User:R. Morgan Taylor/Notebook/Biology 210 at AU
3/25/15 Zebrafish Experiment Analysis
Purpose
When an embryo develops normally the organism ends up without deformities. Because this is such a crucial time for organisms, development of embryos usually takes place in a secure environment; for example, a human embryo forms in the womb of their mother and a chick embryo develops inside an egg. The womb and the egg are both examples of protections for the zygote so that they are not exposed to something that will make them sick. Exposure to chemicals, high or low temptetures, or lack of light can all impede the development of an embryo. Similarly to the development of the human and chick zygotes, the Zebra fish embryo forms in an egg casing, and then they break free of it. The egg casing around the Zebra fish protects it from harmful chemicals, light and temperatures. Zebra fish are an extremely important in biology because they are a test system (a model organism) used to prescreen certain chemicals and drugs before they are exposed to humans (Herrmann, 1995). This is because Zebra fish tend to have the same reactions to chemical and drugs that humans have. I hypothesize that exposing Zebrafish embryos to Retinoic Acid during development will cause an increase of deformities among the fish. If the Zebrafish show neural deformities and decreases in mobility, then Retinoic acid impedes the development of the Zebrafish.
Materials and Methods
Day 0
On the first day of the experiment a control and test group were set. The control was set first by adding 20 ml of deerpark water to a petri dish. 20 clear embryos were selected and placed into the petri dish. The test was set by adding 20ml of Retinoic acid to the second petri dish. After the acid was added 20 clear embryos were chosen and placed into this petri dish. The petri dishes were labeled as “control” and “test.” After recording intial stages of development and checking to make sure that all embryos were viable the petri dishes were sealed with tape and placed on a tray for keeping. After the intial set of the experiment the embryos were observed for stage of developmet, coloration, length and mobility every Monday, Friday and Wednesday. In total, the fish were observed 7 times.
Day 2
Both the test and control groups were obtained from storage and examined under a dissection scope. Coloration and appearance of the body, tail and eye along with criteria denoted in the lab manual on page 61 were recorded. A movement scale was developed ranging from 0-10, with 10 being the quickest and 0 not moving. After observations were made two fungi were removed from the control dish using a dropper and placed into a waste container. Next 10ml of retinoic acid was removed from the test dish and 10ml of water was removed from the control dish and placed into a waste container. 25ml of retinoic acid was added to the test dish and 25ml of water was added to the control dish. The petri dishes were then resealed with tape and placed into a plastic container with a wet paper towel on the bottom.
Day 5
Number of alive and dead fish in each dish was recorded. The live fish were observed under the dissection scope for criteria listed in the lab Manuel on page 61(Bentley et al 2015). 10 ml of water was removed from the control group and 10 ml of retinoic acid was removed from the test group. 25 ml of water was then placed into the control group mad 25ml of retinoic acid was added to the test group.
Day 7
Dishes were checked for number of alive and dead fish as usual and categorical data was recorded. Representative embryos from both the test and control dishes were placed into a depression slide and observed under the compound microscope on 4x. Response to stimuli was observed by approaching a representative fish with a dropper and see how rapidly it moved away. A representative from both control and test were fixed. A dropper was used to place the fish into two separate tubes. 1 drop of trichina solution was added along with 1ml of water. The paraformaldehyde was added to the tube and put into storage.
Day 9
Number of alive and dead fish was observed. Dead fish were removed using a dropper and placed into a waste container. Representative fish from both control and test were placed into a depression slide and viewed under the compound microscope on 4x. Criteria noted on days 5 and 7 were observed again. Afterwards, Petri dishes were resealed and placed into the plastic bin.
Day 12
Number of alive and dead fish was observed. Dead zebrafish were removed from both dishes with a dropper and placed into a waste bin. Remaining live fish were obtained from both the test and control dishes and viewed under the compound microscope. The criteria that were observed on days 4 and 7 were again recorded and compared.
