User:Noy Kaufman/Notebook/Biology 210 at AU
Invertebrates and Vertebrates July 14th, 2015
Purpose:
Invertebrates can be broken down into 14 different phyla and comprise 95% of the known animal species (Freeman, Quillin, Allison, 2014). The complexity of animals can be characterizes by radial symmetry and bilateral symmetry. Radial symmetry means that the animal is in a sphere shape, has a top and a bottom, but not a front (Bentley, Walters-Conte, Zeller, 2015). Bilateral symmetry on the other hand means that the animal has head and tail and has left and right sides (Bentley, Walters-Conte, Zeller, 2015). Internal tissues and organs evolved and developed from the bilateral symmetry; there are three germ layers: endoderm, ectoderm, and mesoderm (Bentley, Walters-Conte, Zeller, 2015). Vertebrates are animal with a bony structure that support the dorsal side of the body, called vertebrae, and a cranium, which is a bony case that enclose the brain (Freeman, Quillin, Allison, 2014). This lab will study invertebrates found using the Berlese Funnel and vertebrates in the transect.
Methods:
Different known organisms were observed under the microscope. The Berlese Funnel was then separated and the top 15mLs of liquid and organisms were poured into a petri dish. The remaining was poured into a second dish. Both plates were examined under a microscope and different organisms were found. The organisms were identified using a key and a website. the organisms were measured and examined. Then the transect was observed and five vertebrates were identified (Bentley, Walters-Conte, Zeller, 2015).
Results:
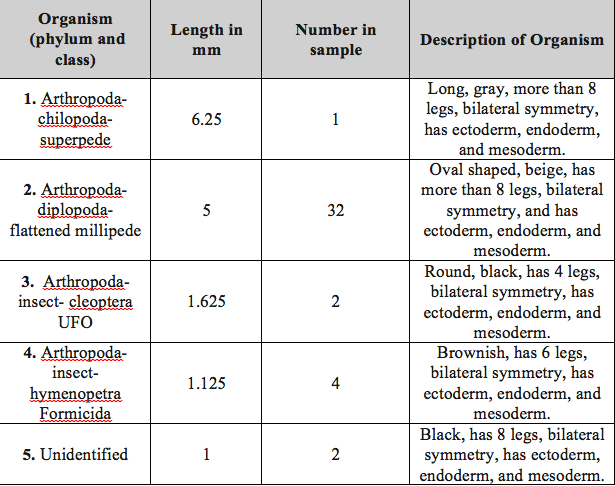 Table 1: identified organisms characteristics
Table 1: identified organisms characteristics
[[[Image:Unidentified.JPG]] Figure 5: unidentified organism
Table 2: vertebrates found in the transect. Classification and survival factors
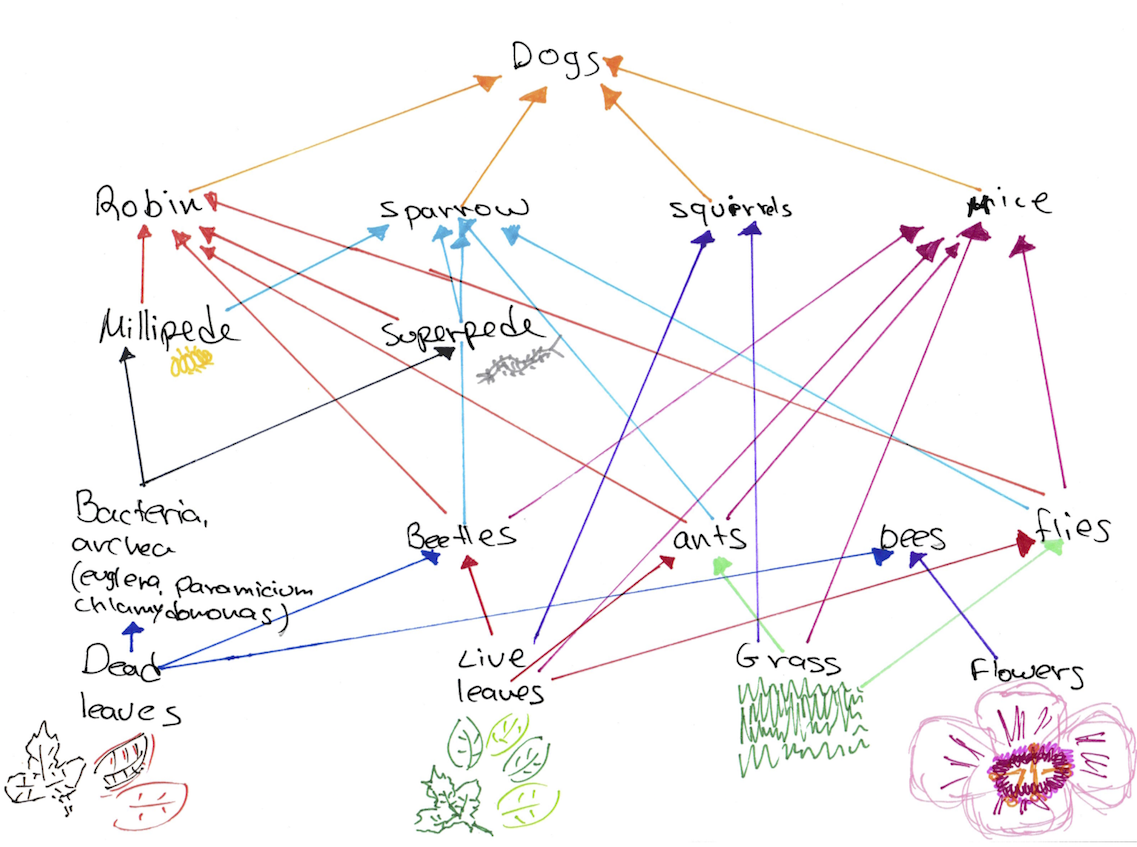
Figure 6: Food web of the organisms found in the transect
