User:SKaret/Notebook/GroupProj/PromRes
From OpenWetWare
Jump to navigationJump to search
Factors affecting output from genetic gene circuits:
- Choice of transcription factor
- Choice of core promoter
- Operator sites
- Number of sites
- Spacing between
- Binding affinity
- Transgene copy number
- Integration into the genome or expression from plasmids
- Promoter activity
- Ribosome-binding sites
- Codon bias of the host
- Transcription rate and tRNA abundance
- Half-life of mRNA
- Substrate and cofactor availability
- Adjustment of enzyme kinetics
- Protein scaffolding
- Sub-cellular localization through the use of microcompartments
These affect expression dynamics, namely strength, leakiness and sensitivity of a promoter
Constitutive promoters:
- Desirable features: different and predictable activation characteristics.
- Problems:
- Few sequences available
- Do not perform as required
- Not transferable to new chassis
Inducible promoters:
- Concentration of inducer can be changed to achieve desired protein production levels
- Problems:
- Promoter hypersensitivity
- Cost (industrial)
- Heterogenous expression levels
- Variations in transgene expression levels, even in clonal populations
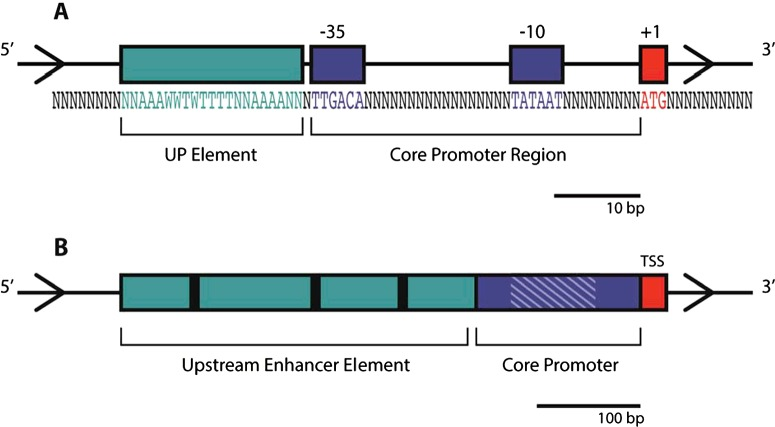
Structure of the prokaryotic promoter (E. Coli)
- Red: Transcription start site (TSS)
- Blue: Conserved hexamers
- 10 and 35bp upstream of TSS
- Key binding sites for RNA Polymerase
- In some species a consensus length of 17bp between the conserved hexamer sequences is found
- Green: UP elements
- Adenine/thymine rich
- Boosts transcription rate by interacting with the C-terminal domain on the a subunit of the RNA polymerase
- KEY:
- N = any deoxynucleotide
- W = Adenine or Thymine
- A = Adenine
- T = Thymine
- C = Cytosine
- G = Guanine
Design principles for a synthetic promoter
1. Number of spacing between response elements:
- Transcription increases with number of operator sites 5’ of the minimal promoter.
- Spacing between response elements, and between response elements + minimal promoter increases steric hindrance
- Changes orientation of bound TF to the minimal promoter
- Steric hindrance = slowing of chemical reactions due to bulk
2. Variants of response elements
- Modified to increase or decrease binding affinity of TF to their cognate sequence
- Enhanced binding --> increase transcription initiation
- Reduced binding --> decrease transcription initiation
3. Choice of minimal promoter
- Modifies efficiency of RNA polymerase recruitment (to TSS)
- Influences basal and maximal transgene expression
4. Choice of reporter gene
- Allows quantification of the upstream promoter
- Intracellular proteins: Faster turnover rates Lower basal levels
- Secreted proteins: monitor gene expression over time
Previous years project: future directions:
- Parts containing B0032/34 RBS and corresponding plasmids had mutations or missing DNA sequences
- Implement the toggle switch in the lab and carry out stability and robustness testing
- Application; Production of essential oils for use in fragrances via metabolic engineering