Day 14
For the final day of observations fixed samples were made from the control group and were observed. The sample was placed into a depression slide and viewed under a compound microscope. Coloration, body length, tail length, eye size, eye shape and eye color were all observed. After observing the fixed specimen all the dead organism were placed into a waste container. The liquids were then placed into a separate waste container for liquids. The dishes were disposed of.
Data and Observations
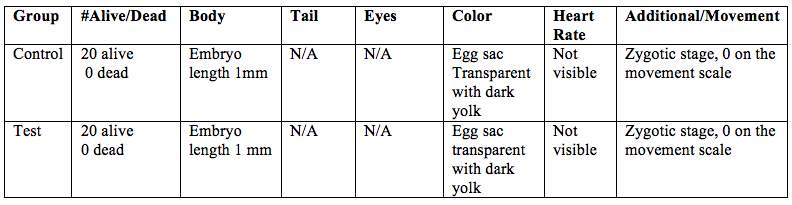
Table 1: Zebrafish observations, day 0, 2/18/15
Table 1 shows observations for the control and test groups on the initial day of the experiment. Criteria observed were number of alive embryos, dead embryos, body observations, tail observations, eye observations, coloration heartbeat and mobility. On the initial day there are 20 alive embryos and 0 dead in both the control and test group. Tail and eyes were not observed becaue they were not present. The embryos in both the control and teset group were about 1mm in length. The eggs sacs in both the control and test group are transparent with a dark yolk center. There is no mobility yet, and thus a score of 0 on the movement scale. Heart rate could not be detected.
Zebrafish embryos, day 0, 2/18/15
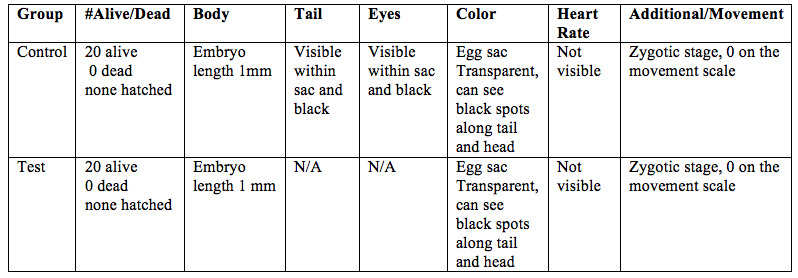
Table 2: Zebrafish observations, day 2, 2/20/15
On the second day of observation there were 20 alive embryos in both the control and test group. None of the eggs had hatched for either group. A heart rate was not able to be detected for either group. Although the fish were not hatched, forming tails and eyes could be seen through the transparent egg sac in both groups. The tails are spotted black and the eyes are black. The embryos still have no mobility becasue they are in the egg sacs. The egg sacs are about 1mm in both groups.
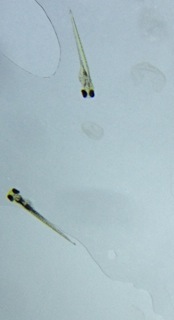
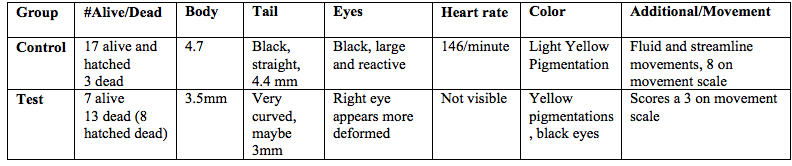
Table 3: Zebrafish observations, day 5, 2/23/15
On the 5th day of observation there were 17 live hatched fish in the control group, and 3 dead egg sacs. The live fish in the control group were about 2.7mm from tail to head. The tail takes up a large portion of the body at 2.3mm, and is straight and black. The bodies of the fish are transparent and spotted black. The eyes are large and black. The heart rate was approximately 140 beats per minute. They have begun to swim and score a 5 on the movement scale as they swim away when touched with a dropper, but not consistently. In the test group there were 15 live hatched fish and 5 dead egg sacs. The fish were about 2.5mm from tail to head. The tail was pretty straight with a possibly hooked end. The eyes are black and appear slightly smaller than the control. The bodies of the test fish are a light yellow with black spots. They score a 4 on the movement scale because they do respond to touch by the dropper but twitch a bit as they swim away.