Discussion:
The invertebrates found on the transect varied in size and shape.The size rage was 1mm to 6.25mm. The smallest organism measured was the unidentified organism and the largest organism measured was the Arthropoda- chilopoda- superpede. The most common type of organisms that was present in the leaf litter is the Arthopoda phylum, in particular the millipedes. Figure 6 presents a food web based on all of the organisms found in the transect during the labs. This food web presents the complex relationships between the different organisms that coexists in the transect, which is their ecosystem. As a part of this ecosystem, and the different communities and niches within it, these organisms share the same resources with regard to the carrying capacity. The carrying capacity is the maximum number of organisms that can live in a particular area (Bentley, Walters-Conte, Zeller, 2015). When there are more organisms than maximum capacity, there are not enough resources, and some organisms will die. The trophic levels of organisms describe the position of the organism in the food chain and the source of energy that they consume (Freeman, Quillin, Allison, 2014). All of the organisms present in the transect are required for the survival of each other, creating a community with the necessary environmental conditions and food sources.
References
“American Robin” Wikipedia, The Free Encyclopedia. 25 Jun. 2015. Wikipedia. (17 Jul. 2015).
Bentley, M., Walters-Conte, K., Zeller, N. 2015. A Laboratory Manual to Accompany General Biology II. American University Department of Biology.
“Dog” Wikipedia, The Free Encyclopedia. 17 Feb. 2015. Wikipedia. (17 Jul. 2015).
“Eastern Gray Squirrel” Wikipedia, The Free Encyclopedia. 25 Jun. 2015. Wikipedia. (17 Jul. 2015).
Freeman, S., Quillin, K., Allison, L. 2014. Biological science (5th ed.). Pearson: Glenview, IL. 1150.
“House Sparrow” Wikipedia, The Free Encyclopedia. 16 Jan. 2015. Wikipedia. (17 Jul. 2015).
