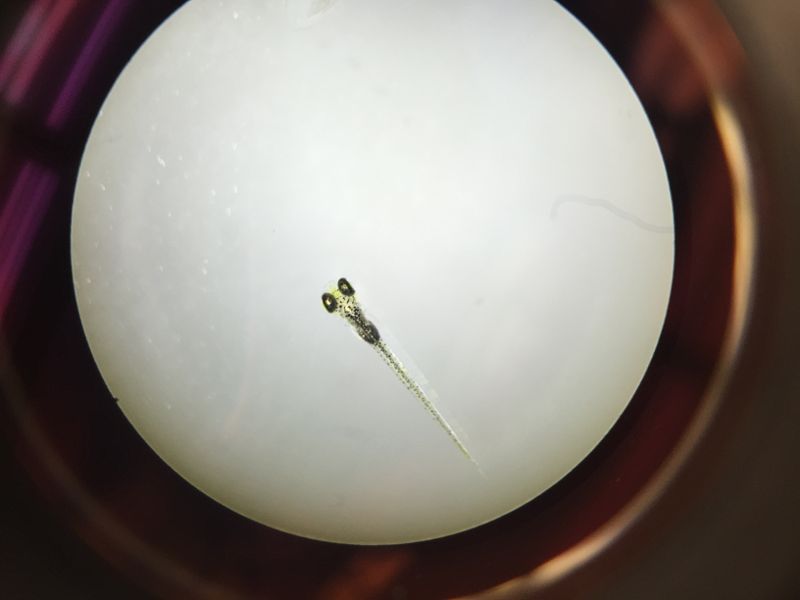
Conclusions and Future Directions Past research concluded that placing zebrafish embryos in a nicotine solution would negatively impact their survival rate through reduces their response time, eye diameter, tail length, and heart rate. In accordance to past experiments, the zebrafish treated with the nicotine solution had a higher mortality rate as well as reductions in other physical traits crucial to survival.
2.20.15
Very good lab book entry.
Detailed description of procedures and invertebrates found. Well organized, especially the contents section at the top. Nice food web and good consideration of the fence at transect.
SK
Lab #5 Invertebrates
Purpose The purpose of this lab was to observe invertebrates in order to understand their importance and to learn how simple systems evolved in to more complex systems.
Materials and Methods
Procedure I: In the first procedure students observed prepared slides of cross sections of Acoelomates, Psuedocoelomates, and Coelomates under a microscope and noticed their movement mechanisms and other body structures.
Procedure II: During this procedure, students observed example organisms from the classes: Arachnida, Diplopoda, Chilopoda, Insect, and Crustacea. Students observed the differences in these organisms such as body parts, body segments, and number of appendages.
Procedure III: In procedure three, students analyzed the invertebrates that were collected from our given transects through the breaking down of Berlese Funnels. First, students poured the top 10-15 mLs of the funnel liquid in to a petri dish, then the remaining liquid in to another - labeling them top and bottom respectively. Students then examined each petri dish under a microscope and identified each organism using a key.
Procedure IV: Finally students considered vertebrates that inhabit and pass through their given transects and analyzed their presence in the transect - which is explained further in the data below.
Data and Observations
[[The table below show