Test group, day 5, 2/23/15
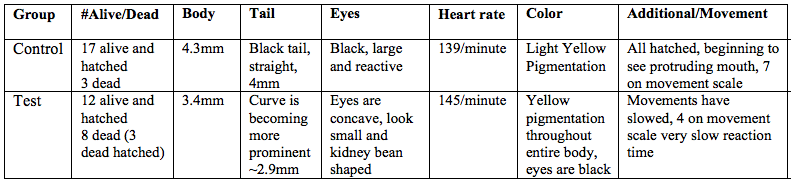
Table 4: Zebrafish observations, day 7, 2/25/15
On day 7 there were still 17 alive fish left in the control group. The body length increased to 4.3mm and was yellow. The tail was roughly 4mm long and is straight. The eyes remaining black and reactive. The protruding jaw is developing and can be seen protruding past the mouth. On Day 7 for the test group, 3 of the hatched fish had died, leaving 12 alive. The fish were beginning to have a yellow pigmentation with black spots along the tail and head and were about 3.4mm long. The heart rate was not measurable. The fish had very curved tails that were difficult to measure because of curvature. At this point, the fish slowly moved when touched with a dropper, twitching slightly.
Table 5: Zebrafish observations, day 9, 2/27/15
Day 9 had the same results as day 7 for the control aside from an increase in size to 4.7mm, tail to 4.4 and movement scale to 8. For the test on day 9, 7 fish were alive, 13 dead. About 3.5mm in body length,3mm of tail, deformed eyes and tail, 3 on movement scale.
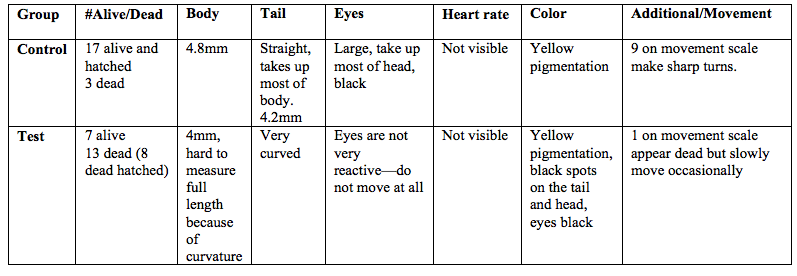
Table 6: Zebrafish observations, day 12, 3/2/15
On the 12th day there were 17 alive fish in the control group, about 4.8mm length with 4mm making up the tail. The fish are still yellow pigmented, and the eyes are still large and black. They score a 9 on the movement scale, as they react almost before the dropper tocuhs them, adn they are extremely hard to catch. On the 12th day 7 fish were still alive in the test group and a total of 13 dead. The fish were roughly 3.5mm in length. The living fish scored a 1 on movement scale. Eyes are black and concave looking and appear to be deformed, remained in a fixed position. The fish do not move often and are only occasionally twitching. They do not respond to the dropper anymore.
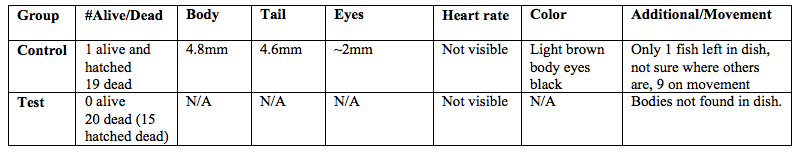
Table 7: Zebrafish observations, day 14, 3/4/15
On the final day only 1 alive fish was found alive. This fish was fixed and observed. The fixed fish was 4.4mm with 4mm of tail and a light brown color, 9 on movement scale. Its tail was straight and eyes were large and black. No signs of deformities. Heart rate was only visble for days 5-9 but remained between 139 and 146 beats per minute.On day 14 all the test fish were dead. There was no evidence of full fish in the dish although some pieces of a few decayed fish were found. We were not able to fix a test fish because there were none left, and unfortunately we messed up our fixation from the week before and the fish did not remain preserved.