“Mouse” Wikipedia, The Free Encyclopedia. 18 Apr. 2014. Wikipedia. (17 Jul. 2015).
N.K
Plantae and Fungi July 9th, 2015
Purpose:
Plants and fungi are crucial to and have an important role in sustaining life on Earth. While plants consume carbon dioxide to release oxygen, which is essential for living organisms, fungi act as decomposers and release carbon dioxide and nitrogen, which will be used by the plants as a nutrient (Bentley, Walters-Conte, Zeller, 2015). There is a strong evidence that plants have evolved from aquatic green algae to land plants. The first land plant was Bryophytes, which is a non vascular plant. The terrestrial organisms have evolved since then and developed vascular systems, seeds, flowers, pollen, and different reproductive systems (Bentley, Walters-Conte, Zeller, 2015). This lab will analyze and examine different plants and their characteristic and structures.
Methods:
A Ziploc bag was filled with leaf litter from an area with soft soil and dead leaves in the transect. Then, five different plants were collected from different locations in the transect. The collected plants were identified, examined and the different characteristics were recorded in a table. The Berlese funnel was then prepared. 25mL of 50:50 ethanol/water solution were added to a 50 mL tube. A piece of screening material was taped to the bottom of a funnel. The tube was taped to the end of the funnel. The leaf litter was then placed inside the funnel. The funnel was attached to a ring stand and both were placed under a 40 watt lamp and everything was covered with foil (Bentley, Walters-Conte, Zeller, 2015)
Results:
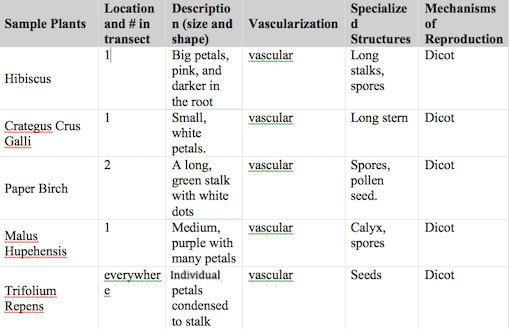 Table 1: Characteristics of plants collected from the transect
Table 1: Characteristics of plants collected from the transect
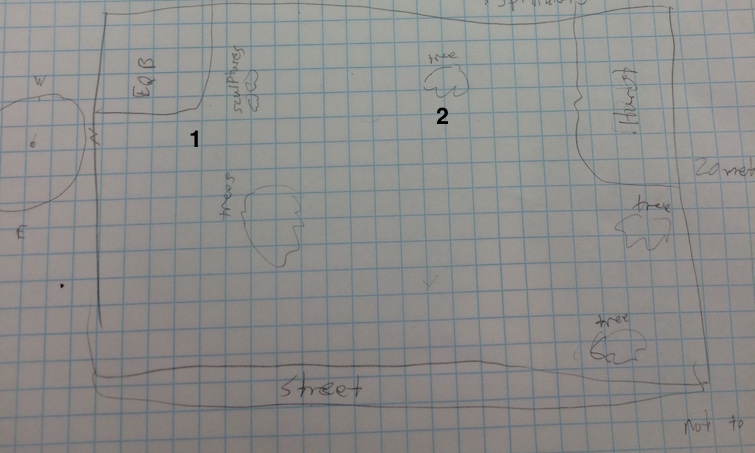 Figure 1: a map of the transect with location numbers.
Figure 1: a map of the transect with location numbers.

Discussion:
Table 1 presents the different characteristics of the plants collected from the transect. It can be seen that all of the plants have vascular system ad that all have a dicot stricture of reproduction. Some of the plants were only found in one location, while others, such as the Trillium reopens are all over the transect. The diversification of the plants was observed and discussed. This lab contributed to the understanding of how plants evolved to have vascular systems and the diversity of plants.
References:
Bentley, M., Walters-Conte, K., Zeller, N. 2015. A Laboratory Manual to Accompany General Biology II. American University Department of Biology.
