User:Randy Jay Lafler/Notebook/Physics 307L/Millikan Oil Drop
Steve Koch 05:20, 21 December 2010 (EST):Very good primary notebook. I can follow it easily. I agree that focusing probably major issue. With more time you would get better data.
Purpose
The purpose of this experiment was to use the Millikan oil drop apparatus to discover the charge on individual oil drops. We are then supposed to notice that the charges are a multiple of some elementry, or minimal charge.
Set up and procedure
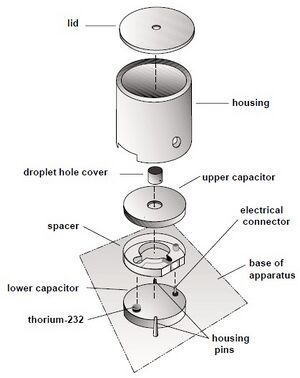
- First we leveled the plateform for the Oil Drop Apparatus.
- We measure the width of the plastic spacer with a micrometer. This is the plate separation.
- Then we focused the viewing scope by using the focusing wire and adjusted the light.
- We connected the power supply to the plate voltage connectors using bannana plug patch plugs and applied 500V. We also connected a multimeter in parralel with the apparatus and power supply to better monitor the voltage.
- We connected another multimeter to the thermistor connectors to measure the resistance in the thermistor. This gave us the temperature within the viewing chamber.
- We used the atomizer to introduce oil drops into the chamber. This was done with the ionization source lever in the Spray Droplet Position, which allowed air to escape the chamber.
- Once there were droplets in the chamber we moved the ionization source lever to the Off Position. We tried to find a droplet with a fall velocity between 7 and 15 seconds. This was one of the hard parts of the experiment.
- When we chose a drop we measured as many fall and rise times as we could. The fall times are the times it takes the drops to fall from one major reticule line to the next. The major reticule lines are 0.5 mm apart. The rise times are the same, but with the voltage applied between the plates so that the drops rise against the force of gravity.
- For the last oil drop we measured several fall and rise times and then moved the ionization lever to the ON position and measured several more fall and rise times.
Equipment



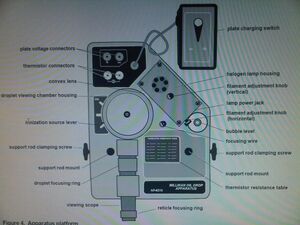
- Millikan Oil Drop Apparatus
- Tel-Atomic (50V and 500V power supply)
- Oil of density 886 Kg/m^3
Data
Particle 2
- averages calculated by summing up the consistent times and dividing by the number of times. I say consistent because some of the times calculated for particle 5 have large jumps in the rise times.
- I computed the standard diviation by taking the difference of the data point and the average value, squaring the result, adding up all these values and dividing by the number of trials, and then taking the square root of the whole thing.
- average fall time = 12.68s
- standard div. fall time = 0.963s
average fall velocity [math]\displaystyle{ v_f=3.94(28)*10^-5m/s\,\! }[/math]
- average rise time = 3.63s
- standard div. rise time = 0.128s
average rise velocity [math]\displaystyle{ v_r=1.38(5)*10^-4m/s\,\! }[/math]
calculating the charge for particle 2
- I calculated the charge q in three parts.
- First I calculated the radius a of the oil drop
- [math]\displaystyle{ a=\sqrt{(b/2p)^2+9nv_f/2gp_r}{-b/2p}\,\! }[/math]
- [math]\displaystyle{ a=5.65(21)*10^-7m\,\! }[/math]
- Then I calculated the mass
- [math]\displaystyle{ m=4*pi*a^3*p_r\,\! }[/math]
- [math]\displaystyle{ m=6.70(78)*10^-16Kg\,\! }[/math]
- Finally I calculated the total charge
- [math]\displaystyle{ a=\frac{mg(v_f+v_r)}{(V/d)v_f}\,\! }[/math]
- [math]\displaystyle{ q=4.25*10(40)^-19C\,\! }[/math]
Elementry charge
- Knowing that the accepted value for e is [math]\displaystyle{ e=1.6*10^-19C\,\! }[/math], it appears that particle 2 has 3e.
- Dividing q by 3 I get:
- [math]\displaystyle{ e=1.4(1)^-19C\,\! }[/math]
Particle 4
- average fall time = 7.80s
average fall velocity [math]\displaystyle{ v_f=6.41*10^-5m/s\,\! }[/math]
- average rise time = 1.58s
average rise velocity [math]\displaystyle{ v_r=3.16*10^-4m/s\,\! }[/math]
Calculating charge on particle 4
- I did not calculate the standard diviation for particle 4
- The radius a of the oil drop
- [math]\displaystyle{ a=7.33*10^-7m\,\! }[/math]
- The mass
- [math]\displaystyle{ m=1.46*10^-15\,\! }[/math]
- The total charge
- [math]\displaystyle{ q=1.22*10^-18C\,\! }[/math]
Elementry charge
- The accepted value for e is [math]\displaystyle{ e=1.6*10^-19C\,\! }[/math], it appears that particle 4 has 1e.
- [math]\displaystyle{ e=1.2*10^-19C\,\! }[/math]
Particle 5
- average fall time = 18.64s
- I neglected the time of 14s.
- standard div. fall time = 3.31s
average fall velocity [math]\displaystyle{ v_f=2.68(40)^-5m/s\,\! }[/math]
- average rise time = 3.56s
- I neglected the time of 10s
- standard div. rise time = 0.90s
average rise velocity [math]\displaystyle{ v_r=1.4(3)*10^-4m/s\,\! }[/math]
Calculating charge on particle 5
- The radius a of the oil drop
- [math]\displaystyle{ a=4.6(4)^-7m\,\! }[/math]
- The mass
- [math]\displaystyle{ m=3.6(1.1)*10^-16\,\! }[/math]
- The total charge
- [math]\displaystyle{ q=2(2)*10^-19C\,\! }[/math]
Elementry charge
- The accepted value for e is [math]\displaystyle{ e=1.6*10^-19C\,\! }[/math], it appears that particle 5 has 1e.
- [math]\displaystyle{ e=2(2)^-19C\,\! }[/math]
Analysis
- We had a lot of error in the calculations for particle 5. Our fall and rise times were not very consistent and it is apparent in the large error I obtained in the charge measurement.
- Our data has the least error, standard diviation, for particle 2. I also calculated an e of particle 2 that is closer to the accepted value of e that for the other oil drops. We observed lots of strange things happen to the oil drops after the second oil drop. The oil drops would disappear or they would fall at very inconsistent times. When we applied a voltage, some drops would accelerate down and some would go up. This might be why the measurements for particle 2 were the most consistent.
- Another reason for our error could because we very adjusting the wrong part of the viewing scope, and may have been changing the distance between the large reticule lines. We adjusted the droplet focusing ring instead of the reticule focusing ring while we took measurements. I used 0.5mm in all of our calculations anyway.SJK 05:18, 21 December 2010 (EST)

05:18, 21 December 2010 (EST)
This is a very good point. - Our largest error in all our calculations is probably due to our fall times, because the particles never fell straight down but drifted from side to side.
- Our measurements for e were to the right order of magnitude, but I think we should have gotten much better measurements. I think if we he had not adjusted the viewing scope we would have been much closer.