Conclusions and Future Directions
The original hypothesis was that exposing Zebrafish embryos to Retinoic Acid during development would cause an increase of deformities among the fish. Results found are consistent with this hypothesis. In comparing results of the control and test groups, it becomes apparent that retinoic acid does have an effect on the development of zebrafsh most notably on the eye size, tail length and thus mobility of the fish. Retinoic acid serves as a signaling pathway during development and when exposed to too much or too little the subjects become deformed due to incorrect positional codes that specify the embryonic anteroposterioir body (Holland et al, 2006). The tail deformity lead to the lack of movement exhibited by the test fish in comparison to the control fish. The results also show that the test group had smaller, concave eyes. Similarly to the tail deformities current literature shows that retinoic acid serves as a signal pathway for the beginning of embryonic eye development (Cvekl and Wang, 2009). In this case, the over exposure to Retinoic acid resulted in deformed eyes that were the wrong size and shape. In order to improve the experimental design for next time, it could be interesting to compare various concentrations of retinoic acid with a control group. Since we only got to see a 20ml diluted concentration in this experiment, we could not see what more or less retinoic acid would do to the development of the fish. I think it would be interesting to see what would happen if the zebrafish were deprived of retinoic acid during development, since it is necessary for development. Next time I may want to set up various of concentrations of retinoid so that I can get a better idea about the effects of a higher or lower concentration of retinoic acid on the zebrafish.
References:
Herrmann, K. (1995). Teratogenicity Effects of Retinoic Acid and Related Substances on the Early Development of the Zebra fish. Toxic in Vitro, 9(3), 267-283
Holland, L. Laudet, V. Marletaz, F. Schubert, M. 2006. Retinoic acid signaling and the evolution of chordates. International Journal of Biological Sciences. 2:38-47
Cell, Ales. Wang, Wei-Lin. 2009. Retinoic acid signaling in eye development. Experimental Eye Research. 89:280-282
-RMT
2/27/15 PCR and 16s sequences The results from Lab 3's PCR reaction came back. Originally, there were two dishes with tetracycline and two without. 1 from each of the two plates was selected to amplify the 16s rRNA gene. This gene was being amplified because it is very diverse in different species. Below is a picture of the Gel Electrophoresis:
The first bacteria that came back from the PCR sequencing was Flavobacteria. Flavobacteria is a gram-negative bacteria that sometimes has motility and is rod shaped. It is typically found in soil and in fresh water. Flavobacterium is known to cause diseases in fish. This was not the bacteria that was originally thought to have been found, but it is gram-negative which was observed previous to sequencing.
The second bacteria found was Epilithonimonas, which is a gram-negative, non-motile, rod shaped bacteria. It is often found in soil and in costal water. This makes sense because the transect is composed of soil, and at the time of extraction the soil was wet due to snow.
-RMT
2/17/15 Lab 5:Invertebrates
1)THe Planaria moves forwards slowly sort of like a slug would. It glides and crawls. Planaria is a acoelomate, meaning that its simple digestive system doesn't have a coelom; this form effects the function of the invertebrates. It's simple structure is reflected in its movement because it moves very slowly, and lacks the ability to make sharp turns or go quickly.
Invertebrates with a pseudocoelomate structure have an incompletely lined body cavity, and Nematodes are an example of this. The nematodes move by sliding back and forth, a wriggling motion. The coelomate Annelida moves by contracting muscles causing the head to extend forwards. The Annedlida phyla have a fully lined fluid-filled coelomate, and the internal organs are placed in a way that they aren't harmed with movement. It appears as though the contracting muscles do not allow the annelid to move backwards.
The size range of organisms observed ranges from .8mm to 2mm. The largest organism measured was the beetle larvae, and the smallest organism measured was the soil mites. The most common organism found in the leaf litter was the soil mites while the least common found were springtail.