N.K
Microbiology and Identifying Bacteria with DNA Sequences July 7th, 2015
Purpose:
There are three Domains of life, which are Archaea, Bacteria, and Eukarya; all include unicellular organisms (Bentley, Walters-Conte, Zeller, 2015). The Bacteria Domain consist of the prokaryote groups Proteobacteria, Chlamydiae, Spirochetes, Actinobacteria, Firmicutes, and photosynthesizing Cyanobacteria (Bentley, Walters-Conte, Zeller, 2015). This lab will study prokaryotes in the Domain Bacteria and from the group specified above. Cell morphology is one way to classify microorganisms, in particular, staining techniques to examine unicellular organisms. Thee basic bacterial cell shapes have been identified using these techniques: bacillus is rod-shaped, coccus is spherical, and spirillum is a twisted spiral (Bentley, Walters-Conte, Zeller, 2015). In this lab, the Gram test was used to identify Bacteria.
Methods:
The Hay infusion was observed and examined for any changes. The agar plates from last week were retrieved and the growth was observed and the colony morphology was determined and recorded in a table. The number of colonies in each plate was counted and recorded. Four colonies, two from the nutrient agar and two from the tet+ agar were chosen. A small amount of each of the four growths was scrapped using a sterile loop and placed on four different slides. A drop of water was placed on each slide and mixed. The area under the sample was circled and the slide was labeled. The slide was then fixed by passing it through a flame several times. The slides were placed on the staining tray and the bacterial smear was covered with the crystal violet solution for one minute. The stain was rinsed off using DI water. The bacterial smear was then covered with Gram's iodine mordant for one minute. The stain was rinsed off using DI water. The slides were then decolorized using 95% alcohol for 10-20 seconds and rinsed with DI water. the bacterial smear was then covered with safranin stain for 20-30 secondhand rinsed off with DI water. the slides were allowed to dry and the stained samples were observed under the microscope and the morphology was recorded. Last, one nutrient plate and one tet+ plate were chosen and a single colony from each was added to tube containing the primer, water and 16S PCR (Bentley, Walters-Conte, Zeller, 2015).
Results:
 Figure 1: top view of Hay infusion
Figure 1: top view of Hay infusion
 Figure 2: side view of Hay infusion
Figure 2: side view of Hay infusion
The smell of the Hay infusion is still pretty foul, however, the mold on the surface disappeared and it seems like the plant material in the bottom has grown. The appearance ad smell of the Hay infusion have changed and would probably change every week due to natural selection and the different microorganisms that are currently living in the infusion. The current communities are different than the parental ones since they adjusted to the new conditions of the environment. I believe the Archaea organisms will not grow on the agar plates due to the fact that Archaea are mostly found in extreme conditions which are not present on our Hay infusion.
 Figure 3: Nutrient + tet agar plates
Figure 3: Nutrient + tet agar plates
 Figure 4: Nutrient agar plates
Figure 4: Nutrient agar plates
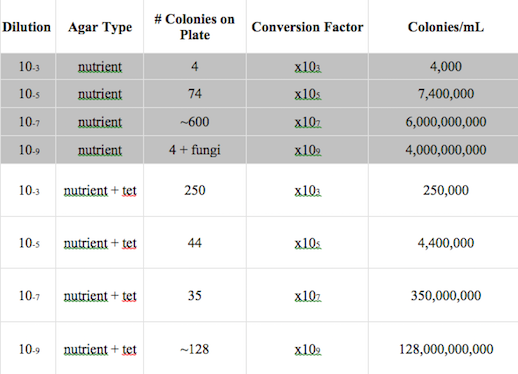 Table 1: 100-fold Serial Dilutions Results
Table 1: 100-fold Serial Dilutions Results
 Table 2: Bacterial Characterization
Table 2: Bacterial Characterization
 Figure 5: drawing of organisms, including magnification.