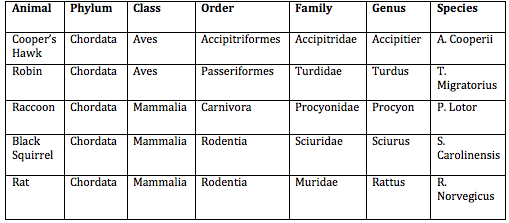
3) 5 Vertebrates that could be found in this transect:
The Hawk and the Raccoon would be at the top of the food web. The Hawk and the Raccoon would both depend on smaller animals as a source of food, and possibly some plants. The Hawk would need access to a tree for nesting, similarly to the robin. The raccoon would live in a den, so they would need to have a shelter of rocks, trees, or bushes available to them so that they could be safe. The robin would depend on small creatures such as worms or grasshoppers for food, and at the same time, the robin could be a perfect meal for a Hawk. The robin would need to be around trees as well, preferably away from predators as to keep hatching eggs safe. The squirrel and mouse would both be sources of food for the raccoon and the hawk, and in addition, the mouse would be food for the robin. The mouse and squirrel would need areas to live, such as in a tree (squirrel) or in a small den, similarly to the raccoons for the mouse. All of these animals would need to have access to water in order to survive. Bacteria and archaea would benefit all the species, because they would feed small invertebrates such as annelids that would in turn feed the squirrels and robins, and so on. Important factors could be dead leaves and grass, because the dead leaves and grass are primary producers, meaning they are the original source of the energy. Bacteria and archaea could feed on the dead leaves, while mice could feed on the grass.
These animals represent the ecological concept of community because they all live together in the same ecosystem, and maintain normality together. Within a food web, there are different trophic levels that track the energy flow through organisms. As the energy moves up the food chain, it decreases. At the bottom of the food chain are the primary producers, and they are always autotrophs that are able to produce their own energy. Heterotrophs from all the other trophic levels are dependent on autotrophs for energy, so even though they are at the bottom of the food chain, they are the most important. The next level is primary decomposer or consumer. Primary consumers feed on the autotrophs (such as the grasses and moss) while the primary decomposers feed on the detritus. In this case, the bacteria volvox, sarcina, and Hypha are the primary decomposers because they feed on the dead leaves. A secondary consumer in this system would be the robin, because it does not get it's food directly from the autotrophs--it gets it second. A tertiary consumer would be the Hawk or the Raccoon, because they are at the top of the food chain. In the decomposer food chain, the Hawk and the RAccoon would be quaternary consumers, because annelids would be the secondary consumer with the robin as the tertiary. Carrying Capacity is the number of individuals that an environment can support. When carrying capacity is exceeded, organisms begin to die out due to starvation, sickness, or predation. The food web is especially important because the preying of higher up organisms on lower organisms makes sure that the carrying capacity does not get exceeded. For instance, if suddenly there was no Cooper's Hawk, there might be a growth in the number of robins because they weren't getting preyed on as much. In turn, they would deplete more of the smaller animals such as the rats, and the rats may become a significantly smaller population. This is just an example, but it demonstrates that each trophic level is essentially important in maintaing the community and carrying capacity.
-RMT
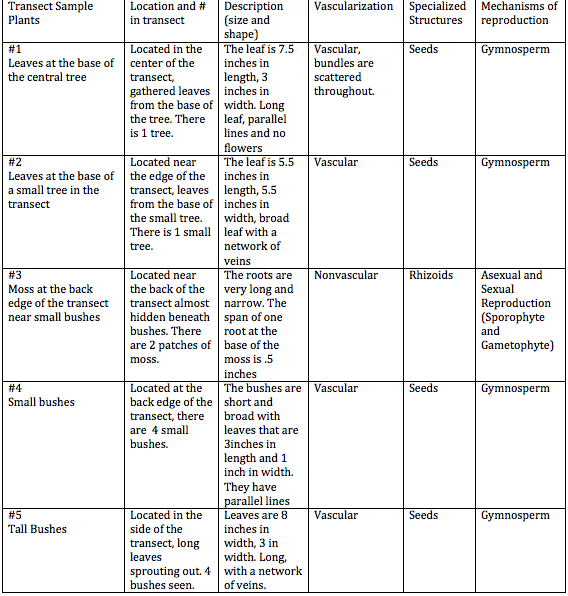
2/10/15 Lab 4:Plantae and Fungi
1)Within the transect we found five different plants, although this was difficult because there aren't many plants within our transect, and many of the plants appear to be of the same species.The plants that we found are listed in the table below.
2)There are no flowering plants in our transect.
3)The Fungi Sporangia have hyphae that grow upward, forming small dark globelike structures. Sporangia contain cells that are called spores, and these spores are released into the air when the sporangia open up. Sporangia are very important for reproduction of fungi. -RMT
2/3/15 Lab 3: Identifying and Studying Bacteria
Pre Procedure questions/observations: 1)I don't think that there will be any Archaea growing on the agar plates. This is because archaea tend to grow in extreme conditions, and the agar plate is not an extreme condition. 2)The Hay Infusion Culture smells even worse this week than it did last week, with a rotten decaying smell. The crusted top that we broke last week to take samples from the bottom has reformed and looks even thicker than it had previously. The water appears to be a darker brown than it was last week, as it is almost black in color; the plant matter in the middle is hardly visible. Hypothesis: The smell and coloration of the Hay infusion changes from week to week as more organisms begin to grow in the Hay Infusion.
Procedure 1: Table 1 100-fold Serial Dilutions Results
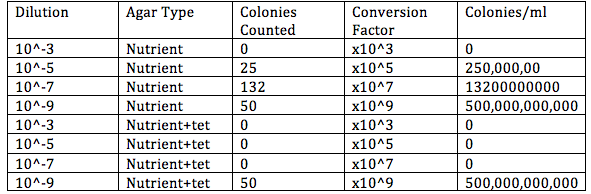
Procedure 2:Antibiotic Resistance 1)Most of the plates that have the antibiotics have no colonies growing in them. With the exception of the Nutrient+Tet dilation 10^-9 tray, the agar plates with tet have no growth. The plates without antibiotic have lots of visible colonies growing on them.This indicates that the bacteria on the tet plates that grew is naturally resistant to the antibiotic, while the plates with no growth were not. The plates without the antibiotic have growth because there was no tet introduced. Overall, the Tet decreased the amount of bacteria on each plate. Oddly, the tet plate with growth did not even show a diminished amount of growth, suggesting that the antibiotic did not effect that strain of bacteria at all. There was fungi on the tet plate with bacteria, which makes sense because an antibiotic would not wipe out a fungi; an anti-fungal would do that. Only one species of bacteria remains resistant to the antibiotic. 2) The mode of action for the antibiotic Tetracycline is to inhibit the protein synthesis in bacteria by preventing aminoacyl-tRNA from associating with the ribosome(Chopra and Roberts 2001.) Tetracycline is used on a variety of bacteria including gram positive and negative bacteria.