Figure 5: drawing of organisms, including magnification.
Discussion:
It can be seen from figures 3 and 4 that there are differences in the colony types between the plates with versus without antibiotics. Figure 4 presents the nutrient plate, which had growth of mostly white colonies and one with a growth of fungi. On the other hand, figure 3 presents the nutrient plate that contained antibiotics, which had a growth of mostly orange colonies. This indicates that the type of bacteria that grow in the orange colonies in resistant to the tetracycline antibiotic. The tetracycline did not reduced the number of colonies dramatically, however, it prevented the growth of fungi. The mechanism of action of tetracycline works in a way that inhibits the synthesis of a bacterial protein by preventing aminoacyl - tRNA from entering the bacterial ribosome (Chopra I, Hawkey PM, Hinton M 1992). The types of bacteria that are sensitive to tetracycline are gram-positive and gram-negative bacteria, chlamydiae, mycoplasmas, rickettsiae, and protozoan parasites (Chopra, I., & Roberts, M. 2001). The Gram test results presented two organisms that are positive, however, most of the samples were negative for the test.
References
Bentley, M., Walters-Conte, K., Zeller, N. 2015. A Laboratory Manual to Accompany General Biology II. American University Department of Biology.
Chopra, I., & Roberts, M. (2001). Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiology and Molecular Biology Reviews, 65(2), 232–260. doi:10.1128/MMBR.65.2.232-260.2001
Chopra I, Hawkey PM, Hinton M. J. (1992). Tetracyclines, Molecular and Clinical Aspects. Antimicrob Chemother, 29(3):245-77.
N.K
IDENTIFYING ALGAE AND PROTISTS July 2nd, 2015
Purpose:
Scientists began classifying organism that are visible to the naked eye into groups, however, after the microscope was invented, scientists were able to observe microscopic organisms and identify and classify them into different groups. There are two major group that will be discusses in this course: prokaryotes and eukaryotes. Prokaryotes are organisms that lack member bound organelles or nucleus and eukaryotes are organisms with a membrane bound organelles and nuclei (Bentley, Walters-Conte, Zeller, 2015). Each of these groups are made up of different lineages. The purpose of this lab is to identify organisms in a sample from the Hay infusion using the Dichotomous keys.
Methods:
The jar containing the Hay infusion was retrieved and its characteristics, such as smell were recorded. Two samples were taken from two different niches; one sample from the surface of the liquid and another sample was taken from the bottom of the jar near a plant. Two wet mounts of each samples were prepared and observed under the microscope. Three organisms were identified using the Dichotomous key. The identified organisms were then recorded and measured. Next, serial dilutions for the next lab were prepared. Four tubes containing 5 mLs of series broth were labeled 10^-2, 10^-4, 10^-6, and 10^-8. The Hay infusion jar was mixed and 50μL were retrieved and added to the 10^-2 tube; this is a 1:100 dilution. The tube was mixed. 50μL from the 10^-2 tube were retrieved and added to the 10^-4 tube; this is a 1:10000 dilution. This procedure was repeated for the 10^-6 and 10^-8 tubes. Four nutrient plates were labeled 10^-3, 10^-5, 10^-7, and 10^-9. 100μL of tube 10^-2 content were added to the 10^-3 plate, 100μL of tube 10^-4 content were added to the 10^-5 plate, 100μL of tube 10^-6 content were added to the 10^-7 plate, 100μL of tube 10^-8 content were added to the 10^-9 plate. The samples were spread on the plate using a sterile stick. The same procedure was repeated for four tet+ plates(starting with the 10^-9 dilution) (Bentley, Walters-Conte, Zeller, 2015).
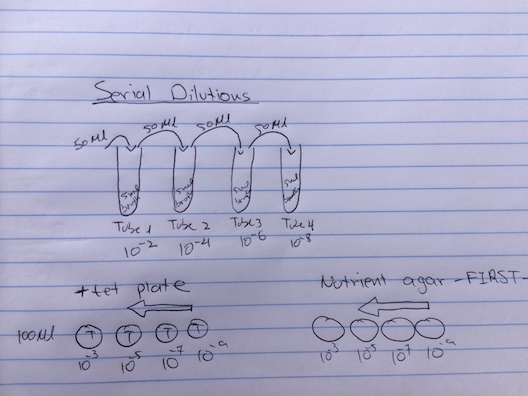 Figure 1: serial dilutions diagram
Figure 1: serial dilutions diagram
Results and Observations:
The Hay infusion: the culture had a foul, mold smell. There were observable mold brown spots on the surface of the liquid. About 3/4 of the mixture was cloudy and dirty liquid and 1/4 was a thick layer of dirt in the bottom of the jar, which also contains a small plant.
Top sample, surface of the liquid:
Euglena (protozoa): measured to be 40 micrometer. This is a small, green organism, very motile due to its flagellum and is photosynthesizing.
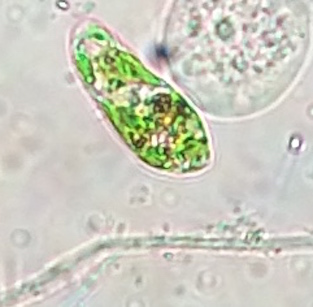 Figure 2: Euglena observed under microscope in 10X.
Figure 2: Euglena observed under microscope in 10X.
Chlamydomonas (protozoa): measured to be 7 micrometers. This is a very small organism, it is colorless and is motile due to its two flagella. It is photosynthesizing.
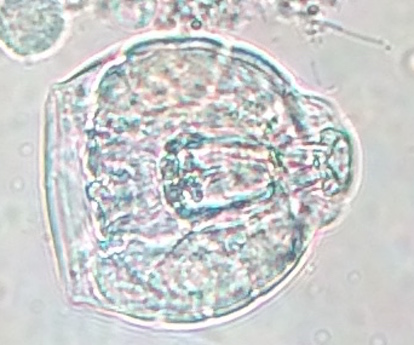 Figure 3: Chlamydomonas observed under microscope in 40X.
Figure 3: Chlamydomonas observed under microscope in 40X.
Didinium (protozoa): measured to be 20 micrometers. It is colorless with two visible bands, a darker one and a clear one. It is not motile and non photosynthesizing. Was present in high quantities.
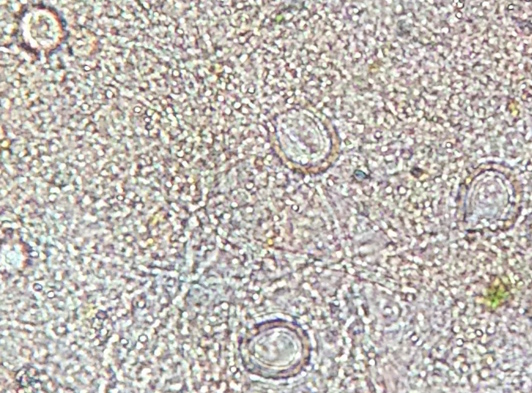 Figure 4: Didinium observed under the microscope in 10X.
Figure 4: Didinium observed under the microscope in 10X.
No organisms were found in the second sample from the bottom layer of the Hay infusion.
If the Hay infusion culture grew for two more months, I would expect an increase in the number of organisms on the surface and an increased growth of mold. This is due to the fact that the organisms on the surface can get light and oxygen more easily than the ones in the bottom of the culture. The organisms in the bottom of the culture will not get the oxygen and nutrients needed for their development and they will die. Moreover, since the jar is open, new organisms are able to enter the culture reproduce, as a result it is possible that a new ecosystem will be created. Some factors that could affect the community are: lack of oxygen, lack of sunlight, temperature of the room, new organisms introduced to the community, sold surface on top of the liquid, lack of nutrients and fresh water.