Procedure 3: Table 2 Bacteria Characterization
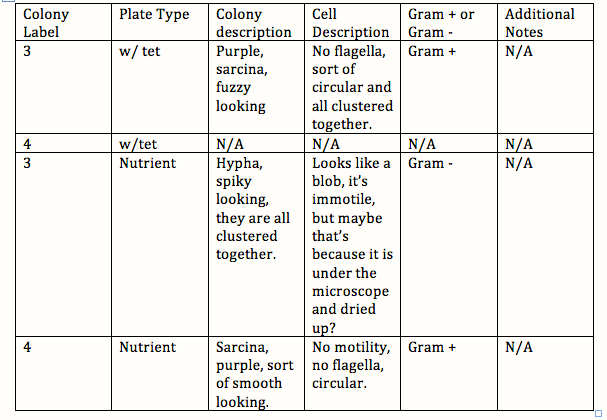
-RMT
Citations: Chopra, I., & Roberts, M. (2001, June 1). Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Retrieved February 3, 2015, from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC99026/ ;
1/29/15 Lab 2: Hay Infusion Culture Observations
Procedure 2: 1)The Hay infusion from the Wildlife Sanctuary has a rotten smell. On the top of the sanctuary there appears to be some mold. For microscopic observation, we took samples from two different locations, from the top near the pant matter and from the bottom. Organisms that are near the plant matter may differ from those that live away from the plant matter because they may feed on different organisms or have different mechanisms, such as being photosynthetic or not. We were only able to locate 4 organisms from our samples, 3 from the top of the hay infusion and 1 from the bottom. On the top, we found Paramecium Caudatum, Colpidum and Volvox. The Paramecium Caudatum is about 230 micrometers in length and is motile (cilia.) The paramecium does not appear to be photosynthesizing, especially with the lack of light that is making its way into the infusion. It is a paramecium. The Colpidum is 55 micrometers, and is motile (cilia.) It does not appear to be photosynthesizing, and it is a protozoa. The Volvox is about 400 micrometers. The Volvox is photosynthetic and is an algae. The volvox is motile and moves with a flagella. The only organism that we could find in the bottom of the Hay infusion was Colpidum, which we also found in the top of the Hay Infusion. There is little light reaching the bottom, and a very foul smell with thick waters so there may not be much life in the bottom.
2)The Volvox is a species that meets all the needs of life. The needs of life are as follows: They can acquire and use energy, they are made up of more than one cell, they are able to process information, they are capable of replication, and they are a product of evolution (Freeman.) The volvox is a type of green algae, meaning that it requires and uses energy through the means of photosynthesis. They are chloroplast containing. Each volvox is made up of 6000 individual cells on average, so it meets that criteria. Since the volvox is algae, and algae are eukaryotic that means that Volvox have nuclei, meaning that they are able to process information. Volvox can reproduce through asexual or sexual reproduction. Eventually, daughter colonies will break off of the parent colonies and form a new colony (Koufopanou.) Finally, the volvox comes from the the volvocine, which was examined earlier in lab. The volvocine line begins with the isogamous unicellular alga called Chlamydomonas, and eventually evolves into the volvox, which is the peak of evolution for this particular line (Bentley et al.) If the Hay infusion culture continued to grow for two months, I think there would be a decrease in life because of the lack of sunlight getting into the infusion. Species such as the volvox are photosynthetic,and thus wont be able to live long without access to sunlight. Sunlight would be the main selective pressure for my sample, although other prey and may be a selective pressure as well. Citations: Bentley, M., Laslo, M., Walters-Conte, K., & K. Zeller, N. (2014). Biological Life at AU. In A Laboratory Manual to accompany: General Biology 2 (1st ed., Vol. 1, pp. 14-16). Washington, D.C: Department of Biology American University. Freeman, S. (n.d.). Biology and the tree of life. In Biological science (Fifth ed., p. 2). Koufopanou, V. 1994. The evolution of soma in the Volvocales. Am. Nat. 143:907–31.
-RMT
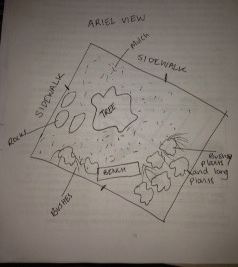
1/25/15 Lab 1:Hay Infusion and Transect
The Wildlife sanctuary is located between two sidewalks. The transect is primarily made up of soil and leafy bushes.In the center of the transect is a tree which is bare, and there are dead leaves on the ground. The soil is quite bare besides the dead leaves and a bit of snow. There is a wooden bench in the transect that seems to have been scraped at by an animal. It does not seem as though there is much foot traffic through the transect, so it is not very disturbed. It seems as though the wind has blown a few wrappers into the transect. Biotic factors within the transect are the tree, moss, small leafy bushes, grass and tall grassy bushes. The abiotic factors are rocks, soil, mulch,snow, wind and litter.
 -RMT
-RMT
1/25/15 This is Morgan Taylor, I've successfully submitted to OWW! -RMT