Discussion:
The culture has low variability in terms of organisms. The only organisms found were only protozoa and were found on the surface of the culture. Other organisms were identified and most of them turned out to be the same as the ones we identifies before. The sample from the bottom of the culture containing the dirt and plant, did not have any identifiable organisms. This could be due to the fact that organisms that are close to the plan matter might differ from the ones on the surface and have different environmental conditions that were not met in this culture and therefore, they were not able to survive. As a result of the low variability, only three organisms were identified instead of four.
The Euglena found on the surface of the culture fulfill the five requirements of life. 1. Obtaining energy: the Euglena is photosynthesizing organism and therefore obtains is energy from the sunlight. 2. Comprised of cells: the Euglena is a unicellular organism, it is a eukaryote and the same as all living organisms, is comprised of cells. 3. Information: Euglena contains the genetic material in its nucleus. The information in the nucleus is processed to eventually produce the proteins required for the function of the organism. 4. Replication: Euglena reproduce asexually (Freeman, Quillin, Allison, 2014). Reproduction was not observed during the lab. 5. Evolution: this is a very long process that obviously cannot be observed under the microscope. However, all organisms are the product of evolution and Euglena has evolved to its current structure.
References
Bentley, M., Walters-Conte, K., Zeller, N. 2015. A Laboratory Manual to Accompany General Biology II. American University Department of Biology.
Freeman, S., Quillin, K., Allison, L. 2014. Biological science (5th ed.). Pearson: Glenview, IL. 2
N.K
EXAMINING BIOLOGICAL LIFE AT AU June 30th, 2015
Purpose:
Life on earth has evolved to be very diverse. Gene flow, gene drift, and mutations are some modes of natural selection that contributed to the biodiversity and to the expending of certain communities. Large communities, or ecosystems, contain both living organisms (biotic) and non living organisms (abiotic), all of which contribute and affect the structure and characteristics of the ecosystem (Bentley, Walters-Conte, Zeller, 2015). In this lab, a sample from a selected ecosystem, also called a transect, will be analyzed and studied. The different communities within the transect inhibit different niches with different environmental conditions and requirements that are best fit for the specific community.
Methods:
Groups of four people were assigned and each group chose their own 20 X 20 meters transect in the AU campus. My group chose a transect behind Hurst Hall. Once the transect was examined thoroughly, the biotic and abiotic organisms were listed and a detailed map was created and pictures of the transect were taken. A sample containing soil and vegetation was retrieved from a representative area of the transect and placed in a plastic bag. 12 grams of the sample were placed in a plastic jar along with 500 mLs of deerpark water. 0.1 grams of dried milk were added to the mixture and the jar was thoroughly mixed for 10 seconds. The jar and lid were labeled and the jar was left open in the lab for three days until the next lab.
Results:
Our transect is located in the space behind the EQB building and Hurst Hall. The transect was mostly composed of grass and some trees; some biotic organisms that were present in the transect are: grass, trees, flowers, fungi, bees, flies, worms, birds, and squirrels. Abiotic factors included: soil, water, air, light, statues, lams, and sprinklers. Since the transect is located in the borders of the campus, there is a sidewalk and a road right next to it, so there is some human interactions and some traffic around the area.
 Figure 1: map of the transect
Figure 1: map of the transect
 Figure 2: the transect
Figure 2: the transect
 Figure 3: te transect
Figure 3: te transect
Discussion:
This experiment was an introduction to the labs to follow. The transect was carefully chosen to meet the requirements and to give a good representation of possible species communities within the AU campus. This transect will be the basis for future experiments regarding the different habitats in it. The Hay infusion prepared will play a role of a new ecosystem for the sample retrieved from the transect. By letting the organisms grow in the new Hay infusion environment we can study the different environmental requirements and conditions of the organisms.
References:
Bentley, M., Walters-Conte, K., Zeller, N. 2015. A Laboratory Manual to Accompany General Biology II. American University Department of Biology
N.K